Low Level Radiation and Living State EDITORS N.G. Huilgol D.V. Gopinath B.B. Singh Narosa Publishing House New Delhi Madras Bombay Calcutta EDITORS N,.G. Hullgol Chief, Division of Radiation Oncology Dr. B alabhai Nanavati Hospital & Medical Research Centre Bombay, INDIA D.V. Gopinath Director, Health Safety & Biomedical Group Bhabha Atomic Research Centre Bombay, INDIA B.B. Singh Head, Radiation Biology & Biochemistry Bhabha Atomic Research Centre Bombay, INDIA Copyright O 1994
Low Level Radiation and Living State
EDITORS
N.G. Huilgol
D.V. Gopinath
B.B. Singh
Narosa Publishing House
New Delhi Madras Bombay Calcutta
EDITORS
N,.G. Hullgol
Chief, Division of Radiation Oncology
Dr. B alabhai Nanavati Hospital & Medical Research Centre Bombay, INDIA
D.V. Gopinath
Director, Health Safety & Biomedical Group Bhabha Atomic Research Centre
Bombay, INDIA
B.B. Singh
Head, Radiation Biology & Biochemistry Bhabha Atomic Research Centre Bombay, INDIA
Copyright O 1994 Narosa Publishing House
N A R O S A P U B L 1 S H I N G H O U S E
6 Community Centre, Panchsheel Park, New Delhi 110 017
35-36 Greams Road, Thousand Lights, Madras 600 006
306 Shiv Centre, DBC Sector 17, KU Bazar PO, New Bombay 400 705 2F—2G Shivam Chambers, 53 Syed Amir Ali Avenue, Calcutta 700 019
All rights reserved. No part of this publication may be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without the prior permission of the publisher.
Exclusive distribution in North American (including Mexico), Canada and Europe by Springer’-Verlag Berlin Heidelberg New York
All export rights for this book vest exclusively with Narosa Publishing House. Unauthorised export is violation of Copyright Law and is subject to legal action.
Published by N.K. Mehra for Narosa Publishing House, 6 Community Centre, Panchsheel Park, New Delhi 110 017 and printed at Rajkamal Electric Press, Delhi 110 033 (India).
Preface
The web of our life is of a mingled yarn, good and ill together.
—William Shakespeare
Chemobyl accident naturally fueled animated debate on consequences of low level radiation and biological systems. This controversial topic continues to attract research workers and increasingly, researchers from the field of molecular biology. The reductionist approach of molecùlar biologists in recent years seems to have obscured earlier holistic approaches.
This book is a departure from this trend wherein scientists from different areas of research related to low level radiation have contributed.
This book covers epidemeological survey of high background areas of India and China, assessment of livestock of same area, in vitro experiments to study genetic consequences, changes in immunity following radiation and adaptive response. Chemobyl data has been revisited with a fresh perspective. Biological rationale for recommendations of goveming bodies and mechanism of risk estimates have been highlighted. Radiation induced carcinogenesis has been assessed on the basis of recent data. The effects of radiation is a mingled yann, good and ill together which we hope has come out through this book.
The editors thank the contributors and also Dr. B.S. Rao of BARC, Mrs. S. Savithri, Mrs. N. Chatterjee and Mrs. Nilamani Huilgol who helped with the manuscripts.
N.G. HUILGOL
D.V. Gopinath
B.B. SINGH
Contents
Preface
- Radiation Induced Cancer 1
1.W. Stather
- Environmental Radiation and Human Cancer 16
D.M. Taylor
- The Effects of Prenatal Irradiation on Carcinogenesis 24
- Einhorn
- Genetic Effects of Radiation and Offspring Cancer 40
- Nomura
- Consequences of Low Level Radiation A review 57
E.B. Burlakova
- International Assessment of Radiological Consequences
of Chernobyl Accident 63
D.V. Gopinath
- Investigation on Dose Assessment and Cancer Mortality
in High Background Radiation Areas of Yang jiang, China 69
- Luxin, Z. Yongru, T. Zufan, H. Weihui,
- Dequing and r. Yongling
- Cancer Risk of the Indian Population from Low Level
Radiation, Projected from the Revised ICRP Risk 77
Coefficients
Y.S. Mayya, P.V. Joshi and K.S.V. Nambi
- Study of Population Exposed to Natural Background
Radiation in Kerala, India 85
M.K. Nair, N. Sreedevi Amma, P. Gangadharan,
- Sankaranarayanan, T.P. Ramachandran,
- Jayalekshmyand K.S. Mani
- Effects of Low Dose X-ray Irradiation on Autologous
Tumour Killing System 93
- Uchida, Kariya, T. Takashi and K. Sugie
viii Contents
- Changesin Subpopulations of Lymphocytes after Exposure to lonizing Radiation in vitro and in vivo
- Dehos, Kriehuber, H. Czempiel, I. Baumgartner,
- Egblomasseand W. Burkart
- Adaptive ResponseStudies in Diploid Yeast Saccharomyces cerevisiae Exposed to HTO Beta and Gamma Radiation
- Rao, K.B. Aiijaria and N. Saiikara Narayanan
- BiologicalBasis of Radiation Protection Standards (ICRP-60)
S.D. Soman and B.S. Rao
- BiomedicalEffects of lonizing Radiation
K.S. Parthasarathy
- Studieson Human Populations Living in Natural High Background Radiation Areas of Kerala: A review
K.V. Aravindan and S.K. Mahajan
- CongenitalDefects and Post-Implementation Mortality in the Offspring of Gamma Irradiated Male Mice
- Bhattacharjee
- DoseRate Effect for the Induction of Genetic Damage in Diploid Yeast Cells Exposed to Gamma or
Beta-Radiation
B.S. Rao, N. Sankara Narayanan and K.B. Anjaria
- MicronueleatedErythrocytes for Low-Dose Effects of lonizing Radiation in the Bone Marrow of Mice: Role of sample size
H.N. Bhilwade, R.C. Chaubey and P.S. Chauhan
- AComputer Software Package for Determining Statistical Significance of Radiation Effect in High Background Radiation Area
M.B. Yadav, S. I yothi and H. Singh
- Health ImpactAssessment of Indoor Radon Levels in Some High Background Areas in India
M.C. Subba Ramu
1. Radiation Induced Cancer
J.W. Stather
National Radiological Protection Board, Chilton, Didcot, UK
Introduction
Repeated and large doses of radiation had been found to cause cancer of the skin within a few years of Roentgen’s discovery of X-rays in 1895. Shortly after this the widespread use of penetrating X-rays and radium in treating disease led to the recognition of a cancer risk in many organs and tissues following high radiation doses which caused gross tissue damage. There was, however, a delay of about 40 years before it became clear that there was a risk of radiation-induced cancer from irradiation at lower doses and that there is no apparent threshold dose below which exposure to radiation can be considered safe. This delay can be attributed to the fact that radiation- induced cancers do not differ in any known way from those occurring naturally or caused by other agents. It is now believed that any radiation dose is capable of inducing cancer and that the probability of its occurrence, but not its severity, depends on the radiation dose. In radiation protection terminology it is termed a “stochastic effect”.
The detection of radiation-induced cancers in exposed groups is more straightforward for tumours which are normally rare, especially if the irradiation is due to local concentrations of inhaled or ingested radioactive substances in certain tissues of the body. Thus, an excess of bone cancer in luminisers in the USA who had ingested radium-226 was found about 50 years ago. With increased knowledge of the effects of radiation attention has been progressively focussed on the effects of lower dose rates. As the numbers necessary to detect a given excess rate are, however, approximately proportional to the inverse square of the dose delivered, it has become necessary to study larger groups of irradiated individuals. Thus for most organs, evidence of the risk of radiation-induced cancer and its relationship to radiation dose has depended on prolonged and accurate medical surveillance of large groups of people who have been exposed in the past to known amounts of external radiation or internally incorporated radionuclides.
The development of risk coefficients for radiation-induced cancer, both for the working population and for members of the public are described in this paper, together with some information on the risk to the foetus.
Radiation-cancer
Cancer is generally understood to develop in a number of stages. That is, for malignancies to be expressed, a series of events must occur in cells and the rate at which they occur is thought to be reflected in the way cancers appear in the population over the course of time. It is not yet feasible to say which stages in carcinogenesis are affected by radiation, whether more than one stage is affected or whether a multistage model is able to fully explain the actual process. It may even be that events postulated at the cellular or subcellular level cannot be easily related to the clinical data on radiation carcinogenesis because of the influence of hormonal, immunological or other host factors.
A limited number of genes, known as oncogenes, have been implicated in the malignant transformation of normal cells. The precise ways in which these oncogenes can be activated by radiation are not known, but so far data have not revealed any modifications that would suggest radiation plays a special role in inducing cancer or that would help to differentiate, at the genetic level, radiation-induced tumours from tumours induced by other carcinogens.
If any induced DNA damage involving strand breaks occurs as a result of exposure to ionising radiations it is usually repaired with high fidelity, returning the DNA structure to its original form. However, repair processes may occasionally be error-prone in that overall DNA integrity is retained but small base sequence changes (point mutations) at the site of the initial lesions occur, or there could be even more gross changes such as gene deletions or rearrangements involving hundreds of bases. These misrepair events may occur in regions containing growth regulating genes (e.g., those thought to be implicated in the initial stages of carcinogenesis), resulting in stable genetic damage in surviving cells. It is on this theoretical probabilistic basis that ICRP assumes no dose threshold for the induction of cancer, an assumption generally supported by in vitro cellular studies.
Much information on carcinogenesis has been derived from the study of the effects of carcinogenic chemicals. Chemicals are often classified into those which are required to initiate the process of carcinogenesis and those which promote it. Initiators cannot themselves produce tumours whereas promoters are only functional on previously initiated cells. This is one reason why carcinogenesis is often regarded as a multistage process. Where radiation carcinogenesis is concerned there is no such clear distinction. Continued exposure to radiation throughout the latent period is not necessary for the development of a radiation induced tumour. Thus it would appear that radiation can act as both initiator and promoter in a single step since, as far as we know, exposure to radiation, however brief, can give rise to tumours which manifest themselves many years later. Expression of the tumour may, however, be influenced by many environmental or host factors such as cigarette smoke, hormones, etc.
Radiation is capable of causing tumours in nearly all tissues of the body, although the frequency of appearance following a unit dose may vary markedly from one tissue to another. Information on the dose related frequency of tumour induction by radiation is gained through follow up of groups of persons exposed to radiation. The observed tumour frequency can then be compared with an age and sex matched control group, not exposed to radiation, to determine the increase in frequency due to radiation exposure.
Tumors induced by radiation are in general indistinguishable from those occurring spontaneously and since cancer is not uncommon (one in five die as a result of it), the problem of determining a relatively small excess due to radiation is difficult. In general large exposed populations are necessary to obtain statistically meaningful results.
The chief sources of information on the risks of radiation-induced cancer are the A bomb survivors exposed to whole-body irradiation in Hiroshima and Nagasaki, patients with ankylosing spondylitis and other patients who are exposed to partial-body irradiation therapeutically, either from external radiation or internally incorporated radionuclides, and various occupationally exposed populations, such as uranium miners and radium-dial painters.
There is always a minimum period of time between irradiation and the appearance of a radiation-induced tumour. This period is termed the latent period and its length varies with age and from one tumour to another. Some types of leukaemia and bone cancer have latent periods of only a few years but many solid tumours have latent periods of ten or more years. For leukaemia and bone cancer there is fairly good evidence that the risk is completely expressed within about twenty five years following exposure. For tumours of longer latency, it is not yet clear whether the incidence of these tumours passes through a maximum and declines with time following exposure or whether the risk levels out or alternatively increases indefinitely during the remainder of life.
To project the overall cancer risk for an exposed population, it is therefore necessary to use models that extrapolate over time the data based on only a limited period of the lives of the individuals. Two such projection models have generally been used:
- Theadditive (absolute) risk model which postulates that radiation will induce cancer independently of the spontaneous rate after a period of latency, variations in risk may occur due to sex and age at
- The multiplicative(relative) risk model in which the excess (after latency) is given by a constant factor applied to the age dependent incidence of natural cancers in the
In most cases this spontaneous risk increases with age and therefore the multiplicative model will predict an increasing incidence of cancer with increasing age. The relative risk model also gives different risks of radiation- induced cancer in different populations, depending on the national cancer incidence. Data are now available from the A-bomb survivors in Japan and from studies on uranium miners that suggest the multiplicative projection model currently gives a better fit to the data, at least for some of the most common cancer types [24] (Table l). Despite this there are indications from a number of exposed groups that the risk of cancer may start to decline many years after exposure. This has been well documented for leukemia, but also been observed in the case of bone cancers (German, 2°4Ra cases), thyroid cancers (US, follow-up study after thymus irradiation), solid cancers
(anklyosing spondylitics) and possibly lung cancers in the uranium miners. These results suggest that for the Japanese population the excess risk may ultimately decrease with time and thus multiplicative projection models applied over a lifetime could result in an overestimate of the cancer risk.
Table 1. Number of deaths from all cancers other than leukemias among Japanese atomic bomb survivors with a DS86 dose of 0.75 Gy or more (from Preston and Pierce, 1987)
Age at Time since exposure (years)
| exposure
(y $) |
5-25 | 24-40 | 5-40 | ||
| < 20 | O‘ | 14 |
|
58 | |
| Eb | 4.03 | 17.8 | 21.8 | ||
| O/E’ | 3.47 | 2.47 | 2.66 | ||
| 20—34 | O | 26 | 48 | 74 | |
| E | 13.0 | 24.4 | 37.4 | ||
| O/E | 2.01 | 1.96 | 1.98 | ||
| 35 | O | 119 | 99 | 218 | |
| E | 86.7 | 68.9 | 155.6 | ||
| O/E | 1.37 | 1.44 | 1.4 | ||
| All | O | 159 | 191 | 350 | |
| E | 103.7 | 111 | 215 | ||
| O/E | 1.53 | 1.72 | 1.63 | ||
‘Observed number of deaths.
Expected number of deaths in an unirradiated population, based on rates among those with a DS86 dose < 0.1 Gy.
’Relative risk.
The A-bomb Survivors in Japan
The mortality experience of the Hiroshima and Nagasaki A-bomb survivors has been the single most important source of information on the radiation- related risk of cancer induction. New data that has become available on this population of more than 90,tXi0 people in the Life Span Study (LSS) followed up since 1950 has necessitatcd a revision of previous risk estimates [16, 19]. There are a number of components to this change. The first is a revision of the dosimetry (DS86) to allow, amongst other factors, for the high humidity in the air over the cities which has substantially reduced the neutron dose at Hiroshima from the earlier 1965 (T65) estimates which were based on measurements in the dry atmosphere of the Nevada desert. Improved estimates have also been made of yield of the Hiroshima bomb (increased from 12.5 to 15 ktonnes), the shielding provided by buildings and of tissue and organ
doses. The second is that the number of excess cancers in the population has increased due to the increased period of follow-up (to 1985) and an estimate of the cancers occurring in the period 1945—1950 have now been made. The third is that multiplicative, rather than additive risk model now appears to provide a better basis for assessing lifetime risk of most solid cancers.
UNSCEAR [24] in a repon to the General Assembly hens provided information on radiation induced cancer risk for a number of tissues in the Japanese population based on both additive and multiplicative projection
models (Table 2). The total cancer risk at high dose and high dose rate is estimated to be 4 to 7 x 10 2 Sv”* using the additive and multiplicative
models respectively and an age averaged risk coefficient. This compares with the Committee’s 1977 assessment of 2.5 x 10*2 Sv°’ at high dose rate using the additive model. Because children and young persons are more sensitive to radiation than adults the application of age specific risk co- efficients increases the predicted number of radiation-induced cancers. The Committee gives an indication of the effect of different age groups on the risk coefficients by applying the two models: 4-8 x 10*2 Sv*1 for a population of working age (25—64 yrs) compared with 4-11 x 10 ° Sv*’ for a population of all ages (Table 2).
Table 2. Projected lifetime risks for a Japanese population following whole body-y-exposure to 1 Gy (low LET) radiation at high dose rate (UNSCEAR, 1988)
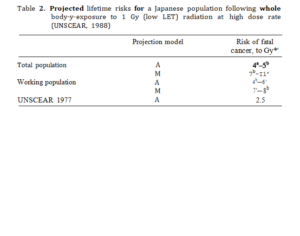
These risk estimates for whole body radiation exposure are based on an extrapolation into the future which is somewhat uncertain for solid cancers, because two-thirds of the Japanese survivors are still alive and two-thirds of the cancer risk has still to be expressed. Up to 1985 about 80 excess leukaemias and 260 excess solid cancers had occurred in the LSS population for whom DS86 doses are available out of a total of about 6000 cancer
*A risk of 1 x lH’ Sv ‘ corresponds to a risk of cancer of 1 in 100 per Sv or 1 in 100,000 per msv.
deaths [16]. The risk of radiation-induced leukaemia is more certain than that for solid cancers, however, as few more excess cases are now expected. There are also uncertainties in extrapolating the cancer risks based on the Japanese population exposed to radiation at high dose rates to low doses and dose rates relevant for radiological protection purposes.
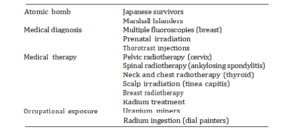
Although the data on the A-bomb survivors provide information on risks of cancer in a range of tissues, to date no information is available under the new dosimetry for radiation-induced cancers of the liver, cells on bone surfaces, thyroid and skin. Information on radiation-induced cancer in these tissues is however, available from other epidemiological studies summarised in Table 3. Details of some of these studies are described below.
Table 3. Human populations available for risk estimation
Bone Cancer
Intakes of Radium-226
An increased incidence of bone cancer and of head sinus carcinoma has been observed in persons exposed to long-lived radium, particularly in painters of luminous dials, but also in radium chemists or persons treated with radium salts for a supposedly therapeutic effect (18]. These persons
became internally contaminated with pure 2° Ra (f1/2- 1,620 yrs) in some cases, and in other cases with various mixtures of °26Ra and 228Ra
(* = 6.7 yrs). These long-lived radium isotopes deposit mainly in the skeleton. By the end of 1983, 62 cases of bone sarcoma and 32 cases of head sinus carcinoma had occurred in a total of 2,352 persons with measured
body contents of radium. A further 23 bone sarcomas and 5 head cancers have appeared in 3,412 unmeasured cases. The majority of the bone sarcomas and head cancers had appeared by 1969, although three bone tumours have appeared since then and head cancers have recently appeared at a greater rate than bone cancers. This no doubt reflects the continued irradiation of the skeleton. The bone sarcomas appear to have been induced by a-particles from either the 22&a or 22’Ra decay series, whereas the head sinus carcinomas are thought to be caused mainly by the accumulation of radon (222Rn) gas in the frontal sinuses and mastoid cells. The radon is produced by the decay of *“Ra in nearby bone.
Except for the bone sarcomas and head sinus carcinomas no definite excess in other types of malignancy, including leukaemia, is presently ascribed to the internal deposition of long-lived radium.
Intake of Radium-224
The effects of intake of radium has also been studied in German patients injected with 22‘Ra shortly after World War II. The study group consists of
a population of 68 l adults and 218 juveniles (age at first injection varied between 1 and 20 years) who received weekly or twice-weekly intravenous injections of 224Ra, mainly for the eeatment of bone tuberculosis or ankylosing spondylitis [9]. The follow-up times from first injection to death or last
known health status ranged from 0—38 years and averaged 22 years. By June 1984 half of the patients were known to have died. As with the 2 Ra
cases bone sarcoma has been the main effect of intakes of 2“Ra with 54 cases observed (36 in juveniles and 18 in adults), compared with only 0.2 expected naturally. The last bone tumour occurred in 1983, 33 years after the injection of 2^Ra into a three-year-old boy and is the only bone sarcoma reported in this series since 1974. Very few new tumours are now expected.
Bone Cancer Risks
Based on the information on bone cancer risk following intakes of radium
ICRP (1991) has adopted a total risk estimate of 5.10“ Sv°’.
Lung cancer
An increased mortality from lung disease has been observed in underground miners working in Czechoslovakia, Canada, United States of America and Sweden exposed to 22*Rn and its decay products [2].
The increase’in mortality from lung cancer has been correlated with air concentrations of radon in different mines and the duration of exposure. Bronchial stem cells and secretion cells in the airways are considered to be the main target cells for the induction of lung cancer resulting from radon exposure. There are many difficulties in calculating the radiation dose to these cells as a result of exposure to radon decay products (expressed in working level months The radiation dose over the working life must be taken into account and the dust loading of the atmosphere known, as it determines the extent of absorption of radon daughters onto the respirable *1 WL is any combination of the short-lived decay products of radon per litre of air which will result in the ultimate emission of 1.3 x 10 MeV of tr-particle energy. A WLM results from exposure to a concentration of decay products in air of 1 WL for an average working month of 170 hours at a breathing rate of 1.2 m° h*’. particles. In addition to any possible synergistic effects between smoking and radon exposure, the presence of dust, diesel fumes and other possible carcinogens in the mine atmosphere causes some uncertainty as to whether an excess of cancer can be attributed to radiation alone. The BEIR IV Committee has suggested a risk of lung cancer following exposure to radon and its decay products of 350 cases per 106 persons per WLM. This corresponds to a risk of 0.420 x 10*2 Sv°l following exposure of the lung (3.5 x 10*2 Sv°1, effective dose), assuming a radiation weighting factor, IVR for a-irradiation of 20 and is similar to the value of 0.68 x 10 2 Sv ‘ adopted by ICRP for a working population based on the A-bomb survivors (Table 6).
Liver cancer
Thorotrast is colloidal thorium oxide. In the late 1920s it began to be injected into the arteries of patients for use in diagnostic radiology as it was an excellent X-ray contrast material. The average dose of about 25 ml of Thorotrast contained 5 g of thorium with an activity of about 20 kBq 23*Th
with additional radioactivity from its decay products. The colloidal Thorotrast was cleared from the bloodstream by uptake into phagocytic cells depositing about 60% ’in liver, 30’7o in spleen and 10% in red marrow. Extensive epidemiological studies in Portugal, Sweden, Denmark, the United States, the Federal Republic of Germany and Japan have shown that retention of thorium oxide particles in the liver and in the bone marrow has resulted in an increased risk of liver tumors and leukemias as well as liver cirrhosis and other cardiovascular diseases [25]. On the basis of an injected dose of 25 ml, the dose to the liver is estimated to be 0.25 Gy y*’. Present estimates, based on a latent period of 20 years, suggest a lifetime risk of liver cancer following exposure to Thorotrast of about 0.15 x l0*2 Sv° (assuming a WR for o-radiation of 20) [2, 7], about half this risk is expected to be expressed by 40 years after exposure.
Thyroid cancer
Groups of children and young persons who received thyroid irradiation and who can be used to derive risk coefficients for thyroid cancer include children who received X-ray treatment for thymic enlargement, patients treated in US hospitals for thyrotoxicosis and other benign lesions of the neck and patients who received X-ray treatment for thyroid diseases [12]. The risk coefficient calculated for the incidence of radiation-induced thyroid cancer in children is 2.5 cancers per 10 -PY Gy. In the majority of cases, particularly in the young, thyroid cancer is not fatal. The mortality from radiation-induced thyroid cancer is expected to be about 10% of the incidence. There is also evidence that the risk in adults is about half that in children and that the risk in females is about twice that in males. For a population uniformly exposed to external radiation the risk of fatal thyroid cancer is estimated to be 8.0 x l& Gym, assuming 5 year latent period [7]. Information on human populations given iodine-131 for non-therapeutic reasons, and who received doses well below 2 Gy indicates a risk coefficient 3 to 4 times less than that obtained following external radiation at high dose rates [12].
Dose and Dose Rate Effectiveness Factors (DDREFs)
Risk coefficients for radiation-induced cancer are mainly based on population groups exposed at high doses and high dose rates. Studies at the molecular, cellular, tissue and whole animal level have demonstrated that radiation damage increases with dose and that, at least for low LET radiation, at high dose rates it is often greater per unit of exposure than at low dose rates. Thus, although the assumption normally made for radiation protection purposes is that the dose response curve for cancer induction is linear, with the risk proportional to dose, in practice a dose and dose rate effectiveness factor (DDREF) has commonly been used to allow for a reduced effectiveness of radiation in inducing cancer in man at low doses and low dose rates. The choice of a suitable DDREF has caused considerable debate with relevant data being available from cellular and animal studies, as well as human epidemiology.
A DDREF of 2 was used by ICRP in 1977 for assessing the risks of cancer induction for radiological protection purposes based on conclusions by UNSCEAR [22] (Table 4). In 1986 UNSCEAR suggested that for many cancers the assumption of a linear response when extrapolating from mformation at high dose rates could overestimate risk at low dose rates by up to a factor of 5. Recently, UNSCEAR [26] stated that risks at low dose rates of low LET radiation may be less than high dose rates by a factor of between 2 and 10. Similar conclusions were reached by the BEIR V Committee [3]. ICRP 1991 have based estimates of DDREF principally on the analysis by Pierce and Vaeth [15] of the data from the Japanese survivors and on theoretical considerations based on a linear-quadratic dose response model. As a consequence they have adopted a DDREF of 2 (Table 4), recognising that the choice is somewhat arbitrary and over-simplistic when applied to
Table 4. Summary of dose and dose rate effectiveness factors
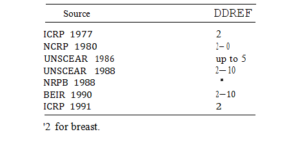
derive probability coefficients for all site specific cancers for a wide range of total doses and dose rates. A better understanding of the mechanisms involved will be essential for improving understanding of the effects of both dose and dose rates on radiation-induced tumour induction in man.
Risk Coefficients for Radiation-induced Cancer
In the last few years a number of studies have been published which have calculated risks of radiation-induced cancer for different populations. They have been based predominantly on information derived from the A-bomb survivors but supplemented by data from other epidemiological studies. Most risks have been calculated for the general population, although a number of reports have also given risks for workers. ‘these tend to be lower (by about 20-40’7) because of the greater risk to children and young persons calculated using the relative risk projection model for most solid cancers. Table 5 summarises the information on somatic radiation risks at high doses and high dose rates published in recent years by UNSCEAR (1988), BEIR (1990), NRPB (1988), ICRP (1991), using mainly relative risk projection models for most solid cancers. In the majority of studies, lifetime risks of cancer have been calculated although NRPB also gave risks to 40 years after exposure (the present period of follow-up of the A-bomb survivors). UNSCEAR calculated risks based on both an age-averaged and an age-
specific constant relative risk models (Table 3).
BEIR V [3] calculated risks to a US population and gave values for a number of tissues using time-varying relative risk models for some cancers
Table 5. Estimated lifetime fatal cancer risks in populations (all ages, both sexes) associated with exposure to low LET radiation at high doses and high dose rates, based on multiplicative projection model

Range based on age-averaged and age-specific constant relative risks.
’Risk calculated to 40 yrs after exposure and lifetime assuming age-specific constant
relative risks. see text (Section 3.7).
°Average value based on USA, UK, Japan, Puerto Rico and Chinese population. Risks for workers 8.0 x 10*’ Sv*’.
Applying BEIR V models. (leukaemia, respiratory tract, breast cancer in females). It is noteworthy that BEIR V, unlike UNSCEAR, calculated excess cancer deaths, not early deaths. The former risk is about 20—25% less than the latter reflecting the baseline cancer rate in the populafion. ICRP (1991) in the recent recommendations calculated risks for a ‘world’ population based on average value for five populations (Japan, UK, USA, Puerto Rico, China) and on transferring both absolute and relative risks across populations.
Overall, the lifetime risks calculated in recent years are not too different for the various studies, the lowest value being for UNSCEAR (1988) using age-averaged risk coefficient. ICRP (1991) have adopted a rounded value of 10 x l0*2 Sv°’ for the risk coefficient for cancer at high doses and high dose rate following exposure of a population. Applying a DDREF of 2 gives a
risk of 5 x 10°* Sv*’ for radiation protection purposes. Risk coefficients for individual tissues are given in Table 6. For workers the risk coefficient adopted for radiation protection purposes is 4 x 10 2 Sv°1.
Table 6. Risk coefficients for fatal cancer adopt
| Population | Workers | ||
| Bladder | 0.30 | 0.24 | |
| Red bone marrow | 0.20 | 0.50 | 0.40 |
| Bone surface | 0.05 | 0.05 | 0.04 |
| Breast | 0.25 | 0.20 | 0.16 |
| Colon | 0.85 | 0.68 | |
| Liver | 0.15 | 0.12 | |
| Lung | 0.20 | 0.85 | 0.68 |
| Oesophagus | 0.30 | 0.24 | |
| Ovary | 0.10 | 0.08 | |
| Skin | 0.02 | 0.02 | |
| Stomach | 1.10 | 0.88 | |
| Thyroid | 0.05 | 0.08 | 0.06 |
| Remainder | 0.50 | 0.50 | 0.40 |
| Total | 1.25 | 5.00 | 4.00 |
| Irradiation in Utero | |||
For the developing embryo and foetus there is evidence that cancer induction may occur following irradiation in utero. Information on the risk of cancer following irradiation in utero has been reviewed by UNSCEAR (1972; 1977) and by the BEIR-III Committee (BEIR, 1980). Current risk estimates for radiation-induced childhood cancer (<15 years) are based mainly on data collected in the Oxford Survey of Childhood Cancers (OSCC) concerning obstetric radiography [4). Information is also available from other studies of prenatal X-ray exposure that have been performed in the USA and elsewhere,
and from the study of those irradiated in utero as a result of the atomic bombings of Hiroshima and Nagasaki. Based orr data from the OSCC and information from UNSCEAR (1972) on doses received in utero from obstetric radiography, the number of excess cancer cases is calculated to be about 6 x 10°2 Sv°1 for in utero exposure [14]. Since slightly less than 50% of childhood cancers consist of leukaemia and other lymphatic/haemato- poietic cancers (OPCS, 1981) and the relative risks are similar for these and other cancers, a risk of 2.5 x 10*2 Sv*1 is calculated for leukaemia and
3.5 x 10*2 Sv*l for solid cancers. Also, as approximately half of all childhood cancers are fatal (OPCS, 198 1), the number of excess cancer deaths will be 3 x l0‘2 Sv°1 comprising 1.25 x 10 2 Sv‘1 for leukaemias and 1.75 x 10*° Sv*’ for solid cancers [20]. These risks are
derived from follow-up studies on children irradiated in utero with radia- tion doses up to a maximum of 10—20 msv. In separate analyses Bithell and Stiller [5] estimated the risk from the OSCC to be about 2.2 x 10*2 Sv°’ and Mole [10] estimates the risk to be 4—5 x 10 2 Sv*’ (95% C I 0.8—9.5 x 1W2) based on a survey of the doses associated with routine obstetric radio- logy in the UK in the period 1958—1961. Recent results from the Life Span Study [26] for the period 1950—1984, based upon DS86 dosimetry and an incidence of two childhood cancers out of 1630 children irradiated in utero, indicate an upper bound of risk on 95% confidence limits of
2.8 x 10 2 Sv 1.
Summary of Risk Factors for the General Population and Workers Used in Setting Dose Limits
The International Commission on Radiological Protection now considers four components of the detriment (health effects) due to irradiation of the tissues and organs of the body at low doses when assessing the overall effects of radiation. These include the probability of fatal cancer; the probability of non-fatal cancer and the probability of severe hereditary disease, both .weighted for severity values assigned to the non-fatal cancers and severe hereditary disease (including multifactorial diseases), each amount to about one-fifth of the detriment associated with fatal cancer. In summary the aggregated detriment amounts to 7.3 x 10*2 Sv°1 for a nominal population (Table 7). It is slightly less (5.6 x 10 ° Sv* ) for a population aged 18—64 years who are occupationally exposed, when account is taken of the omission of younger persons who are more radiosensitive and the shorter mean potential period of reproduction. In addition, the temporal pattern of fatal cancer risk is such that the period of maximum risk occurs in the seventh and eighth decades of life if the multiplicative projection model is used to calculate the lifetime expression of the cancers in persons exposed continuously to small annual doses at or below the dose limits.
Table 7. Risk factors for protection, 10‘2 Sv°’
| Fatal cancer | 1.25 | 5.0 | 4.0 |
| Severe hereditary effects | 0.40′ | i.ob | 0.6b |
| Total | 1.65 | 6.0 | 4.6 |
| Hereditary effects | |||
| (weighted) | 1.3 | 0.8 | |
| Non-fatal cancers | |||
| (weighted) | — | 1.0 | 08 |
| Total (weighted)‘ | — | 7.3 | 5.6 |
‘First two generations.
All generations.
°To allow for non-fatal cancers and years of life lost for cancers and hereditary disease.
dFor fatal cancer the detriment coefficient is equal to the probability coefficient.
Future Prospects
There are a number of important questions that are to be answered in the assessment of the risk of radiation-induced cancers in human popu- lations. Very limited information is available at the low doses and low dose rates that are important for radiation protection and the risks have to be assessed from populations exposed at high doses and dose rates by applying an appropriate dose and dose rate effectiveness factor. Increasingly, however, epidemiological studies on groups of workers in the nuclear industry are providing information on exposures at low doses and dose rates although at present any estimates of risk have large uncertainties associated with them. With the development of these national studies and by pooling them internationally these uncertainties should be progressively reduced. The projection of lifetime risk remains uncertain, particularly for those exposed at younger age groups, and is largely based on empirical fits to the epidemiological data obtained to date. Continued follow up of exposed populations, in particular the A-bomb survivors in Japan is needed for validating current models. It seems likely that epide- miological studies will be unable to answer all the questions concerned with the effects of dose, dose rate, radiation quality and individual sensitivity on cancer induction. Ultimately this must depend on a much better understanding of sensitivity of tissues to radiation. This will come partly from carefully controlled animal studies but increasingly from cellular and molecular studies on the fundamental mechanisms involved in cancer induction.
REFERENCES
- BEIR III. Committee on the Biological Effects of lonising The effects on population of exposure to low levels of ionizing radiation. National Academy of Sciences. National Academy Press, Washington DC 1980.
- BEIR Health Risks of Radon and Other Internally Deposited Alpha-emitter. National Academy Press, Washington DC 1988.
- BEIR V. Health Effects of Exposure to Low Levels of lonizing Radiation. NationalAcademy Press, Washington DC
- Bithell,F. and Stewart, A.M. Pre-natal irradiation and childhood malignancy: A review of British data from the Oxford Survey. Br. J. Cancer 1975; 35: 271.
- Bithell, J.F. and Stiller, C.A.A new calculation of the carcinogenic risk of obstetric X-raying. Statistics in Medicine 1988; 7: 857—864.
- International Commission on Radiological Protection. Recommendations of theInternational Commission on Radiological Protection. ICRP Publication 26, Pergamon Press, Oxford
- 1990 Recommendations of the International Commission on Radiological Protection. ICRP Publication 60. Annals of the ICRP 1991; Vol. 21: No. 1—3.
- Mays, C.W., Spiess, H., Chemelevsky, D. and Kellerer, A.In: Proc. Symp. “The Radiobiology of Radium and Thorotrast”. W. Gossner, G.B. Gerber,
- Hagenand A. Luz. Neuherberg 29—31 Oct, 1984. Strahlentherapie 1986; 80
Suppl. 14-21: pp 27—31.
- Mays,W. Alpha-particle-induced cancer in humans. Health Phys. 1988; 55: 637—652.
- Mole,H. Fetal dosimetry by UNSCEAR and risk coefficients for childhood cancer following diagnostic radiology in pregnancy. J. Radiol. Prot. 1990; 10: 199—203.
- Muirhead,R. and Kneale, G.W. Prenatal irradiation and childhood cancer. J. Rad. Prot. 1989; 9: 209—212.
- NationalCouncil on Radiation Protection and Measurement, Induction of thyroid cancer by ionising radiation. Report No. 80, 1985.
- Influence of dose and its distribution in time on dose-response relationships for low-LET radiation. Washington DC, NCRP Report No. 64, 1980.
- Otake,, Yoshimaru, H. and Schull, W.J. Severe retardation among prenatally exposed survivors of the atomic bombing of Hiroshima and Nagasaki: A comparison of the T 65 DR and DS 86 dosimetry systems. Hiroshima, Radiation Effects Research Foundation, RERf TR 16—87; 1988a.
- Pierce,A. and Vaeth, M. Cancer risk estimation from the A-bomb survivors: Extrapolation to low dose, use of relative risk models and other uncertainties. In: Low Dose Radiation-Biological Bases of Risk Assessment (Eds. K.F. Baverstock and J.W. Stather). Taylor and Francis, London, 1989; pp 54—75.
- Preston,L. and Pierce, D.A. The effect of changes in dosimetry on cancer mortality risk estimates in the atomic bomb survivors. Hiroshima, Radiation Effects Research Foundation, RERf TR 0—87, 1987.
- Rowland,E. and Lucas, H.F. Radium dial workers. Radiation Carcinogenesis: Epidemiology and Biological Significance, Ed. J.D. Boice and J.E. Fraumeni. Raven Press, New York, 1984; 231—240.
- Rundon,, Kaene, A.T. and Lucas, H.F. et al. Current (1984) status of the study of 22‘Ra and °°’Ra in Humans at the Center for Human Biology. In: Proc. Symp. “The Radiology of Radium and Thorostrast”. Eds. W. Gossner, G.B. Gerber, U. Hagen and A. Luz. Neuherberg 29—31 Oct, 1984. Strahlentherapie 1986; 80
- Shimizu, Y., Kato, H. and Schull, W.J.Studies of the Mortality of A-Bomb 9. Mortalité 195Œ-1985. Part 2. Cäncer Mortality Based on the Recently Revised Doses (DS 86). Radiat. Res. 1990; 121: 120—141.
- Stather, J.W., Muirhead, C.R., Edwards, A.A., Harrison, J.D., Lloyd, D.C. and Wood,R. Health Effects Models Developed from the 1988 UNSCEAR Report. NRPB-R226, 1988.
- UnitedNations Scientific Committee on the Effects of Atomic Sources and effects of ionising radiation. 1977 report to the General Assemblé, with annexes, 1977.
- lonizing Radiation: Levels and Effects. 1972 Report to the General Assembly with Annexes. Vol. II: Effects. New York, United Nations, 1972.
- Ionising Radiation: Sources and Biological Effects. 1982 Report to the General Assembly, with annexes.
- Genetic and Somatic Effects of Ionising Radiation. Report to the General Assembly with annexes.
- Sources, Effects and Risks of Ionising Radiation. 1988 Report to the General Assembly, with annexes.
- VanKaick, , Muth, H. and Kau1, A. The German Thorostrast Study. Report No. IEUR 9504 EN Luxembourg CEC (quoted in BEIR IV), 1984.
- Yoshimoto,, Kato, H. and Schull, W.J. “Risk of cancer among children exposed in utero to A-bomb radiations, 1950—84”. Lancet, 1988; Sept. 17: 665—
Environmental Radiation and Human Cancer
David M. Taylor
Institute for Pharmacologie (Radiotoxicology) University of Heidelberg, Heidelberg, Germany
School of Chemistry and Applied Chemistry University of Wales of Cardiff, Cardiff, UK
Throughout its existence on this planet Homo Sapiens has been exposed to both external and internal irradiation from environmental sources. The external radiation comes from cosmic rays and from the y- and Q-rays emitted by the natural radioelements in the earth’s crust. The internal radiation comes from the inhalation of radon and thoron and their daughter products and from the ingestion of 4°K, 14C and radionuclides of the uranium and thorium series in food and water, in most parts of the world internal irradiation accounts for about 80’7« of the radiation dose received. In general the annual radiation dose received from environmental sources is very small, about 1 to 3 msv (Fig. 1), and it has been assumed to be a negligible hazard to human health. However, in recent years the recognition that about 60% of the internal dose arises from n-particle emitting radionuclides, predominantly from radon and thoron and their daughter product, has prompted suggestions that this radiation may cause a significant fraction of spontaneous human lung cancer as well
HUMAN ANNUAL EXPOSURE TO NATURAL RADIATION BY SOURCE (microsieverts)
Average Dose In MlcroSleverts per Year
Cosmic Gamma Internal Radon Thoron
Fig, 1. The average natural radiation background dose to humans living in the UK but the dose may be regarded as fairly typical for Europe as leukaemia, prostate and other cancers [3, 4, 15]. Some of these suggestions are at present highly controversial, but they are important in that they appear to challenge the established dogma and stimulate new thinking on the radiobiology of internally deposited radionuclides.
In UK the annual effective dose equivalent delivered to the lungs by inhalation of radon and thoron decay products is about 1200 jisv, or about 559» of the total environmental radiation dose from all natural sources, but the corresponding doses to bone marrow and other tissues at about 120 jisv are an order of magnitude lower [13]. Early calculations based on data from uranium miners suggested that perhaps some 10 to 40’7» of human lung cancer might be due to exposure to radon and radon-daughter products, but this was considered by others to be an overestimate and a value between 1 and 5’7o was thought more likely. The data for the UK suggest that perhaps some 6% of lung cancer may be caused by radon exposure. For tissues receiving radiation doses about ten times less than that delivered to the lung, assuming a similar risk factor for all cancer induction, a significant contribution from radon and thoron to cancers in those tissues appears unlikely. However, Richardson and his colleagues [15], noting the high solubility of radon in fat have calculated that bone marrow doses may be higher than previously thought and have suggested that this radiation could account for 6 to 12’7« of myeloid leukaemia.
These calculations assume an averaged radiation dose to the tissues toncemed, however, e-particle irradiation is very nonuniform and the average dose to the tissue does not necessarily reflect that actually delivered to sensitive cells. In fact, it is not improbable that a high fraction of cells traversed by e-particles will be so severely damaged that the formation of a heritable mutation which could give rise to a cancer is unlikely. One tissue which does appear to be especially sensitive to cancer induction by n-particles is bone, and for this tissue we do have information about cancer induction and microdosimetry for ‹x-particle emitting radionuclides, both for animals, and to a lesser extent for humans.
This information on the radiotoxicity, in terms of bone tumour
[osteosarcoma] induction by tr-particle emitting radionuclides in humans comes from the studies in the radium dial painters, 2 Ra, the °24Ra patients and the Thorotrast studies [18], while data for 2“Ra, °28Th, 237Np, 2′ Pu, 241Am, 244Cm [11, 12, 17, 19] comes from the studies in beagles, rats and mice which have been carried out in the USA, UK, Germany and Russia during the past forty years. The animal studies are all based on the concept
of the Toxicity Ratio, introduced by Robley Evans forty years ago [11]. This may be stated quite simply in the following words, as radium in humans is to plutonium in humans so is radium in the animal to plutonium in the
animal. For plutonium we may read any other bone-seeking n-emitter. From this it is clear that the 2“Ra data for humans are crucial since they set the
risk for all the other nuclides. Figure 2 shows a plot of the results for 982 dial painters from lhe USA and the UK, the data have been fitted to a linear
Radium – induced Osteosarcomata in Human
Averaged Dose to Bone (Kilo Sieverts)
Fig. 2. The bone cancer incidence in female radium dial painters from the USA and the UK. The risk factor shown is the slope of the regression line assuming a linear fit to the data.
relationship which suggests a slope, or Risk Coefficient of 0.025%/Sv average a-radiation dose to bone, which is the relationship proposed by Mays and Lloyd in 1972 [11]. The Risk Coefficient may overestimate the true risk, by a factor of about 5, since only one bone tumour was observed in 586 persons whose averaged bone dose was < 14 Sv. Table 1 shows the Toxicity Ratios and Risk Coefficients for the major a-emitting bone seekers calculated from the combined data for humans, beagles, rats or mice. The values have been rounded to the nearest single digit to avoid giving the impression of unjustified precision.
Table 1. Toxicity Ratios and human Risk Coefficients for bone sarcoma induction by e-particle emitting radionuclide
Radionuclides Toxicity Ratio’ Risk Coefficient (&oSv)
| Radium-226 | i | 0.025* |
| Radium-224 | 3 | 0.07* |
| Radium-228 | 2 | 0.05 |
| Thorium-228 | 8 | 0.2 |
| Thorium-232 | 2 | 0.05* |
| Americium-241 | 3 | 0.07 |
| Curium-243, 244 | 3 | 0.07 |
| Califomium-249 | 3 | 0.07 |
| Plutonium-239 | 16 | 0.40 |
| Neptunium-237 | 16 | 0.40 |
*Values bases on direct human observations.
‘Toxicity Ratios calculated from data for humans, beagles, mice and/or rats.
This table shows quite clearly that there are marked differences in the radiotoxicity of the most important bone-seeking radionuclides, differences which are not yet fully explained but depend most probably on chemical- biochemical differences in the deposition patterns of the radionuclides in bone or on bone surfaces and the variations in the microscopic distribution of the o-radiation dose which result therefrom. This aspect of bone radiobiology will not be discussed further here.
Despite the very difficult problems of analysing accurately very small quantities of radionuclides in the limited amounts of human tissue which can ethically be made available for analysis, a number of studies of the concentration of the most important natural n-emitting radionuclides, and also plutonium, in human bone have been made on samples taken from various parts of the world [1, 6, 10, 16]. Table 2 lists a compilation of the mean values calculated for the elements radium, thorium, uranium and plutonium in human bone, together with the range of values which is very broad. The samples were taken mainly from adult humans.
Table 2. Concentrations of n-particle emitting radionuclides in human bone of persons living in areas of average natural radiation
| Radionuclides | Mean concentration | Range of concentration |
| (mBq/kg) 4 ISD | (mBq/kg) | |
| Radium-226 | 190 -k 120 | 50 to 510 (o > 1700) |
| Thorium-228 | 67 A 70 | 12 to 86 (n = 26) |
| Thorium-230 | 39 d- 41 | 4 to 182 (n = 41) |
| Thorium-232 | 18 A 18 | 2 to 37 (n = 39) |
| Uranium-234 | 50 + 11 | 8 to 242 (ii = 29) |
| Uranium-235 | 2 d- 1 | 0 to 7 (n — 20) |
| Uranium-238 | 35 A 8 | 5 to 172 (ii = 29) |
| Plutonium-238 | 4 – 2 | 0 to 20 (n = 16) |
| Plutonium-239 | 10 A 2 | 0 to 54 (n = 28) |
| n = Number of ddta sets. |
Using these data the e-particle radiation dose rate has been calculated for each radionuclide and integrated over a period of fifty years. Table 3 shows the computed median and the range of averaged o-radiation doses to bone for each of the radionuclides. The data are presented as msv and they are of the same order of magnitude to the cumulative doses to bone marrow from radon and thoron over 50 years which can be inferred from the data of Richardson ct a1 (15).
Using these cumulative doses and the Risk Coefficients shown in Table 1, it is possible to calculate tentative values for the risks of bone tumour induction over a 50 year period. These tentative risks, calculated for persons living in areas with a more or less normal radiation background are shown graphically in Fig. 3. The data are presented as percentages of the spontaneous risk of developing osteosarcoma over 50 year period. This spontaneous risk
Table 3. Calculated cumulative averaged n-radiation dose to human bone from environmental radionuclides, assuming 50 years of exposure
| Radionuclides Median dose (mSv/50 yr) | Range of dose
(mBq/kg) |
|
| Radium-226 6.3
Thorium-228 1.0 Thorium-230 0.5 Thorium-232 0.2 Uranium-234 0.8 Uranium-235 0.02 Uranium-238 1.3 Plutonium-238 0.04 Plutonium-239 0.3 |
1.7 to 17
0.3 to 8.0 0.1 to 4.3 0.04 to 0.8 0.04 to 1.2 0 to 0.04 0.1 to 2.3 0 to 0.1 0 to 0.5 |
|
|
Total |
||
| Pu-239 | ||
| Pu-238 | ||
| U-238 | ||
| MEDIAN Risk Over 50 years | ||
| U-234 | ||
| Th-232 | ||
| Th-230 | ||
| Th-238 | ||
| Ra-226 | ||
| 0 0.2 0.4 0.6 | 0.8 | 1.2 |
% of the Natural Risk [0.05%] in 50 yrs.
Fig. 3. The estimated possible risk of bone tumour induction from natural environmental n-particle radiation over a period of 50 years. The data are expressed as percentages of the natural, or spontaneous risk, which is assumed to be 0.05% in 50 years.
was calculated from the data presented in the IARC Publication “Cancer in Five Continents” (14] to be 5 + 2 cases per 10,000 for males andñ 4 2 per 10,000 for women, or about 0.05% in 50 years. On the basis of the
median values, 2“Ra, 2’2Th, 23Pu might make the greatest contribution, in total the risk at this level from all the radionuclides shown appears to be 1.1% of the spontaneous risk and even for the highest values listed the total risk might be only about 5’7o. Taking account of the widespread of the measured radionuclide concentrations in bone and the large uncertainties in the estimations of the Risk Coefficients the total bone tumour risk from n-particle emitting radionuclides in bone is probably less than 5′ of the natural risk and could well be zero. These calculations have been based on data for people living in average natural radiation areas, few data on the concentrations of n-emitters in the bones of persons living in high background areas, such as Kerala or parts of China are available [l]. What little information is available’ suggests that the mean radiation doses might be about a factor of 10 greater than those calculated here. If such an increased radiation dose to bone manifested itself as a similar increase in bone cancer incidence this might be detectable if a very carefully conducted epidemiological study would be possible in the area concerned.
Recent reports of increased leukaemia incidence in children living near certain nuclear plants [5] has led to claims that occupational contamination of the father by plutonium, or other actinides, might be responsible for the cancer in the child. This suggestion of transgeneration carcinogenic effects, although according to current thought extremely unlikely [2], raises interest in the possible a—radiation doses to the testes from natural environmental radionuclides as well as from any occupational contamination. Information on the concentrations of elements such as thorium, uranium and radium in the human testes are lacking, but some information, accurate perhaps to within an order of magnitude, may be derived from the radionuclides concentrations in human bone discussed above, by using ICRP models to calculate whole body content and then the testicular concentrations. For thorium and plutonium, the ICRP 48 [8] model may be used assuming that of the material reaching the blood 45% goes to bone and 0.035% to the testes. For uranium it has been assumed using the ICRP 30 [7] model that about 50% of the body uranium is in the skeleton and 25’7o is distributed uniformly throughout about 20 kg of soft tissue. On the basis of these assumptions, and also assuming steady state concentrations in the testes, approximate estimates of the testicular concentrations and a-radiation doses have been made. These are shown in Fig. 4. The data in this figure shows that the estimated mean doses range from 0.7 ySv/a for plutonium to 7 jiSv/a for thorium and the total annual dose is about 15 ySv/a with a range from 2 to 64 ySv/a. On the basis of ICRP Risk for severe hereditary effects, of which an induced cancer in a child, a transgeneration cancer might be one, the ICRP Publication 60 [9] has recommended a risk factor of l.3E-2/Sv, the risk at the above level of testicular irradiation might be about less than one in a million/a at the upper end of this range less than one in ten million at the lower end.
The currently available information suggests that, with the possible exception of lung cancer, environmental radiation plays a miniscule role, if any, in the causation of human cancer. However, to be certain of this we need much more information about the sensitive cells for the induction of each type of cancer, the mechanisms of induction and the radiation risk estimates, preferably age-related risk estimates, for the individual types of cancer. Further, especially for e-particles, soft Q-particles and Auger electrons,
Annual Alpha Dose-mIcroSleve»l«
Fig. 4. Estimated radiation dose to the human gonads, testes or ovaries, from
natural or environmental o-particle irradiation.
we need good data on the biodistribution, biokinetics and intracellular deposition of the radionuclides of interest. Further we need information, hopefully for humans. on the cellular micromorphometry and the distribution of the stem cells in tissues in order to determine the microscopic distribution of radiation dose in the region of the sensiñve cells in bone, bone marrow and other tissues. Only when we have such data can we hope to be able to assess with any real certainty, the contribution to human cancer by the oldest and most ubiquitous of our environmental pollutants.
REFERENCES
- Chen,Xing-An. in Radiological Protection 1987; No 84: 13—20.
- Cox, in Radiological Protection Bulletin 1992; No 129: February, 15—23.
- Eatough,P. and Henshaw, D.L. Lancet 1990; 335: 1292.
- Edling, Br. J. Indust. Med., 1985; 42: 721—722.
- Gardner,J., Snee, M.P., Hall, A.J., Powell, C.A., Downes, S. and Terrell,
- Harley,H. and Fisene, N.H. Health Phys., 1990; 58: 513-518.
- InternationalCommission on Radiological Publication 30, Part 1, Annals of the ICRP, 2 (3/4), 1979.
- International Commission onRadiological Protection, Publication 48, Annals of the ICRP, 16 (2/3),
- InternationalCommission on Radiological Protection, Publication 6, Recom-
mendations of the ICRP, 1990, Annals of the ICRP, 21 (1-3), 1991.
- Kawamura,, Yamamoto, M., Igarashi, Y., Shiraishi, K. and Ueno, K. Health Phys., 61, 615—622, 1991.
- Mays, C.W.and Lloyd, R.D. Radiobiology of Plutonium, (Ed.) W.S.S. Jee, JW Press, Salt Lake City, pp. 409—430.
- Mays, C.W., Lloyd, R.D.,Taylor, N. and Wrenn, M.E. Health Physics, 52, 617—624, 1987.
- Miles,C.H. and Cliff, K.D. Radiat. Prot. Dosim. in the press, 1992.
- Muir, (Ed. Cancer in Incidence in Five Continents) Vol. 5, IARC Sci. Publ. 88, 1988, Table 12.2.
- Richardson, R.B., Eatough, J.P. and Henshaw, D.L. Brit. J. Radiol. 64,608— 624,
- Singh, N.P., Lewis, L.L. and Wrenn, M.E. in Metals in Bone, Commission of theEuropean Communities (Radiation Protection), MTP Press, Lancaster, UK, 1985, 231—241.
- Taylor, G.N.,Mays, C.W., Lloyd, R.D., Gardner, P.A., Talbot, L.R., McFarland, S.S., Pollard, A., Atherton, D.A.,Van Moorhem, D., Brammer, D., Brammer, T.W., Ayoroa, G. and Taysum, D.H., Radiant. Res. 95, 584-601. 1983.
- Taylor, D.M., Mays, C.W., Gerber, G.B. and Thomas, R.G.(Eds.). Risks from Radium and Thorotrast, BIR Report 21, The British Institute of Radiology, London,
- Taylor,M. Science Total Environ. 83, 217—225, 1989.
The Effects of Prenatal Irradiation on Carcinogenesis
Lena Einhorn
Department of Oncology, Radiumhemmet, Karolinska Hospital, Stockholm, Sweden
Most published studies have demonstrated a higher incidence oi cancer among children exposed to irradiation in utero (10, 13, 20, 24, 27, 35, 41,
47, 48, 55, 59, 75, 79, 80, 84], although there are still some remaining
doubts about casuality [39, 40, 89, 90]. The 1988 UNSCEAR report [90] finds that the overall evidence does not, however, support a particular first- trimester sensitivity to radiation carcinogenesis, as suggested by some authors. Initially, it appeared that data from the Hiroshima and Nagasaki atom bomb explosions did not support an association between prenatal irradiation and cancer development: A 10 year follow-up of children exposed in utero [30], found no increase in the incidence of malignant tumours, which was in marked contrast to the early cancer incidence among those exposed as adults [36]. However, a recent follow-up has revealed that as those exposed in utero_approach middle-age, they do seem to develop a higher than normal rate of adult malignancies—but not leukemia or embryonic tumours [100].
This may indicate that although an immediate carcinogenic effect was not detectable, in utero cancer initiation may have taken place.
Experimental studies in animals may offer additional information about the connection between prenatal X-ray exposure and subsequent tumour development.
In a review, Brent [8] presents a compilation of the effects of irradiation at various stages of gestation in rat and mouse. He reported, that the late foetal stages appear to be sensitive to radiation-induced carcinogenesis. In contrast, when mice or rats were irradiated before or during organogenesis (before day 12 in mouse or day 13 in rat) no clear increase in tumour development was observed. This early period is also the period when the embryo is most susceptible to malformation. Thus, during embryonic development, the period of maximum sensitivity to teratogenesis appears to be the period of minimum sensitivity to carcinogenesis, and vice versa. Sasaki et al [641and Vesselinovitch et a1 [92] irradiated mice on either day 12 or day 16-18 of gestation. Both groups found an increase in tumour development after the late stage irradiation but not after the early stage irradiation. Rugh et a1 (63) and Upton et al [91] irradiated mouse embryos at different time points of gestation and their tables show a similar pattern. In two studies by Schmahl et a1, no overall increase in malignancy was found when mouse embryos were irradiated during organogenesis [67, 70].
The Effects of Prenatal Irradiation on Carcinogenesis 25
A third study did, however, show an increase [69]. Friedberg et al found no significant increase in malignancy after irradiation of mouse pronuclear zygotes [21]. A study on dogs shows a similar pattern [6]. After four years of follow-up, it was found that dogs, irradiated just before or after birth had a significant increase in malignancy. In contrast, no increase was found among dogs irradiated early in gestation or several months after birth.
Most studies on animal embryos thus indicate that the early embryonic stage is less susceptible to radiation-induced carcinogenesis than the later foetal stage. There are, however, some exceptions to this pattern: Wegner and Graul [95] found an excess of tumours in rats after irradiation on day 9 of gestation, and Streltsova and Pavlenko Mikhailov [81] found that rat embryos irradiated on day 7 of gestation had a higher incidence of tumours than those irradiated later.
Sikov and Lofstrom 76, 77] found that irradiation of rats (20 R or 100 R) on day 10 of gestation led to an overall dccrease in mammary tumors for the first 20 months, after which there was a sudden increase. In contrast, irradiation with 50 R on day 15 of gestation led to a much earlier increase in mammary tumours. However, when the dose given on day l5 was raised to 185 R, it almost completely obliterated mammary tumorigenesis, while at the same time rendering the gonads atrophic. This thus points to a postnatal hormonal promoting effect, effective also after early stage irradiation.
Indeed, other studies have found that irradiation of rodents during early gestation can enhance the carcinogenic effects of postnatal‘treatment with chemical carcinogens [54]. Thus, it appears that early gestation can be susceptible to initiation. In the last few years, there has also been increased interest in the possibility of multigeneration carcinogenic effects (for review, see [49]).
What then is the picture after administration of chemical carcinogens to embryo? A number of groups have studied transplacental administration of carcinogens, particularly to rodent embryos (for reviews, see [53, 60, 61, 83]). The best studied of these carcinogcns is ethylnitrosourea (ENU), a direct acting alkylating nitroso compound [50]. The emerging picture from these studies is very similar to that seen after prenatal irradiation. Carcinogens administered before or during the period of major organogenesis produce a high rate of malformations but very rarcly tumours. In contrast, if the chemical carcinogen is administered after the completion of organogenesis, susceptibility to malformation rapidly declines at the same time as the embryos become susceptible to tumorigenesis. Indeed during the latter part of gestation, the animals are often more susceptible to chemical carcinogenesis than adult animals. It should be noted, that in humans, the major period of organogenesis lasts only through the eighth week of pregnancy.
The Relationship between Development and Neoplasia
The susceptibility of a tissue to cancer is related to its proliferative capacity, and most tumours arise in continuously regenerating tissues. Early embryonic life is the period when the rate of growth is higher than during any other time of life. In view of this, the observations on the low incidence of tumours after early gestation carcinogenic insults seem paradoxical. The observations have previously led the author to suggest that there are developmentally active factors present during early embryonic life, and to a lesser degree later, that can act to prevent malignancy [15]. In summary, the hypothesis is based on the following findings:
- Only3 in 100,000 newborn humans are born with a tumour or develop a tumour during the first month of life [15—45].
- Onlyexceedingly rarely is a malignant tumour found in spontaneously aborted embryos or foetuses [42, 51, 78].
- Asdescribed above, attempts to induce cancer in early stage animal embryos by irradiation or by transplacental chemical carcinogenesis have largely been unsuccessful, even when exposed animals have been observed throughout their lifetime. At later stages of gestation animals rapidly become susceptible to carcinogenesis [53, 60, 61, 83].
- Whenremoved from the embryonic environment, embryonic cells can easily be transformed, and thus the refractiveness to tumour induction lies not in the single cell [60].
- Whenmalignant cells are transplanted to embryos they frequently lose their malignant characteristics [9, 45, 56, 65, 94].
- Socalled embryonic tumoun in humans are tumours that arise mainly during the first 4 years of life and are comprised of immature tissues. However, in spite of their embryonic appearance, the most common embryonic tumours in humans—Wilms’ tumour, neuroblastoma, medulloblastoma and retinoblastoma—arise in organs with an unusually late ongoing organogenesis (97]. These organs partly retain their embryonic appearance until shortly before birth or even for some time after It thus seems, that in spite of the embryonic histology of these tumours, they probably develop at the end of gestation or after birth.
- The younger thechild is, when it develops an embryonic tumour (but not leukemia), the better is generally the prognosis [4, 7, 14, 18, 23, 29, 44, 82, 96]. This is irrespective of the degree of spread at
- Unlike adult cancer, spontaneous regression of embryonictumours is not an uncommon finding [17]. These spontaneous regressions almost never occur after the age of 2 The modes of regression seem to be either necrosis of the tumour—as seen in retinoblastoma—or cytodifferentiation of the malignant tumour to a more highly differen- tiated benign tumour form—as seen in cases of spontaneous regression of neuroblastoma to benign ganglioneuroma (see [15]).
These mechanisms—differentiation and necrosis—are not unique to the regulation of tumour growth. In fact, normal embryonic development seems to be controlled by cell differentiation and cell death.
The Effects oy Prenatal Irradiation on Carcinogenesis 27
Cancer is by its very nature a developmental deviation. And from the evidence, it seems likely that it can be controlled by mechanisms similar, or identical, to those that regulate normal development. The developmental control mechanisms are strongest during embryonic life, which could explain why carcinogenesis seems to be particularly hampered during this period. The unusual sensitivity of the embryo, foetus and young child for developmental disturbances, due to the high rate of growth, seems to be counterbalanced by development-regulatory mechanisms which are most active during the most active periods of growth.
The degree of developmental or the regenerative potential of an organ seems to determine how active the tumor inhibiting control mechanism is. Amphibia are capable of regenerating whole limbs. When malignant tumours are transplanted to a regenerating limb, or a limb amputated distally of a chemically induced tumour, it often leads to differentiation and spontaneous regression of the tumour (62, 73, 74, 85, 86, 87].
That carcinogenesis may be irihibited during periods of active development is in itself an exciting notion. In addition to this, however, some observations in the literature raise the possibility that perhaps, in some situation, carcinogens themselves may act to inhibit, rather than induce, carcinogenesis. The author has previously published a review of some of these data [16].
The Effects of Prenatal Irradiation on Transplacental
Chemical Carcinogenesis in Rats
One of the currently most accepted hypotheses on carcinogenesis is that cancer arises through an accumulation of mutations. But there are certain indications that this may not always hold true during embryogenesis.
There have been a few published studies on rats where prenatal irradiation has been combined with prenatal transplacental chemical carcinogenesis [31, 68, 88, 93]. As mentioned, when ENU is administered after day 12 of gestation in rats it produces neurogenic tumours in a high proportion of the offspring. But, surprisingly, when the ENU-treatment is preceded by irradiation, the result has been a drastic decrease in the development of brain tumours (see Table l(a)). In a typical case, tumour incidence decreased from 69 to 39% when carcinogen treatment was preceded by 1 Gy irradiation, and to 15% when it was preceded by 2 Gy. It has also been shown that when the ENU-treatment is preceded by treatment with methylazoxymethanol (MAM) the tumour incidence decreases [32]. This is surprising, since both irradiation and MAM are themselves mutagenic and in fact, during the later stages of gestation carcinogenic [37). In addition, similar combined carcino- gcnic treatments in adults usually lead to an increase in malignancy [3, 22, 28, 38, 46, 72, 99]. That irradiation of the embryo could actually protect it against carcinogenesis is an observation which seems contradictory and begs for alternative explanations.
One possible explanation for these findings is that the tumour-prone animals die from radiation damage before they have a chance to develop tumour’s.
Table 1(a). Rat embryos irradiated on day 16 of gestation + administered ENU (10 mg/kg body weight of mother) on day 20 of gestation (from Kalter et al, 1980 [74])
X-ray dose (Gy)
| 0 + ENU | 0.05 + ENU | 0. l + ENU | 0.25 + ENU | 1.0 + ENU | 2.0 + ENU | 2.5 + ENU | |
| % Offspring | 68.9 | 58.5 | 65.0 | 46.2 | 39.2 | 14.9 | 14.8 |
| w/neurogenic tumours* | |||||||
| % Alive at | |||||||
| 4 wks | 97.8 | 89.8 | 88.9 | 98.5 | 83.6 | 79.1 | 38.6 |
| 4 mos | 97.8 | 88.1 | 86.7 | 92.4 | 80.3 | 67.3 | 27.2 |
| Mean no. of neurogenic | 1.2 | 1.2 | 1.3 | 1.3 | 1.2 | 1.2 | 1.2 |
| tumors per affected rat | |||||||
| Latent period (days) | 314.7 | 289.5 | 312.3 | 364.3 | 324.2 | 328.5 | 417.3 |
| *Of those alive at 4 wks. |
However, in one study [68], the reduction of the litters due to early radiation- related deaths is insignificant (Table 1(b)), in spite of a decrease in tumour incidence form 74 to 25’7o with 1.5 Gy irradiation. In another study [31], a significant proportion of the irradiated embryos did die from radiation damage before 4 months of age. But in this study the decrease in tumour incidence at almost all doses of irradiation is also larger than the proportion of non- survivors (Table 1(a)). In addition, prenatal mortality is shown not to be significantly related to X-ray dose [31]. Thus, the decrease in tumour incidence in prenatally irradiated animals is not likely explained by a loss of the tumour-prone population due to radiation damage.
Another possible explanation for the decrease in tumour incidence could be radiation-caused destruction of the target cells for ENU-induced tumorigenesis. Prenatal irradiation does cause a decrease in brain-size (68). If, however, the decrease in tumour incidence is due to a reduction of target cells, the number of brain tumours per tumour bearing animal would also be expected to decrease with irradiation. In fact, as can be seen in Tables 1(a) and 1(b), the number of brain tumours per tumour-bearing animal remain the same, despite a drastic decrease in tumour incidence. In addition, the latency periods remain the same [31, 78].
Thus, it seems that irradiation makes the majority of animals resistant to tumorigenesis, whereas the remaining animals are totally unaffected in tumour responsiveness, despite a decease in brain size.
In addition, when instead of irradiation, the neurotropic teratogen methylnitrosourea is applied two days before ENU application, there is no significant reduction in brain tumor incidence, despite considerable microcephaly [l].
What could be the mechanism behind this radiation-induced resistance to tumorigenesis?
The Presence of Neuronal “Rosettes”
ENU-treatment induces major dysplasia of the brain. Tumours are more commonly found in rats which develop this ENU-induced dysplasia. Irradiation also induces major dysplasia. But, surprisingly, in this case the dysplasia seems to be involved or associated with the tumour protective mechanisms. When prenatal ENU-treatment is combined with irradiation, tumour incidence appears to be drastically reduced specifically in brains that have developed major radiation-induced dysplasia. And again, the mean number of tumours per tumour bearing animal remains unchanged at 2.5 in animals with major dysplasia—despite a reduction in tumour incidence from 69 to 49c after 1.5 Gy irradiation ([68], Table 1(a)).
How can we explain the differences between ENU-induced and radiation- induced major dysplasia? Schmahl and Kriegel [68] have made the important observation, that after irradiation, there is one specific form of dysplasia, the existence of which seems to rule out simultaneous tumour development: ectopie neuronal nodules. These cell clusters develop in the cerebral cortex
Table 1(b). Rat embryos irradiated on day 13 of gestation + administered ENU {SO mg/kg body weight of mother) on day 13 of gestation (from Schmahl and Kalter, 1985 [75])
X-ray dose (Gy)
| 0 + ENU | 0.05 + ENU | 0.1 + ENU | 0.25 + ENU | 1.0 + ENU | 2.0+ ENU | 2.5+ ENU | |
| ‘7o Offspring w/brain | 73.8 | 68.2 | 44.0 | 25.0 | |||
| tumours | |||||||
| % Alive at | 84.4 | 90.4 | 90.4 | 91.4 | |||
| 4 wks
Mean no. of brain tumours |
1.62 |
1.67 |
1.63 |
1.56 |
|||
| per affected rat | |||||||
| Offspring w/major | |||||||
| dysplasia: | |||||||
| to that also has brain | 69.4 | 61.9 | 21.9 | 4.3 | |||
| tumours (gliomas)
Mean no. of gliomas |
2.44 |
2.53 |
2.57 |
2.50 |
|||
| per affected rat | |||||||
from so called “rosettes”, induced by irradiation of the rat or mouse embryo from day 10 to day 16 of gestation [19, 26]. Ectopic neuronals nodules were found in up to 40% of the animals which had been irradiated in conjunction with the ENU-treatment, but not in one single case did an animal have both a tumour and ectopic nodules [68]. ENU in itself does not induce rosettes or ectopic neuronal nodules at this dose. It is an effect of the irradiation.
What are these “rosettes”?
Radiation induced rosettes morphologically resemble organogenesis of the neural tube, and they are very similar to the rosettes seen in malignant retinoblastomas and neuroepitheliomas [26]. They have been described as attempts at regeneration after the radiation damage [11]. There is a high rate of proliferation in the rosettes which, however, has ceased by the time these rosettes have developed into ectopic neuronal nodules.
Wilson et al [98] have described development of what they called “tumours” in the head region of rat embryos irradiated on day 9 of gestation. The “tumours” grew rapidly for a few days, after which they completely regressed. Wilson’s “tumours” and the rosettes share the features of being radiation- induced aberrant cell proliferations that never develop into uncontrollable malignant growths.
How can we explain that the presence of what used to be aberrant growth proliferations, rosettes, means that the organ seems to be in a tumour resistant state?
One interpretation is that rosettes are potentially tumorigenic growths which the organism manages to control, and that the later tumour resistance in the same organ is a result of the retained state of control after the carcinogenic insult.
Another way of interpreting it is, that the rosettes are attempts at regeneration after the induced radiation damage, and that with this resumed organogenesis, automatically the tumour control is also reactivated.
In either case, it appears that the irradiation has reactivated the strong tumour inhibiting control mechanisms that were present during organogenesis.
Malformation vs. Tumour Induction
As mentioned, during organogenesis the embryo is highly susceptible to malformation, but not to tumorigenesis. It is a reasonable assumption that some of what we call malformation—usually a developmental arrest—in fact is a result of the organism’s protection against uninhibited growth— tumorigenesis. No doubt, irradiation, and also chemical carcinogens such as ENU, in themselves cause cell destruction-and ensuing developmental arrest. But the general separation between major periods of susceptibility to teratogenesis and carcinogenesis, and the seemingly arrested rudiments of tumour development that can be observed during the periods of teratogenesis, makes it likely that malformation to some extent is a result of the organism’s protection against developmental deviations such as malignancy.
We have thus described a pattern in rat brain where during late gestation a weak carcinogen appears to moderate the carcinogenic effects of a strong carcinogen. In contrast, in the adult, similar combined carcinogen treatments tend to have a synergistic effect on tumour induction. Studies on combined treatment with irradiation and chemical carcinogens in newborn rats show that any tumour inhibitory effect of irradiation decreases soon after birth [25, 33, 34, 43, 57].
Experiments on Mice
The problem with comparing the above experiments in rats with similar experiments in mice is that mouse embryos in general are not susceptible to induction of brain tumours. After transplacental ENU treatment, they tend to display a different spectrum of tumours—the most common being lung, liver, uterine and ovarian tumours. And studies on prenatal irradiation combined with prenatal or postnatal carcinogen treatments, do not show consistent results: Nomura et al [52, 54] found a synergist/c carcinogenic effect of prenatal irradiation and postnatal urethan or TPA treatment in mice, an effect which increased with X-ray dose. In contrast, Schmahl found that high doses (>1.5 Gy) of foetal irradiation tend to inhibit postnatal ENU carcinogenicity, whereas low-dose foetal irradiation seems to enhance postnatal ENU carcinogenicity [16]. Obviously, since in these experiments the chemical carcinogen treatment occurred postnatally, and often several weeks after the irradiation, conditions are different from those described in the abovementioned rat experiments. When Schmahl and Kriegel irradiated moiise embryos (3 Gy) on gestation days l 1—13, and treated with ENU pn gestation day 17, the results were ambiguous. Irradiation seemed to increase the incidence of leukemia and ovarian tumours, and decrease the incidence of lung and liver tumours [69].
A study by Sasaki et al [64] docs, however, suggest that in mice too, there may be a tumour inhibitory effect of early prenatal irradiation. They compared the effects of a single dose of irradiation (2 Gy) at different stages of gestation in C57BL/6XWHT mice: Although late foetal irradiation (days 16—18) increased the overall tumour incidence in males from 46’7» (controls) to 73’7o, and in females from 65 to 76’7» the same dose of irradiation administered on day 12 of gestation instead reduced the tumour incidence to only 11% in males and 15’7o in females. The same pattern was seen for almost all tumour types (Table 2). As mentioned, early stages of development tend to be tumour resistant, whereas foetuses at later stages become susceptible to tumorigenesis. This does not, however, explain why the early irradiation should produce a tumour incidence much below that of normal, non-treated controls. In females, reduction in body-weight was almost the same after early and late stage irradiation, although the difference in tumour incidence was more than five-fold. This argues against the tumour inhibitory effect of early stage irradiation being attributable to target-cell destruction alone. Finally there was a distinct delay in tumour development in the group irradiated on day 12, as compared to the other groups. Schmahl et al
Table 2. BGWF1 mouse embryos irradiated (2 Gy) on day 12 or days 16—18 of gestation. Incidences of the most common tumours vrom Sasaki et al, 1978 f24])
Percentage females Percentage males
| Control | Irr.
days 16-18 |
Irr.
day 12 |
Control | Irr.
days 16-18 |
Irr.
day 12 |
|
| All tumours | 65 | 76 | 15** | 46 | 73** | 11** |
| Lung tumours | 17 | 39** | 4 | 24 | 56** | 5* |
| Lymphoreticular | ||||||
| tissue tumours | 30 | 24 | 6** | 16 | 10 | 2* |
| Liver tumours | 7 | 10 | 0 | 7 | 17 | 0 |
| Uterine tumours | 14 | 11 | 0* | |||
| Ovarian tumours | 1 | 14* | 0 | |||
| Mammary tumours | 5 | 1 | 0 | |||
| Pituitarytumours | 1 | 9* | 0 | 0 | 1 | 0 |
| Soft part tumours | 4 | 4 | 2 | 6 | 4 | 2 |
| Skintumours | 0 | 2 | 0 | 0 | 5 | 0 |
| Splenietumours | 0 | 1 | 0 | 0 | i | 2 |
| Cardiactumours | 0 | 0 | 0 | 2 | 2 | 0 |
Statistically significant difference from control: *0.01 < p < 0.05; **p < 0.01.
f67, 70] in two studies on mouse embryos also found that irradiation on day 12 (0.5—2 Gy) or days 11—13 (0.8 x 3 Gy) of gestation reduced the overall incidence of neoplasms, although a third study (days l 1—13, 100 R x 3) did not support this [69].
An interesting experiment involving foetal mouse brain has recently been published by Plendl et al [58]. They found that prenatal irradiation (1 Gy on day 14 of gestation) inhibited tumour development after subsequent xenotransplantation of glioma cells into the mouse brain. Thus, it would appear that prenatal irradiation of the brain has created a tumour inhibitory field, also affecting the transplanted tumour cells. It should, however, be noted that a similar phenomenon has been demonstrated also in adult mice irradiated immediately prior to subcutaneous inoculation of sarcoma cells [2]. To clarify the tumour inhibitory effect of prenatal irradiation on the mouse brain, it would be desirable to create the same carcinogen induced brain tumour model as that described above in rats. It is therefore of interest that a couple of groups recently have managed to induce brain tumours with ENU in certain mouse hybrids [12, 66]. Hopefully, further experiments will determine whether preceding irradiation has the same effects on these tumours in mice as it has in rats.
Conclusion
Tumour development is known to be restricted to tissues with a capacity for cell proliferation. Nevertheless, it appears that during periods of strong proliferative activity the organism has an increased capacity to prevent developmental deviations such as cancer. The mechanisms it utilizes appear to be similar to those regulating normal development.
In addition, certain studies indicate that these tumour inhibitory mechanisms under certain conditions can be activated after a carcinogenic insult. Thus, a prenatal carcinogenic insult can be not only tumour producing but possibly also tumour inhibitory. Which result predominates seems to depend on the nature of the carcinogen, on the target organ, and on its development stage. If active development is always accompanied by a similarly active protection against developmental deviations—and less active development by a similarly less active protection—then tumour induction, throughout life, is the result of an inability to retain the delicate balance between developmental activity and protection against developmental deviation.
REFERENCES
- Alexandrov,A. and Napalkov, NT . Experimental study of relationship between teratogenesis and carcinogenesis in the brain of the rat. Cancer Lett. 1976; 1: 345.
- Anderson,E., Tokuda, S., Williams, W.L. and Warner, N.L. Radiation induced augmentation of the response of A/J mice to SaI tumor cells. Am. J. Pathol. 1982; 108: 24.
- Arseneau,C., Fowler, E. and Bakemeier, R.F. Synergistic tumorigenic effect of Procarbazine and ionizing radiation in (Balb/c x DBA/2)F1 mice. J.Natl. Cancer Inst. 1977; 59: 423.
- Bachmann, K.D. and Kroll W. Der Wilms, Tumor im ersten Lebenshajr, inbesondere uber 62 Nephroblastome des Dtsch. Med. Wochenschr. 1969; 94: 2598.
- Bader,L. and Miller, R.W.U.S. cancer incidence and mortality in the first year of life. Am. J. Dis. Child. 1979; 313: 157.
- Benjamin, S.A.,Lee, A.C., Angleton, G.M., Saunders, WI., Miller, GJ’t., Williams, S., Brewster, R.D. and Long, R.I. Neoplasms in young-dogs after perinatal irradiation. J. Natl. Cancer Inst. 1986; 77: 563.
- Bill, A.H. The regression of neuroblastoma. J. Pediatr. Surg. 1968; 3: 103.
- Brent,L. Radiation Teratogenesis. Teratology 19fi0; 21: 281.
- minster, R.L. Theeffect of cells transferred into the mouse blastocyst on subsequent J. Exp. Med. 1974; 410: 1049.
- Court,Brown M., Doll, R.R. and Bradford, Hill A. Incidence of leukemia after exposure to diagnostic radiation in utero. Br. Med. J. 1960; 2: 1539.
- Dekaban,S. Effects of x-radiation on mouse foetus during gestation: emphasis on distribution of cerebral lesions, pan II. J. Nucl. Med. 1969; 10: 68.
- Denlinger,H., Koestner, A. and Wechsler, W. Induction of neurogenic tumors in C3HeB/FeJ mice by nitrosourea derivatives: observations by light microscopy, tissue culture and electron microscopy. Int. J. Cancer. 1974; 13: 559.
- Diamond,, Schmerler, H. and Lilienfeld, A.M. The relationship of intrauterine radiation to subsequent mortality and development of leukemia in children. A prospective study. Am. J. Epidemiol. 1973; 97: 283.
- Donnellan, W.A. and Swenson, O. Benign and malignant sacroGoccygeal teratomas. Surgery1968; 64: 834.
- Einhorn, Are there factors preventing cancer development during embryonic life? Oncodevelopmental Biology and Medicine 1983; 4: 219.
- Einhorn, Can prenatal irradiation protect the embryo from tumor development? Acta Oncol. 1991; 30: 291.
- Emerson,C. and Cole, W.H. Spontaneous Regression of Cancer. Saunders, Philadelphia. 1966.
- Favara, B.E.Origins of cancer in the Am. J. Pediatr. Hematol. Oncol. 1981; 3: 187.
l9. Ferrer, J., Xumetra A. and Santa, Maria J. Cerebral malformation induced by prenatal x-irradiation: An autoradiographic and Golgi-study. J. Anat. 1984; 138: 81.
- Ford,D., Patterson, J.C.S. and Treuting, W.L. Foetal exposure to diagnostic X-rays and leukemia and other malignant diseases in childhood. J. Natl. Cancer Inst. 1959; 22: 1093.
- Friedberg, W.,Hanneman, G.D., Faulkner, D.N. et Fast neurton irradiation of mouse embryos in the pronuclear-zygote stage: mortality curves and neoplastic diseases in 30-day postnatal survivors. Proc. Soc. Exptl. Bio1. Med. 1976; 151: 808.
- Furth, I‘. and Boon, M.C. Enhancement of leukemogenic action of methylcholantrene bypre-irradiation with x-rays. Science 1943; 98: 138.
- Garwics, S.,Henrikson, H., Komfalt, A., Landberg, T. and Mortensson, W., Neuroblastom i Lund 1950-1974. Lkartidningen 1976; 73: 3980.
- Harvey, E.B., Boice, J.D., Honeyman, M. and Flannery, J.T. Prenatal X-ray exposure and childhood-cancer in N. Engl. J. Med. 1985; 3 l2: 541.
- Hasgekar, J.N., Pendse, A.M. and Lalitha, V.S. Effect of irradiation on ethylnitrosoureainduced neural tumors in Wistar rat. Cancer Letters 1986; 30:
- Hicks, S.P.Acute necrosis and malformation of developing mammalian brain caused by x-ray. Proc. Soc. Biol. Med. 1950; 75: 485.
- Hopton, P.A., McKinney, P.A., Cartwright,A., Mann, J.R., Birch, J.M., Hartly, A.L., Waterhouse, J.A.H., Johnston, H.E., Draper, G.J. and Stiller, C.A. X-rays in pregnancy and the risk of childhood cancer. Lancet 1985; 2: 973.
- Hoshino, H.,Tanooka, H. and Fukuoka, F. Summation of carcinogenic effects of 4-nitroquinoline 1-nxide and Q-rays. GANN 1968; 59: 43.
- Izant, Jr. R.J. and Filston, H.C. Sacrococcygeal teratomas. Analysis of forty- three cases. Am. J. Surg. 1975;130: 617.
- Jablon, S. and Kato, H. Childhood cancer in relation to prenatal exposure to atomicbomb radiation. Lancet 1970; 2: 1000.
- Kalter,, Mandybur, Ti I., Ormsby, I. and Warkany, J. Dose-related reduction by prenatal X-irradiation of the transplacental neurocarcinogenicity of ethylnitrosourea in rats. Cancer Res. 1980; 40: 3973.
- Kalter,, Ormsby, I. and Warkany, J. Congenital malformations of the central nervous system and transplacental carcinogenesis: Modification of ethylnitrosourea- induced brain tumors in rats by pretreatment with methylazoxymethanol. Int. J. Biol. Res. Pregnancy 1982; 3: 93.
- Knowles,F. Changing sensitivity of neonatal rats to tumorigenic effects of N- Nitroso-N-ethylurea and x-radiation. J. Natl. Cancer Inst. 1985; 74: 853.
- Knowles,F. Reduction of N-Nitroso-N-ethylurea-induced neurogenic tumors by x-radiation: A life-span study in rats. J. Natl. Cancer Inst. 1984; 72: 133.
- Knox,G., Stewart, A.M., Kneale, G.W. and Gilman, E.A. Prenatal irradiation and childhood cancer. J. Soc. Radiol. Prot. 1987; 7: 3.
- Kohn,I. and Fry, R.J.M. Radiation Carcinogenesis. N. Engl. J. Med. 1984; 310: 504.
- Laqueur, G.L. and Spatz, M. Transplacental Induction of Tumours and Malformationsin Rats with Cycasin and In: Transplacental Carcinogenesis. L. Tomatis and U. Mohr, editors. IARC Sci. mbl. No. 4, Lyon. 1973: 59.
- Little,B., McGandy, R.B. and Kennedy, A.R. Interactions between Polonium- 210 a-irradiation, Benzo(a) pyrene, and 0.99c NaCl solution instillations in the induction of expérimental lung cancer. Cancer Res. 1978; 38: 1929.
- MacMahon, Childhood cancer and prenatal irradiation. In: Cancer Aehievements, Challenges and Prospects for the 1980’s. Vol. 1. J.H. Burchenal and H.F. Oettgen, editors. Grune and Stratton, N.Y. 1981: 223.
- MacMahon, Prenatal X-ray-exposure and twins. N. Eng1. J. Med. 1985; 312: 576.
- MacMahon, B.Prenatal X-ray exposure and childhood cancer. J. Cancer Inst. 1962; 28: 1173.
- Mall, On the frequency of localized anomalies in human infants and embryos at birth. Am. J. Anat. 1917; 22: 27.
- Mandybur,I., Ormsby, I., Samuels, S. and Mancardi, G.L. Neural pituitary, and mammary tumors in Sprague-Dawley rats treated with x-irradiation to the head and N-ethyl-N-nitrosourea (ENU) during the early postnatal period—A statistical study of tumor incidence and survival. Rad. Res. 1985; 101: 460.
Marsden, H.B. and Steward, J.K. (Eds). Tumours in Children. Springer-Verlag, Berlin, 1976: Chapter 7, 10.
- Mintz, and Illmenzee, K. Normal genetically mosasic mice produced form malignant teratocarcinoma cells. Proc. Natl. Acad. Sci. USA 1975; 72: 3585.
- Mixer, H.W. andKirschbaum, Additive effects of X-rays and methycholantrene in inducing mouse leukemia. Radiology 1948; 50: 476.
- Mole,H. Antenatal irradiation and childhood cancer : causation or coincidence. Br. J. Cancer 1974; 30: 199.
- Monson, R.R. andMacmohon, B. Prenatal X-ray exposure and cancer in children. ln: Radiation Carcinogenesis : Epidemiology and Biological Significance (Boice,D. and Fraumeni, J.F. eds.) Raven Press, New York 1984 : 97.
- Napalkov, N.P., Rice, J.M., Tomatis, L. and Yamasaki, H. Perinatal and multigeneration IARC Sci. Publ. No. 96, Lyon, 1989.
- Napalkov,P. Some general considerations on the problem of transplacental carcinogenesis. In: Transplacenatal Carcinogenesis, L. Tomatis and U. Mohr, editors. ÏARC Scientific Publications No. 4, Lyon. 1973: 1.
- Nelson,, Oakley, G.P. and Shephard, T.H. Collection of human embryos an fetuses. In: Monitoring Birth Defects and Environment. E.B. Hook, D.T. Janerich and I.H. Porter, eds. Academic Press, N.Y. 1971: 45
- Nomura,, Nakajima, H., Hatanaka, T., Kinuta, M. and Hongyo T. Embryonic mutation as a possible cause of in utero carcinogenesis in mice revealed by posmatal treatment with 12-0-Tetradecanoylphorbo1-13-acetate. Cancer Res. 1990; 50: 2135.
- Nomura, An analysis of the changing urethan response of the developing mouse embryo in relation to moriality, malformation and neoplasm. Cancer Res. 1974; 3: 2217.
- Nomura, Induction of persistent hypersensitivity to lung tumorigenesis by in utero x-radiation in mice. Environmental Mutagen. 1984; 6: 33.
- Oppenheim, B.E., Griem, M.L. and Meier, P. Effects of low-dose prenatal irradiationin humans analysis of Chicago lying-in data and comparison with other Radiat. Res. 1974; 57: 508.
- Papaioannou, V.E., McBumey, M.W., Gardner, R.and Evans, M.J. Fate of teratocarcinoma cells injected into early mouse Nature 1975; 258: 70.
- Peraino, C., Gydina, DJ.and Carnes, B.A. Synergistic induction of altered hepatocyte foci by combined gamma-radiation and diethylnitrosamine administered to neonatal rats. Carcinogenesis 1986; 7:
- Plendl,, Schmahl, W. and Heinzmann, U. The effect of fetal irradiation on the growth of postnatally xenotransplanted tumour cells. Int. J. Radiat. Biol. 1989;
- Polhemus,W. and Koch, R. Leukemia and medical radiation. Pediatrics 1959; 23: 453.
- Rice,M. Editor. Perinatal Carcinogenesis. NCI Monograph No. 51. Washington DC. 1979.
- Rice,M. Effects of prenatal exposure to chemical carcinogens and methods for their detection.In: Developmental Toxicology. C.A. Kimmel and J. Buelke- Sam, eds. Raven Press, N.Y. 1981: 191.
- Rose,M. and Wallingford, H.M. Transformation of renal tumors of frogs to normal tissues in regenerating limbs of salamanders. Science 1948; 107: 457.
- Rugh, R., Duhamel, L. andSkaredoff, L. Relation of embryonic and fetal X- irradiation to life time average weights and tumour incidence in Proc. Soc. Exp. Biol. Med. 1966: 121: 714.
Sasaki, S., Kasuga, T., Sao, F. and Kawashima, N. Late effects of foetal mice X-irradiated at middle or late intrauterine stage. GANN 1978; 69: 167.
- ,Avinskaya,I. Transplantability of tumors during various periods of the individual development of the animal. In: Zabolevanie Vyzdorovlenic Moscow. 1952: 323. (Translation No. RTS 4059, British Library, Lending Division.)
- Schamahl, W., Luz, A., Leierseder-Bauer, and Neuhuser-Klaus, A. Diaplacental induction by ethylnitrosourea of tumours at the pial border of the CNS (TxHT)F1 mice, accepted for publication, Carcinogenesis 1990; 11: 1913.
- Schmahl,, Kriegel, H. and Senft, E. Can prenatal x-irradiation in mice act as an initiator stimulus in a modified 2-stages BerenblumfMoteam experiment with postnatal promotion with phorbol ester TPA. J. Cancer Res. Clin. Oncol. 1980; 97: 109.
- Schmahl, and Kriegel, H. Correlations between the degree and type of forebrain malformations and the simultaneous neurooncogenic properties of ethylnitrosourea after diaplacenatal exposure in rats, alone and in combination with X-irradiation. Teratogenesis, Carcinogenesis and Mutagenesis 1985; 5: 159.
- Schmahl, W.and Kriegel, Oncogenic proporties of transplacentally acting Ethylnitrosourea in NMRI-mice after antecedent x-irradiation. Z. Krebsforsch. 1978; 91: 69.
- Schmahl, Long-term effects after prenatal irradiation. In: Effects of Prenatal Irradiation with Special Emphasis on Late Effects. EUR-8067 1984: 89.
- Schmahl, Synergistic induction of tumors in NMRl mice by combined foetal X-irradiation with low dose and ethylnitrosourea administered to juvenile offspring. Carcinogenesis 1988; 9: 1493.
- Segaloff, and Maxfield, W.S. The synergism between radiation and eseogen in the production of mammary cancer in the rat. Cancer Res. 1971; 31: 166.
- Seilem-Aspang, F.and Kratochwil, K. Experimental activation of the differentiation ability of malignant cells. Wein. Klin. Wschr. 1963; 75: 337.
- Seilem-Aspang, F. and Kratochwil, Induction and dirferentiation of an epithelial tumor in the Newt {Triiurus cristacus). J. Embryol. Exp. Morphol. 1962; 10: 336.
- Shiono,H., Chung, C.S. and Myrianthopoulos, N.C. Preconception radiation, intrauterine diagnostic radiation and childhood neoplasia. J. Natl. Cancer Inst. 1980; 65: 681.
- Sikov,R. and Lofstrom, J.E. Increased tumour incidence following prenatal x-irradiation of the rat. In: Radiation Effects in Physics, Chemistry and Biology (Eberts, M. and Howard, A. eds.), North Holland, Amsterdam 1963: 372.
- Sikov,R. Carcinogenesis following prenatal exposure to radiation. Biol. Res. in Pregnancy 1981; 2: 159.
- Singh, R.P. and Carr, D.H. Anatomic findings in human abortions of known chromosomal Obstet. Gynecol. 1967; 29: 806.
- Stewart,M., Webb, J., Giles, D. and Hewitt, D. Malignant disease in childhood and diagnostic irradiation in utero. Lancet 1956; 2: 447.
- Stewart, A.M.,Webb, J. and Hewitt, A survey of childhood malignancies. Br. Med. J. 1958; 1: 1495.
- Streltsova, V.N.and Pavlenko-Mikhailov, N. On tumours arising in animals exposed to irradiation in their embryogenesis period. Vopr. Onkol. 1978; 24: 25.
- Surveyof neuroblastoma among the surgical fellows of the American Academy of J. Pediatr. Surg. 1968; 3: 191.
- Tomatis, and Mohr, U. (Eds). Transplacental Carcinogenesis. IARC Scientific Publications No. 4, Lyon. 1973.
- Totter,R. and MacPherson, H.G. Do childhood cancers result from prenatal X-rays? Health Phys. 1982; 40: 511.
- Tsonis, P.A. and Eguchi, G. Abnormallimb regeneration without tufnor production in adult newts directed by carcinogens, 20-methylcholanthrene and benzo(a) pyrene. Develop. Growth 1982; 124: 183.
- Tsonis, P.A. and Eguchi, G.Carcinogens on regeneration. Effects of MNNG and 4NQO on limb regeneration in adult newts. 1981; 20: 52.
- Tsonis, P.A. and Eguchi. Effects of a carcinogen, MNNG, on blastema cells and blastema formation in newt limb regeneration. Develop, Growth Differ. 1983;25: 201.
- Umemura, S., Yoshida, J., Kobayashi, T., Kameyama, Y. and Kageyama, N. Effectof irradiation to carcinogenesis by transplacental ethylnitrosourea in BD- IX rats. No To Shinkei 1978; 30:
- United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR), Genetic andSomatic Effects of Ionizing Radiation, New York 1986: 16—18, 325—341.
- UNSCEAR,Sources, Effects and Risks of Ionizing Radiation, New York 1988:
427—429
- Upton, A.C., Jenkins, V.K., Walburg, H.E.,Tyndall, R.L., Conklin, J.W. and Wald, Observations on viral, chemical, and radiation-induced myeloid and lymphoid leukemias in RF mice. Natl. Cancer Inst. Monograph. 1966; 22: 328.
- Vesselinovitch, S.D., Simmons, E., Rao, K.VG. and Michailovich, Carcinogenic hazards of irradiation during embryonal developmental in mice. Radiat. Res. 1974: 59: 101.
- Warkany, J., Mandybur, T.I.and Kalter, H. Oncogenic response of rats with X-ray induced microcephaly to transplacental J. Natl. Cancer Inst. 1976; 56: 59.
Webb, C.G., Gootwine, E. and Sachs, L. Developmental potential of myeloid leukemia cells injected into midgestation embryos. Developm. Biol. 1984; 101: 221.
Wegner, G. and Graul, E.H. Tumoren als Sp(tsch{den der Nachkommenschaft von Wistar-Ratten nach Ganzkirperbestrahlung am 9. Gravidit { tstag. Strahlentherapie 196; 123: 609.
Willnow, U. Tumoren im Kindesalter. Z. Urol. Nephrol. 1980; 73: 935. Willis, R.A. The Borderland of Embryology and Pathology London: Butterworths, 1962: chapters 3, 11.
Wilson, J.G., Brent, R.L. and Jordan, H.C. Neoplasia induced in rat embryos by roentgen irradiation. Cancer Res. 1952; 12: 222.
Yokoro, K., Sunni, C., Jto, A., Hamada, K., Kanda, K. and Kobayashi, T. Mammary carcinogenic effect of low-dose fission radiation in Wistar/Furth rats and its dependency on proclactin. J. Natl. Cancer Inst. 1980; 64: 1459.
Yoshmoto, Y., Kato, H. and Schull, W.J. Risk of cancer among children exposed in utero to A-bomb irradiation 1950—84. Lancet 1988; 2: 665.
4. Genetic Effects of Radiation and Offspring Cancer
Taisei Nomura
Department of Radiation Biology, Faculty of Medicine Osaka University, Osaka, Japan
Introducti6n
Animal experiments anticipated that parental exposure to radiation and chemicals would induce varieties of defects in the offspring including tumours and malformations [15, 17, 18, 20, 33]. However, these findings have not been supported by a large scale epideminogical survey of the children of atom-bomb survivors in Hiroshima and Nagasaki until the age of 20 [361, although the case control studies in the United States [10, 30] and China [31] suggested a higher risk of leukaemia and childhood cancer in the children of mothers and/or fathers who received preconceptional diagnostic X-ray exposure. Recently, Gardner et a1 [9] documented a 6 to 7 times higher risk of leukaemia (mostly acute lymphatic leukaemia) in the children of fathers who were employed at the Sellafield nuclear reprocessing plant (in the UK) and exposed to more than 100 msv as external doses before conception, especially more than 10 msv during 6 months before conception, while the estimated external doses were about one-fourth and one-fourteenth, respectively, of that in Hiroshima and Nagasaki (433 msv) [24, 36] and radiation exposure at Sellafield was protracted for long period at low dose rate.
In this paper, previous animal studies on tumours and other defects in the preconceptionally irradiated offspring were reviewed and reanalysed to reconcile the problems that lie at the basis in the human studies.
Differential Sensitivity of Developing Germ Cells
Development of germ cells and experimental procedures are shown in the scheme (Fig. 1). In the testis, stem cells (spermatogonia) undergo meiosis (primary and secondary spermatocytes) after a period of mitotic proliferation and then differentiate into mature gametes from spermatids to spermatozoa. The development goes as if they are on the moving side-walk. Adult male mice were exposed to X-rays l to 7 days before conception for the treatment of spermatozoa stage, 15 to 21 days for the spermatid stage, and 64 to 80 days or more for the spermatogonia stage. In the ovary, meiosis finished in part during foetal age, and oocytes and follicles undergo maturation. Meiosis starts again a few hours before ovulation. Adult females were also exposed to X-rays at various intervals before conception to treat oocytes at various follicular stages [17, 18, 21]. In addition to the treatment of adult ICR mice,
Fig. 1.
Scheme for the experimental procedures to detect fertility, embryonic deaths (dominant lethals), congenital anomalies, tumours etc. in the Ft offspring after parental exposure to X-rays and chemicals (Nomura, 1988 with permission). An estrous female mouse was mated with males in the evening. Next morning vaginal plug was examined to determine the day of conception, and males were removed thereafter. Conception (fertilization) occurs at the time of ovulation (about 2:00 on the plug day), when the mouse room is lit from 4:00 to 18:00 [26]. Male or female mice had been treated or not treated with X-rays at various intervals before conception. Ft offspring of irradiated and unirradiated mice were nursed in the same mouse room at 23—25°C with mouse diet CA-f and tap water. All mice were inspected every day until sacrifice at 8 months (ICR) or 12 months (NS). Right upper: embryonic deaths (dominant lethals) and living Ft foetuses of male ICR mice exposed to 5.04 Gy of X-rays at spermatozoa stage (examined on the 18th day of gestation). Right lower: malformed (kinky and short tail, left) and normal (right) offspring resuscitated and foster-nursed after the cesarian operation (from Nomura, 1988 with permission.)
the 8th day embryos, 14th day foetuses, neonates, and young mice (21 days old) were exposed to X-rays to treat primordial germ cells, spermatogonia and immature oocytes at different ages.
Mouse oocytes at early follicular stages are extremely sensitive to radiation. One Gy of X-rays were enough to kill these oocytes, resulting in sterility in 5 weeks after irradiation [17, 21J. Especially, immature oocytes at neonatal and young ages were much more sensitive to radiation killing [5, 17, 21, 28]. Histologically, there were no oocytes in the irradiated mouse ovary, while human oocytes were highly resistant to 2 Gy and more doses of radiation [3]. These differences in the radiation sensitivity between mouse and human oocytes may be caused by the 10 fold difference in the cytoplasm (much repair enzymes), suggesting the difficulty to extrapolate radiation risk to human oocytes from mouse oocytes data.
For the male treatment, germ cells at a spermatogonial stage are very sensitive to radiation in both mouse and humans. Consequently, reduction of the sperm number occurs 7 weeks or 4 months after irradiation (2—5 Gy) in mice and men, respectively, resulting in temporal or permanent sterility (depending on gonadal doses) and also morphological abnormality of the survived sperm [12, 29] (Fig. 2).

Calendar Year
Fig. 2. Changes in the number of spermatozoa in Japanese fishermen exposed to radioactive fallout at Bikini in 1954. Total whole body gamma doses were 1.7 to 6.9 Gy [12] (from Kumatori et a1, 1980 with permission).
The Ft offspring derived from these irradiated germ cells developed embryonic deaths, congenital malformations, tumours, etc. [17, 18, 19, 20, 21, 23].
Embryonic Deaths
Preconceptional irradiation of ICR mice induced high incidence of dead embryos (dominant lethals) which correspond to abortion or miscarriage in human (Fig. 1). As shown in Fig. 3, dominant lethals scored as embryonic deaths per implant increased almost linearly with parental doses of X-rays at the postmeiotic stages of male germ cells (spermatozoa and spermatids). Thus, father’s exposure to radiation can kill the offspring. X-irradiation of mouse oocytes at late follicular stages also increased the incidence of
Fig. 3.
Embryonic deaths and birth-rate in the Ft offspring of ICR mice preconceptionally exposed to X-rays. An estrous ICR female (63—65 days old) was mated with adult male ICR mice (63-65 days old) exposed to a single dose (closed symbols and solid line) or protracted dose (open symbols and dotted lines) of X-rays at spermatozoa (1—7 days before conception; Y, 9); spermatid (15—21 days; P, V), or spermatogonia (64- 80, days; M, O) stage. Adult female ICR mice (63—65 days old) were also treated with a single (D) or protracted (O) dose of X-rays at late follicular stage (1-14 days before conception). The upper panel shows the average frequency of embryonic deaths (early deaths) per implant as an indicator of dominant lethals, and the lower panel shows the average number of live-births, per dam. Figures around the symbols indicate the number of parents examined. Doses of X-rays to parental ICR mice were given on the abscissa. A Toshiba KC—18—2A (Toshiba Medical Co., Ltd., Tokyo, Japan) was used for irradiation, operating at 20 mA and 180 KVp with a filter of 0.5 mm of aluminum and 0.5 mm of copper. Dose rate was 0.72 Gy/min at 45 cm distance (measured by Fricke dosimetry). For protracted irradiation, 0.36 Gy of X-rays was given at 2 hr intervals. embryonic deaths. However, there was no increase of embryonic deaths at
2.16 Gy but a litile increase at 5.04 Gy, if at all, after the spermatogonial irradiation. Doubling dose (doses double the spontaneous incidence) was extremely large (3.2 Gy) for spermatogonia, while these were approximately
0.3 Gy, 0.24 Gy and 0.39 Gy for spermatozoa, spermatids, and oocytes at late follicular stages, respectively (Table 1). Spermatogonia with large
Table 1. Induced rate and doubling dose of embryonic deaths (dominant lethals) in F, offspring of ICR mice exposed to X•rays before conception
| Induced rate/Gy
(x 102) |
Doubling dose
(Gy) |
|
| Spermatozoa | 6.7 (7.8) | 0.38 (0.33) |
| Spermatids | 10.9 (11.7) | 0.24 (0.22) |
| Spermatogonia | 0.24 – 0.8 (0) | 3.2 – 10 (00) |
| Oocytes | 6.5 (6.0) | 0.39 (0.43) |
Induced rates and doubling doses were calculated and averaged from the value at doses of 2.16 and 5.04 Gy for spermatozoa and spermatids, and at doses of 2.16 and
3.6 Gy for oocytes (Fig. 3). Doubling doses were calculated as uniparental exposure following Luning and Searle [13}. As for spermatogonia, two independent data at 5.04 Gy were used for calculation, since there was no increase of embryonic deaths at a dose of 2.16 Gy. Figures in parentheses are for the protracted irradiation by dose fractionation. Experimental procedures are given in the legend to Fig. 3. Two-thirds of data were derived from Nomura [17, 18, 21].
chromosomal changes, which cause dominant lethals, died during meiosis, and only the sperm without such damages could be ejaculated, resulting in no increase of embryonic deaths by the protracted irradiation of postmeiotic sperm. However, slight reduction was observed by the protracted irradiation of spermatogonia and oocytes (Fig. 3). Human oocytes must be much more resistant to radiation for embryonic deaths, i.e., dominant lethals, than mouse oocytes [21], because of more repair activity in human oocytes in parallel with their volume of cytoplasm. Neither prenatal and neonatal deaths nor resultant decrease of live-births were detected in the Ft offspring of atom- bomb survivors [11] which involved both spermatogonial exposure (in males) and immature oocytes exposure (of females). Negative results in human must be caused by the reduced sensitivities to radiation of these germ cell stages as indicated by the mouse experiment (Fig. 3). Consequently, genetic risk of abortions and stillbirths and consequent reduction of live-births should be carefully estimated in humans, because these are produced in the offspring only when the fathers are exposed to radiation during a few months before conception (at postmeiotic stages of male germ cells) [27].
Chemical mutagens also induced dominant lethals in mice, but with more stage-specific sensitivity [8].
Congenital Malformations
Parental exposure of ICR mice to X-rays and chemicals increased the incidence of prenatal and postnatal malformations which wefe detected just before birth (on the 18th day of gestation) or 7 days after birth, respectively 15, 17, 18, 21, 23). Morphological malformations (dwarf, cleft palate, tail anomalies, open eyelid, exencephalus, hydrocephalus, gastroschisis, etc.) increased with paternal doses of X-rays up to 2.16 Gy, but the incidence levelled off at a very high dose (5.04 Gy). In general, higher incidence of malformations was observed prenatally than postnatally, simply because many malformations were lethal shortly after birth (Fig. 4). However, it is noted that most lethal malformations in mice are viable in human after surgical operation [21]. The induced rate of congenital malformations per Gy by the postmeiotic exposure was – 2-3 times higher than that by the spermatogonia exposure (Table 2). High incidence of malformations was also detected by oocyte exposure. Malformation incidence was slightly reduced by the protracted irradiation of spermatogonia and oocytes but not of postmeiotic sperm [18]. As shown in Table 2, induced rate of malformation per Gy was about 4 times higher than that of dominant mutation affecting
Table 2. Incidence (9•) and induced rate per Gy (x1H) of congenital malformation in the Ft offspring (before and after birth) of ICR mice exposed to X-rays before conception in comparison with those of dominant skeletal mutation
Congenital malformation Skeletal mutation (Dose : 2.16 Gy) (Dose : 6 Gy)
| Before birth’ | After birthb | Before birth’ | After birthd | |
| Postgonia | 3.0% (11.6) | l.l3% (4.5) | 2.4% (3.3) | 1.8% (2.4) |
| Gonia | 1.8% (6.6) | 0%
1.ldc (1.9) |
1.1% (1.2) | 1.3% (2.1) |
| Oocytes | 2.4% (10,4) | 1.7% (6.2) | ||
| Control | 0.41% | 0.12% 0.429’r | ||
Figures in parentheses show the induced rate of congenital malformation and dominant mutation affecting skeletal bones which were detected before or after birth.
bNomura (1978, 1982, 1988); detected on the 18th day (day 19) of gestation or 7 days after birth.
*Bartsch-Sandhoff (1974); detected in day 19 foetuses.
dEhling (1966; 1984); detected 4 weeks after birth.
‘5.04 Gy.
skeletal bones, because overall anomalies were detected in my experiment [17, 21]. Doubling doses were 0.12, 0.27 and 0.19 Gy for acute irradiation of spermatozoa, spermatogonia and nature ‹xicytes, respectively [21]. However, acute l Gy exposure of spermatogonia increases only 0.2 of malformations. This indicates that congenital malformation will not be an adequate mark
Fig. 4. Incidence of congenital malformations in the F, offspring of male or female ICR mice treated with X-rays at spermatozoa, spermatogonia or mature oocyte stage. Adult male or female ICR mice {6$-65 days old) were treated at spermatozoa (1-14 days before conception), spermatogonia (64-180 days before conception). Malformations were examined prenatally (?, on the 18th day of gestation) or postnatally (?, 7 days after birth). Figures in parentheses show numbers of Ft foemses or live-born Ft mice examined. Vertical bars indicate 90% binomial confidence intervals. For untreated controls, 1967 foetuses and 809 live offspring were used. Details were given in Nomura, 1988 (from Nomura, 1988 with permission).
to estimate genetic risk of radiation in Hiroshima and Ngasaki, i.e., negligible increase of congenital malformations (about 0.l’7o) must be detected amongst high background incidence (4.7%) in the children of atom-bomb survivors who were exposed to the average dose of 0.435 Sv at the spermatogonial stage.
Recently, we reported that preconceptional treatment of male ICR mice with ethylnitrosourea (ENU) induced respiratory distress syndrome (RDS), i.e., asphyxia, in the offspring [21]. Although about half of RDS foetuses had specific morphological malformations (dwarfism and gigantic thymus), the remainders showed no morphological changes (Fig. 5).
Offspring showing functional defects (respiratory distress) after paternal exposure to ethylnitrosourea. An estrous ICR female was mated with adult ICR mice (63—65 days old) treated with ethylnitrosourea (ENU) 1—21 days or 90—180 days before conception so iliat postmeiotic stages or spermatogonial stage of male germ cells were treated. Pregnant mice were killed on the 18th day of gestation by cervical dislocation, and then foetuses were taken out by cesarian operation. Foetuses were resuscitated immediately after the cesarian operation by patting very gently with tissue paper and wiping amniotic fluid from nose, mouth and body surface. Foetuses were then classified as apneic or pneic. Incidence of apneic foetuses showing respiratory distress syndrome (RDS) was given on the ordinate. Left: post-meiotic treatment. Right: spermatogonial treatment. Figures upon the histogram indicate the number of foetuses examined. H; RDS with morphological malformations (dwarf, cleft palate, gigantic thymus, and exencephalus), Q; RDS without morphological malformation. ’p < 0.02, “p < 0.001 -[271
In our preliminary results, preconceptional X-ray exposure of male mice also induces RDS. Since functional defects like RDS are more commonly observed in human neonates than major morphological defects (Avery, 1987), the study should be extended both experimentally and epidemiologically to variety of physiological and biochemical defects.
Tumours in the Offspring
Parental exposure of ICR mice to X-rays induced a significant increase of tumours in the Ft offspring (Tables 3 and 4). Predominant type of tumours observcd in botlt irradiated and unirradiated offspring was lung tumour
Table 3. Incidence (9•) of leukaemia and other tumours in the Ft offspring of male ICR mice exposed to X•rays before conception
| State at | Dose | Leukaemia | Lung tumour | Others |
| exposure | (Gy) | (%1 | (%) | |
| A. Single exposure | 0.36 | I/141 (0.71) | 5/141 (3.5) | |
| postgonia | ||||
| 2.16 | 3/356 (0.84) | 36/356 (10.1)’ | 6 OC, 1 ST | |
| 5.04 | 2/173 (1.16) | 26/173 (15.0)’ | 2 OC | |
| Spermatogonia | 0.36 | 3/132 (2.3) | 6/132 (4.5) | 4 OC |
| 2.16 | 1/276 (0.36) | 22/276 (8.0)b | 2 OC | |
| B. Protracted | 5.04 | 0.412 (0.0) | 5/41 (12.2) | 2 OC |
| exposure | 0.36 x 6 | l/170 (0.59) | 13/170 (7.6) | 5 OC |
| postgonia
Spermatogonia |
0.36 x 14
0.36 x 6 |
0/105 (0.0)
— |
20/105 (19.0)a
— |
3 OC |
| 0.36 x 14 | 0/135 (0.0) | 7/135 (5.2) | 1 OC | |
| C. Controls | 0 | 2J548 (0.36) | 26/548 (4.7) | 1 OC |
| 0 | 0J4 (0.0) | 14J431 (4.1) | 1 OC, IRT |
Adult ICR males were exposed to 0.36, 2.16 and 5.04 Gy of X-rays at a dose rate of 0.72 Gy/min l to 28 days or more than 64 days before conception for post- spermatogonial (postgonial) or 0.36 Gy of X-rays were given at 2 hr intervals. Mice were sacrificed 8 months after birth. Details for experimental procedures were given in the legends to Fig. 1. All ovarian cysts (> 4.0 mm) and two small lung tumours (adenomas: A single exposure to 2.16 Gy at postgonia) were included in the present reanalysis. Two controls during the experiments (1971 to 1976) were listed in the table; controls from 1971 to 1974 (upper; Nomura, 1975; 1976; 1978; 1982) and those from 1974 to 1976 (lower). The abbreviations used are: OC—ovarian cystoadenoma, ST—stomach tumour (adenocarcinoma), RT—reticulum cell enoplasia.
’p < 0.01, b p = 0.06, p < 0.05 by test vs unirradiated controls. -test was applied with Yates’ correction, when the figure in a cell is less than 5.
(about 90%). The remainders were lymphocytic leukemia, ovarian tumour ilnd stomach tumour. The incidence of lung tumours in the FI offspring increased almost linearly with X-ray doses to paternal ICR. Lung tumours were observed less frequently in the offspring after spermatogonial exposure than post-spermatogonial exposure. Although there was no increase of tumours
in the Ft offspring after maternal exposure to X-rays up to 1 Gy, a large
increase was observed with higher doses (Table 4).
Table 4. Incidence (to) of leukemia and other tumors in the Ft offspring of female ICR mice exposed to X-rays before conception
| A. Single exposure
0.36 |
0/144 (0.0) |
6/144 (4.2) |
1 OC |
| 1.08 | 0/73 (0.0) | 4/73 (5.5) | l OC |
| 2.16 | 0/231 (0.4) | 31/231 (13.4)‘ | 2 OC |
| 3.6 | 1/66 (0.0) | 9/66 (13.6)’ | l OC |
| 5.04 | 2J10 (0.0) | 2/10 (20.0) | I OC |
| B. Protracted exposure
0.36 x 6 |
1/100 (1.0) | 7/100 (7.0) | 1 OC |
| 0.36 x 6 | 1/137 (0.7) | 8/137 (5.8) | 1 OC |
| 0.36 x 10 | 0/157 (0.0) | 11/157 (7.0) | |
| 0.36 x 14 | 0/29 (0.0) | 1/29 (3.4) | |
| C. 0.36b | 0/159 (0.0) | 10/159 (6.3) | 1 OC |
| D. Controls | |||
| 0 | 2/548 (0.36) | 26/548 (4.7) | 1 OC |
| 0 | 0/341 (0.0) | 14/341 (4.1) | 1 OC, RT |
Adult ICR females were exposed to X-rays 1 to 28 days before conception. Details are given in the legend to Table 3.
’p < 0.01.
Exposed to X-rays more than 64 days before conception (adult females remain fertile at 0.36 Gy).
When X-ray doses were fractionated to small amounts (0.36 Gy) and given protractedly at 2 hr intervals, there was no increase of tumour incidence after spermatogonia and oocyte exposure, indicating some repairs of X-ray damages in these germ cells (Tables 3 and 4). However, such reduction of tumour incidence by protracted irradiation was not observed when post- spermatogonial stages (spermatozoa and spermatids) were treated (Table 3) [17, 18]. Induced rates of lung tumours by acute irradiation were 2.2d, 1.49 and 3.17 x 10*°/Gy for post-spermatogonial, spermatogonial and oocyte exposure, respectively (calculated from Tables 3 and 4 and averaged at doses 2.16 and 5.04 Gy for male exposure and 2.16, 3.6 and 5.04 Gy for female exposure), and those by protracted irradiation were 2.09, 0.09 and
0.29 x lW2/Gy, respectively. These germ-line alterations causing lung tumors transmitted to the next generation as a dominant mutation with reduced penetrance or expressivity (Nomura, 1982; 1986). Since such dominant inheritance of lung tumors was not observed in the offspring of unirradiated ICR mice, germ-line alterations must be novel mutations produced in this mouse population [18, 20, 22]. The same result was observed in other strain of mice, N5 [20, 22].
To compare with human studies, leukemia incidence in the Ft offspring was also listed in Tables 3 and 4. Spermatogonia X-irradiation of ICR mice did not increase the incidence of leukemias in the Ft offspring, while post- spermatogonial irradiation resulted in slight (but not significant) increase of leukaemia incidence (acute lymphocytic leukaemia) wi’.h paternal doses of X-ray (Table 3). Treatment of female ICR mice with both acute and protracted doses of X-rays did not increase the incidence of leukemias in the Ft offspring (Table 4). Thus, spermatogonia and oocytes of ICR strain were highly resistant to radiation-induced germ-line alteration causing leukemia. These mouse data reconcile, in part, the negative result of leukemia incidence in the children of atom-bomb survivors (until the age of 20) who were exposed at the spermatogonia and immature oocytes stages [24].
Table 5 gives the comparison of tumors in the F, offspring after male and female germ cells exposure to 2.16 Gy of X-rays at different ages. There
Table S Comparison of tumour incidence in the Ft offspring of ICR mice exposed to 2.16 Gy of X-rays before conception at various germ cell stages at different ages
| Age at exposure | Sex | Leukaemia | Lung tumour | Others |
| 8th day embryo | Male | 1/122 (0.8) | 11/122 (9.0) | |
|
14th day foetus |
Female
Male |
0J28 (0.0)
0/34 (0.0) |
2J28
0f34 (0.0) |
— 1 OC |
| Female | 1/27 (0.0) | 2/27 (7.4) | — | |
| Young (21 days) | Male
Female |
1/55 (1.8) | 3/55 (5.5) | l ScT |
Male ICR mice were exposed to 2.16 Gy of X-rays at embryonic, or young age and then mated with untreated ICR females about 10 weeks after irradiation. Ft offspring were sacrificed 8 months after birth to examine tumours. For adults and controls, see Tables 3 and 4. ScT, subcutaneous tumour (fibrosarcoma). As for congenital malformations, scc Nomura (1978; 1985).
were no substantial differcnces in the incidence of leukaemia and other tumours at treatcd ages except at the 14th day of foetal age (Nomura, 1983). Since male primordial germ cells in the genital ridge of the 14th day foetus were extremely sensitive to X-rays (Nomura, 1983) and chemicals (Nomura, 1983), selective cell killing might cause the difference (Nomura, 1983).
A majority of lymphocytic leukaemias (15 of 17) observed in the Ft of irradiated ICR mice (Tables 3, 4 and 5) originated in the thymus, while both in controls did not. Four mice died of acute (thymic) lymphocytic leukaemias 3, 6, 8 and 8 months after birth. Remainders were detected at the time of sacrifice.
Further Studies with Other Strains of Mice
Predominant types of tumours induced in LT and N5 offspring were found
in the lung after parental exposure to X-rays as in the case of ICR strain. However, varieties of other solid tumours and leukaemias were also observed in the F offspring of irradiated LT and N5 males, e.g., reticulum cell neoplasia, uterine hemangioma, ileum tumour, stomach tumour, subcutaneous tumour, hepatoma, lipoma, undifferentiated tumours, etc. [20). Preliminary report by Takahashi et al [32a also showed about 10 times higher incidence of hepatomas in the B6C3Ft mice exposed preconceptionally to 0.5 Gy of 2’°Cf radialions (about 65% neutron). Consequently, most of tumours induced in the F t offspring by parental irr;tdiation were adult type tumours which were commonly observed in these mouse strains. Germ-line alterations causing tumours may elevate (for the most part) the incidence of various kinds of commonly observed tumours. Such changes might be concerned with an array of genes, e. g. antioncogenic or suppressor genes, modifying immunological, biochemical and physiological conditions which control the tumour incidence [17].
Preconceptional irradiation of ICR mice did not induce lymphocytic leukaemias in the Ft offspring, except for the treatment at the post- spermatogonial stages. Furthermore, the incidence of leukaemia induced by the post-spermatogonial irradiation was very low (Table 3). However, different strains LT and N5 developed significantly high incidence of lymphocytic leukaemias in the F; offspring after paternal exposure to X-rays [20, 25]. When male LT mice were exposed to 5.04 or 3.6 Gy of X-rays at spermatozoa stage, preliminary results indicated 7.4 or 4.5 times higher incidence of leukaemias in the F offspring than that of unirradiated controls, respectively, while spermatogonial treatment with 3.6 Gy of X-rays resulted in no increase of leukaemia as in the case of ICR strain [20, 25].
In contrast to ICR and LT strains, however, N5 strain developcd about
10 times higher incidence of leukaemias in the F; offspring after spermatogo’nial exposure to 5.04 Gy of X-rays (Table 6). Furthermore, preliminary results showed about 18 times higher incidence of leukaemias after X-ray exposure (5.04 Gy) at spermatozoa stage. Thus, there are huge differences in the sensitivity to leukaemia induction by radiation in F, among used strains of mice ICR, LT and N5, in addition to the large difference in the sensitivity between treated stages of male germ cells. The large difference in leukaemia incidence in offspring among thc three mouse strains reflects tho difference in genetic predisposition to leukaemia induction by radiation.
The number of leukaemias per gamete per Gy was 0 to 6.9 x l0‘3 and 1.9
to 13.8 x 10°’ for the treatment of spcrmatogonia and spermatozoa, respectively (Table 7). These values were close to those of dominant skeletal mutations in mice (Table 2), but about 10 times higher than those of other mouse mutations (recessive specific-locus, dominant cataract and enzyme-activity mutations) which involve 7 to 15 gene loci [4, 7, 22].
52 Norm
Table 6. Incidence (to) of leukaemia and other tumours in the F; of male N5 mice exposed to X-rays before conception
| Stage at exposure | Dose | Leukaemia (Gy) | Lung tumour (%) | Others (to) | Anomalies tumours |
| Spermatozoa | 5.04 | 2/27 (7.4) | 5/27 (18.5) | 1 RT, 3 OC | |
| Spermatogonia | 5.04 | 9/229 (3.9)’ | 48/229 (21.0)b | 2 RT, 1 ScT, | 7 T, 2DW |
| 28 OC. 1 IT,
1 ST, lLi |
|||||
| Control | 0 | 1/224 (0.4) | 35/244 (14.3) | 3 He | 4 T, 2DW |
Adult male NS mice were exposed to 0.04 Gy of X-rays at a dose rate of 0.5 GyJ min during 2 weeks before conception for treatment of spermatozoa Ft ffspring were killed 12 months after birth to examine tumours and anomalies. Abbreviations used are: RT, reticulum cell neoplasia; OC, ovarian cysts (240 mm); lT, ileum tumour (lymphoma); ScT, subcutaneous tumour (fibrosarcoma); Li, lipoma; ST, stomach tumour (adenocarcinoma); He, hepatoma; ’I, kinky tail; Dw, dwarf.
Peliminary results at spermatozoa stage and some tumours (ScT, Li) were not reported in the previous paper (Nomura, 1986). Concurrent Ft controls of male N5 mice which had not been irradiated at spermatozoa and spermatogonia stages were combined. Seven of 12 lymphocytic leukaemias originated in the thymus, and b mice died of leukaemia before sacrifice.
‘p < 0.05, Up — 0.06 by ;t2-test with Yates’ correction.
Comparison with Human Studies
Epidemiological study in Sellafield and Shanhai showed higher risk of leukaemia by paternal exposure to radiation before conception, especially extremely high risk of postmeiotic exposure, but not by maternal preconceptional doses (Shu et al, 1988). Although these results are well explained by my mouse experiments [18, 24], there is no increase of leukaemia incidence in the Ft offspring of atomic-bomb survivors in Hiroshima and Nagasaki {36]. There are several reasons for the differences in human studies. A few Ft offspring of atomic bomb survivors in Hiroshima and Nagasaki were conceived during 6 months after bombing, i.e., most of F offspring were exposed to atomic bomb radiation at the spermatogonial stage (for males) or at immature oocyte stages (or females) which show the reduced sensitivity to radiation for leukaemia induction in the mouse experiment (Tables 3 to 6). In contrast to ICR and LT strains, however, high incidence of leukaemias was induced in the F, offspring of NS strain not only after postgonial exposure but also after spermatogonial exposure (Table 6). Suth a difference in genetic predisposition could also account for the different population response of Sellafield and Hiroshima/Nagasaki (24]. In fact, the incidences of acute lyymphocytic leukaemias and non-Hodgkins lymphomas in Japan are half to one-third of those in the UK, the incidences are also low in the actomic bomb survivors and their children, while most of leukaemias in the UK human studies are these types.
Table 7. Leukaemia in the Ft <spring after paternal exposure to radiation in
men and mice
Dose (msv)
Sellafield (Gardner et al, 1990)
Relative risk
Doubling Induced rate/msv dose (msv) (x106)
| All stages | * 100 | 6.24 (1.5-25.8) | 11.9 (2.5-126) | 22.2 (2.1-105) |
| postgonia‘ | 5—9 | 3.54 (0.3-38.9) | 1.5 (0. l- ) | 179 (0-2680) |
| * 100 | 7.17 (1.7-30.5) | 1.0 (0.2-8.8) | 260 (30-1250) |
Hiroshima and Nagasaki (Yoshimoto and Mabuehi, 1991) Spermatogonia 435 1.24 900
Mouse
ICR (Nomura, 1978, 1982)
Spermatogonia 360-5040 l
0.23
0
Postgonia 360-5040 1.9-3.2
LT (Nomura, 1986, 1991)‘
950b
1.9b
| Spermatogonia | 3600 | 1 | 0 | |
| Spermatogonia | 3600-5040 | 4.5-7.4 | 450 | 9.0 |
| N5 (Nomura, 1986, | 1991)G | |||
| Spermatogonia | 5040 | 9.6 | 300 | 6.9 |
| Spermatogonia | 5040 | 18.1 | 150 | 13.8 |
Doubling doses in mice and men were calculated as uniparental exposure in contrast to the previous paper (Nomura, 1990), because fathers and males were exposed to radiation in the Sellafield and mouse studies and conjoint parental gonad doses were used for the Ft study of the atom-bomb survivors. Induced rate of leukaemia per msv in Sellafield study was calculated by the formula; (background incidence of leukaemia relevant to Sellafield study, 53 x 10 ) (Gardner, M.J., personal communication) x (excess risk value)/(paternal dose, msv). External doses to employees were in the range of 100-150 msv for all stages; 5—9 and 10—15 msv for postgonia (Gardner, M.J., personal communication). Average doses used for the calculation in this table were 125, 7.5 and
12.5 msv respectively.
‘Fathers’ exposure during 6 months before conception indicates postspermatogonial exposure for the most part, but includes spermatogonial expsosure in part.
bAverage value at 2160 and 5040 msv.
‘Includes some unpublishied data (see text and Table 4).
It is also possible that early developing acute lymphocytic leukaemias were missed by different diagnostic classification or modified by environmental (microbial) factors. It is known in the mouse study that lymphocyte leukaemias (but not other tumours are suppressd by stress or silent bacterial infection (or its products) (Yokoro, K., personal communication).
Even if these possibilities are taken into account, however, more quantitative comparison of doubling doses and induced rate of leukaemia showed several disparity between the mouse and Sellafield human study. Doubling doses for the induction of leukaemia in the children of the workers at Sellafield were 30 to 150 times lower than those in my mouse experiment (N5) lower
the doubling dose, the higher the sensitivity (Table 7), assuming that the doses estimated by rilm badges well represent gonad doses in the Sellafield study. The induced rate of ieukaemia in Ft offspring per paternal dose of radiation was also 3 to 20 times higher in the Sellafield study (Table 7) but within the large variance of risk values, which is caused by the small sample size. Recently, no raised relative risk of leukaemia was found for father’s dose of ionizing ’radiation before conception in Dounreay nuclear installation, but with much smaller sample size [34].
Although the most recent study in the children of the atom-bomb survivors revealed slightly higher relative risk of leukaemia (1.24) and heritable tumours (1.19), an excess that was not statistically significant [36]. In my mouse experiment, most of tumours induced in the Ft offspring after parental exposure to radiation were adult type tumours in the lung, ovary, stomach, liver uterus, etc. Although leukaemia incidence has not been elevated in the children of the atom-bomb survivors in Hiroshima and Nagasaki because of reduced sensitivity of exposed germ cells, we must await to conclude that human beings are resistant to the atomic radiation for tumour induction in the offspring: The mouse experiments anticipate that adult type cancer could increase when this population reaches cancer-prone age.
Consequently, epidemiological elucidation should be carried out, especially for adult type tumours and other constitutive or functional disorders in the human populations exposed to atomic radiations at Hiroshima/Nagasaki and nuclear plants including those involved in Chernobyl and Kykshtym accidents, etc., those exposed to diagnostic or therapeutic radiations and dnigs, and those exposed to herbicides and other hazardous agents contaminating our planet [23].
Unfortunately, genetic markers which had been examined in Hiroshima and Nagasaki were proven to be inappropriate from our mouse experiments [21, 22, 23, 24]. Especially electrophoretic mutations of serum proteins are caused by base substitution which is rarely induced by radiation [4, 14, 22]. An analysis of radiation-induced DNA alterations, mostly deletions, must be important to reconcile high risk of tumours in mice and men and to estimate genetic risk of radiation to human. Such studies are also in progress in Japan.
Acknowledgements
This review was written by the support of the Inoue Prize for Science. The author would like to thank Drs. T. Kanzaki, H. Tanaka, T. Namba, H. Goloh, H. Nakajima for their assistance and help, and Drs. M.J. Gardner,
- Kumatori, K.Yokoro and K.Mubuchi for their advices to prepare the manuscript.
REFERENCES
- Avery,B. Neonatology, 3rd Edn., J.B. Lippincott Co., Philadelphia, 1987.
- Bartsch-Sandhoff, Skeletal abnormalities in mouse embryos after irradiation of the size. Humangenetic 1974; 25, 93—100.
- Blot,J. and Sawada, H. Fertility among female survivors of the atomic bombs in Hiroshima and Nagasaki. Atomic Bomb Casuality Commission Technical Report, 1971; 26: 1—21.
- Charles,J. and Pretsch, W. Enzymes-activity mutations detected in mice after paternal fractionated irradiation. Mutat. Res. 1986; 160: 243—248.
- Dobson, R.L.and Kwan, C. The tritium RBE at low-level exposure-variation with dose, dose rate, and exposure duration. Curr. Top. Radiat. Res. 1977; 12, 44-d2.
- Ehling,H. Dominant mutations affecting the skeleton in offspring of X-irradiated mice. Genetics 1966; 54: 1281—1389.
- Ehling,H. Methods to estimate the genetic risk. In: Mutations in Man, Obe,
- (ed)1984,’ pp. 292—318. Springer-Verlag: Berlin.
- Ehling,H. and Nauhauser-Klaus, A. Induction of specificlocus mutation and dominant lethal mutations in male mice by chlormethine. Mutat. Res. 1989; 227, 81—89.
- Gardner,J., Snee M.P., Hall, A.J., Powell, C.A., Downess, S. and Terrell,
J.D. Results of case-control study of leukaemia and lymphoma among young people near Sellafield nuclear plant in West Cumbria. Br. Med. J. 1990; 300, 423—429.
- Graham, S., Levin, M.L., Lelienfield, A.M., Schuman, L, Gibson, R. and Dowd, J.E. Preconception, intrauterine, and postnatal irradiation as related to leukemia. Natl. Cancer Inst. Monogr. 1966; 19, 347—371.
- Kato, Early genetic survey and mortality study. J. Radiat. Res. 16, 67—74.
- Kumatori,, Ishihara, T., Hirashima, K., Sugiyama, H., Ishii, S. and Miyoshi,
- Follow-up studiesover a 25-year period on the Japanese fishermen exposed to radioactive fallout in 1954. In: The Medical Basis for Radiation Accident Preparedness, Hubner, K.F. and Fry, S.A. (eds) 1980; pp. 33-54. Elsevier/North- Holland: New York.
- Luning, K.G. and Searle, A.G. Estimates of the genetic risks from ionizing irradiation. Res. l97l; 12: 291—304.
- Nee1,V., Satoh, C., Goriki, K., Asakawa, J., Fujita, M., Takahashi, N., Kageoka,
- andHazama, R. Search for mutations altering protein charge and/or function in children of atomic bomb survivors: final report. Am. J. Hum. Genet. 1988; 42: 663—676.
- Nomura, Transmission of tumours and malformations to the next generation of mice subsequent to urethan treatment. Cancer Res. 1975; 35: 264—266.
- Nomura, Comparison of tumour susceptibility among various organs of foetal, young, and adult ICR/Jcl mice. Br. J. Cancer 1976; 33: 521—534.
- Nomura, T.Changed urethan and radiation response of the mouse germ cell to tumour induction. In: Tumours of Early Life in Man and Animals, Severi L. (ed) 1978; 873—897. Pergia Univ. Press: Perugia.
- Nomura, Parental exposure to X-rays and chemicals induces heritable tumours and anomalies in mice. Nature 1982; 296: 575-577.
- Nomura, X-ray induced germ-line mutation leading to tumours. Its manifestation in mice given urethane post-natally. Mutat. Res. 1983; 121: 5M5.
- Nomura, Further studies on X-ray and chemically induced germ-line alterations causing tumors and malformations in mice. In: Genetic Toxicology of Environmental Chemicals, Part B: Genetic Effects and Applied Mutagenesis, Ramel, C. (ed) 1986; pp. 13—20. Alan R. Liss: New York.
2t. Nomura, T. X-ray and chemically induced germ-line mutation causing phenotypical anomalies in mice. Mutat. Res. 1988; 198: 301—320.
- Nomura, Role of radiation-induced mutations in multigeneration carcinogenesis. IARC Sci. Publ. 1989a; 96: 375—387.
- Nomura, Congenital malformations as a consequence of parental exposure to radiations and chemicals in mice. J. Univ. Occupat. Environ. Health 1989b; 11: 406—415.
- Nomura, Of mice and men? Nature 1990; 345: 671.
- Nomura, Paternal exposure to radiation and offspring cancer in mice: Reanalysis and new evidence. J. Radiat. Res. 1991; in press.
- Nomura, T., Hatt, S., Shibata, K. and Kusafuka, T. (1987). Killing of preimplantation mouse embryos by main ingredients of cleansers AS and Mutat. Res. 1987; 190: 25—29.
- Nomura,, Gotoh, H. and Namba, T. (1990). An examination of respiratory distress and chromosomal abnormalities in the offspring of male mice treated with ethylnitrosourea. Mutat. Res. 1990a; 229: 115—122.
- Oakberg,F. (1962). Gamma-ray sensitivity of oocytes of immature mice. Proc. Soc. Exptl. Biol. Med. 1962; 109: 763—767.
- Searle,G. and Beechey, C.V. (1974) Sperm-count, egg-fertilization and dominant lethality after X-irradiation of mice. Mutat. Res. 1974; 22: 63—72.
- Shiono,H., Chung, C.S. and Myrlanthopoulos, N.C. (1980). Preconception radiation, intrauterine diagnostic radiation, and childhood neoplasia. J. Natl. Cancer Inst. 1980; 65: 681—686.
- Shu, X.O., Gao, Y.T., Brinton, L.A., Linet, M.S., Tu, J.T., Zheng, W. and Fraumeni,F. Jr. (1988). A population-based case-control study of childhood leukaemia in Shanghai. Cancer 1988; 62: 635—644.
- Takahashi,, Watanabe, H., Dohi, K. and Ito, A. (1992). ^Uf relative bio1ogiea,t effectiveness and inheritable effect of fission neutrons in mouse liver mmorigenesis. Cancer Res. 1992; 52: 1948—1953.
- Tomatis,, Cabral, J.R.P., Likhachev, AJ. and Ponomarkov, V. (1981). Increased cancer incidence in the progeny of male rats Exposed to ethylnitrosourea. Int. J. Cancer 1981; 28: 475-478.
- Urquhart, J.D.,Black, J., Muirhead, M.J., Sharp, L., Maxwell, M., Eden, O.B. and Jones, D.A. (1991). Case-control study of leukaemia and non-Hodgkin’s lymphoma in children in caithners near the Dounreay nuclear installation. Br. Med. J. 1991; 302: 609—692.
- Yoshimoto, and Mabuchi, K. (1991). Mortality and cancer risk among the offspring (Ft) of atomic bomb survivors. J. Radiat Res. 1991; 32: 294-300.
- Yoshimoto, Y.,Neel, V., Schull, W.J., Kota, H., Soda, M., Eto, R. and Mabuchi,
- (1990).Malignant tumors during the first 2 Am. J. Hum. Genet. 1990; 46: 1041—1052.
5. Consequences of Low Level
Radiation—A review
B.B. Burlakova
Institute of Chemical Physics
Academy of Sciences of Russia, Moscow
The consequence of low level radiation is a topic currently debated and, investigated all over the world. This interest is not only due to the fascinating practical and theoretical dimensions of the problem but also due to larger environmental concerns.
Low dose radiation biology became a subject of interest and importance following N-bomb detonation for destruction, rapid proliferation of nuclear plants for energy, and nuclear waste disposal leading to radioecological problems with a direct bearing on living state. The study is also motivated by the awareness of additive effects of super low doses of chemicals and drugs on cells, organisms and humans.
Effects of low level radiation were studied both in vitro and in vivo with cells and organisms following Chernobyl accident. A number of effects were identified in biological systems within an area of 30 km radius following Chernobyl fall out. Results of investigation may be summed up as:
- Recognisablelevels of ‘dose-effect’
- Highvariability in sensitivity of biological systems to low dose (of the order of 20 times).
- Presenceof synergistic effects with confounding factors, which is particularly evident for low radiation
- Predictabletime interval for the onset of effects and possibilities of
- Dependenceof consequence on variables such as emotional status, living conditions, predisposition to spontaneous cancers, average life, health status
One of the basic tasks of radiobiology should be to define sensitive parameters for determining the effects of low dose radiation on population. It is extremely important as low dose radiation biology differs from that of high dose radiation. The effectiveness of seemingly similar doses of low level radiation are very often different.
For example, Szepeshi and Filender studied haematological changes in patients who suffered from accidents in atomic power station. Patients were divided into 6 groups on the basis of degree of damage, modality of treatment received and predilection. It was shown that in one group of people, radiation received differed by 20 folds. It was also shown that there was a decrease
58 BURLAKOVA
in heterogeneity of consequence at higher doses. A similar observation was made in ‘Liquidators’, whose blood content of Vit E and A was studied. The conclusion following evaluation of changes in transfer in blood studied by ESR were also similar. Heterogeneity in biological effects is seen at its peak following low dose radiation. Synergism mentioned above was noted in humans as well. For example the incidence of lung cancer in minors exposed to higher levels of radiation and, who also smoked was 4 times higher than in non-smokers. This synergism can be explained by the well established theory of inducer and, promoter of carcinogenesis.
The complex scenario presented above behooves us to analyse the effects of low level radiation on people who live in an area of elevated background radiation. There is a need to evolve a comprehensive model to understand elevated level of illness in population settled around atomic industry or in an area contaminated by radio-nucleides following accidents. Classical biology may be found wanting in explaining all the observed events in a single frame work.
It is by now common knowledge that a large area of Russia, Ukraine and Belorussia have been contaminated following Chernobyl accident. A survey by the medical staff has noted an increase in cardiovascular and neuro- physical disease besides thyroid dysfunction in children.
It is hence logical that a lack of precise dose-effect relation does not necessarily mean a lack of cause and effect relationship.
Synergism of confounding factors with ionising radiation is an area which is under active investigation. Synergistic action with radiation is noted in conjunction with carcinogens like pesticides, ozone, disturbed hormonal millieu etc. For example an increased effect of irradiation under simultaneous action of radiation and nitrates was demonstrated in the institute of nuclear energetics of Belorussia academy of sciences. An interaction of the two show a superadditive effect on number a of mutations in Drosophila as compared with simple additive mutagenic effects of the two.
Emotional stress as a synergistic agent with radiation was an unexpected finding. Free radical content when assessed with ESR in stressed and non- stressed animals showed synergistic effects all through the study, i.e. nearly for 300 hours. Free radical content in some way reflects the extent of bimolecular damage. It was noted that an increase in synergism between radiation and stress by a factor of 2, was present.
Representatives of the official medical staff refused to attribute to radiation any sharp deterioration of health in the case of people who worked in highly contaminated area. The media attributed their illness to “radiophobia”. As a proof a data was presented which did not show any dose-effect relationship. Our study in contrast has been conducted on the premise that there is difference in dose dependence of the degree of damage of biological targets on one hand and dose dependence of their repair capability on the other hand.
Small quantum of radiation may not activate repair system or may activate it only partly. Therefore the damage to biological structure at these low
Consequences of Low Level Radiation 59
dose levels may be greater than for higher doses at which the repair systems
can function effectively (Fig.1).
Dose barb . un i ts
Fig. 1. Dose-response dependence.
Repair systems due to increased dose can lead to situations where enhanced repair prevails over induced damage because of which no change in biological targets is perceived or sometimes a stimulatory effect is seen. This dose range is followed by one at which damage due to radiation is more than the repair systems can cope with, as a consequence of which damage to biological system becomes apparent. Thus the dose-effect dependence is bimodal perhaps polymodal. Figure 1 shows an example, i.e. scheme of 3 ranges of different dose effect.
Dublin in 1964 demonstrated that chromosomal aberrations were higher with low dose radiation than high dose rate radiation even when a similar total dose of radiation was delivered. Thus irradiation with doses of 2.6 and 3 Gy at a rate of 10 r per hour and 1810 r per hour produced a two fold increase in the number of chromosome aberrations in Wriths embryos under lower radiation intensity. This was assumed to be due to lack of induction of repair system.
Investigations in “adaptive response” are basically similar to the data presented above. Numerous experiments on plant animal cells as also in organisms have revealed the effect of increasing the sensitivity of cells and organisms to the subsequent action of high dose of radiation if they were previously irradiated with a low dose i.e. upto 25 r. It is important to note that either increase or decrease in the dose of prior radiation leads to loss of the adaptive response. Activation of repair system at a dose level of 5—15 r results in an increase in resistance to subsequent radiation due to
60 BURLAKovA
induction of repair mechanisms of cells. However, absence of adaptive response noted at a level of 1—10 r may be due to lack of repair mechanisms to become operational. This observation is extremely important while investigating the effects of low level radiation.
Dr. Spitkovsky from the Institute of Molecular Genetics demonstrated bimodal dependence of low dose radiation on chromatin abberration in lymphocytes. His results are similar to those of 7 other laboratories which conducted similar studies. The changes observed in these experiments were higher than those that could have been obtained by a linear extrapolation of effects due to high dose radiation. Dr. Spitkovsky interpreted his results as being due to certain proportions of programmable subpopulations in an heterogeneous group undergoing structural changes. Generally, this is followed by cell’s auto-induction of chromosomal anomalies or structural rearrange- ments which could lead to either better adaptation to altered environment or may trigger the mechanisms of oncogenesis and, other genetically connected disease.
Effects of radiation on cell membrane have been studied in our laboratory. The salient effects observed are, at low dose, radiation damage to membrane and membrane related functions are higher than genomic damage; repair system of membranes are activated at higher dose of radiation than those responsible for genomic repair, and finally the quantum of damage to membrane lipids increases with decreasing dose of radiation. Such bimodal effects of radiation on cell membrane were obtained by studying the effects of irradiation on physico-chemical and, biochemical parameters such as lipid antioxidative activity (AOA), lipid content, and membrane structure etc.
Changes in membrane lipids AOA of liver of rats trapped at various
distances (upto 30 km) from Chernobyl which thus received differing level of radiation, were studied. Activity of AOA and other cell membrane related functions were studied in the laboratory following irradiation of rats to a spectrum of dose levels as received by entrapped ones around Chernobyl. It was iaoted that the changes occurring at half lethal and at the 2 orders lower ones produced the same quantitative alterations in membrane lipid content, AOA, and lipid bilayer structure. It is apparent that cell membrane, which has the function of regulating cellular functions can be affected by a very low dose of radiation. The changes mentioned above were also seen in innumerable experimental studies conducted on animals living in the contaminated areas as well as those who were fed contaminated fodder. Biochemical and biophysical characteristics of cells obtained from liquidators and children living at the contaminated site of Chernobyl also showed such changes. Neifakh N.A. demonstrated a decrease in serum levels of antioxidants vitamin A and E in liquidators. It is possible that this decrease in absolute values of antioxidants in liquidators may reflect on their health status. It may be that they would become more susceptible to neoplastic disease.
The experiments did not show any simple classic dependence on dose but
Consequences of Law Level Radiation 61
significant changes of varying degree were shown in the liquidators who received the doses above 5 r. These differences are possibly due to changes in cell membrane characteristics.
It is known that mechanics of cell membrane repair involves elimination of damaged molecules and replacement with new molecules. Few of the damaged sites on the membrane are repaired without displacement but through repair system involving phospholipases, disulfareductases, ligases etc. Synthesis of membrane proteins and lipids is accelerated at a dose around 300 cCly. In this connection it may be surmised that low level radiation leads to enhanced changes in cell membrane, related functions. This may lead to many pathologic consequences in an organism. Pathogenesis is due to decrease in lipid antioxidant activity, fluidity of lipid content of cell membrane, disruption of intracellular regulatory mechanisms.
It may be noted that number of diseases of cardiovascular system, mental disorders, gastrointestinal diseases are preceded by decreased antioxidant activity which enhances the risk. The cell membrane damage also alters sensitivity of organisms and, animals to other genotoxic agents.
A definite increase in the number of diseases such as neurological, cardiovascular, and gastrointestinal diseases in contaminated area has been attributed to “radiophobia” or stress, by the official medical team. However, it may not be totally true. The observed changes in health status in Chernobyl area may be explained by bimodal expression of radiation damage and, susceptibility of cell membrane instead of classical radiobiology. The data acquired from liquidators and population residing around contaminated area has been analysed on the assumptions of bimodal behavior and cell membrane sensitivity at low dose of radiation.
APPENDIX
Analysis of the liquidators disease has revealed that there are statistically significant differences between I and VII groups of liquidators for disease groups, namely,
- Neurologicaldisease
- Haematologicaldisorders
- Diseasesof digestive system
- Musculo-skeletalorder
However, careful review of the same data of liquidators by us revealed a bimodal dependence of illness with radiation. The data from Art Medical Academy Yu.N. Shi-shmarev and A.M. Niuiforov on the health status of liquidators following Chernobyl accident was reviewed. The disease pattern closely resembled the data of Japanese N-bomb survivors.
Review of data from Chernobyl and other related studies suggests that a simple extrapolation of high dose radiation effect to low dose regions is
62 B RLAKOVA
inappropriate. The risk cœfficient for neoplastic transformation at lower doses is higher than what is generally assumed. It is more appropriate to assume low dose as the causative factor for deterioration in health status of population exposed to low level radiation following Chernobyl accident. Radiophobia and other environmental factors cannot account for sudden increase in illness.
The molecular mechanism involving cell membrane functions and, impaired repair system may be one of the explanations for our findings. However, the data which is accumulating neçds constant review, for more definitive conclusion.
6. International Assessment of Radiological Consequences of Chernobyl Accident
D.V. Gopinath
Health Safety and Environment Group
Bhabha Atomic Research Centre, Bombay, India
The major accidents that occurred at the Chernobyl Nuclear Power Plant on April 26, 1986 caused acute radiation injuries and death amongst the plant workers and fireman. It also led to radiation exposure to thousands of persons involved in the rescue and clean up operations. There has been severe radioactive contamination in the area within 30 km of the plant and contamination of different degrees of severity in extensive areas upto hundreds of kilometers from the site. This called for gigantic efforts on the part of USSR Government in delineating the contaminated areas, decontaminating and restructuring the environment to the extent possible and relocation of the people. Besides, over the past 5 years more than 50 laboratories in USSR have been involved in the studies on environmental contamination, radiaion exposure and health monitoring of the people in the area.
Understanding, there has been a serious concern world wide about the health and environmental impact of Chernobyl accident. Notwithstanding all the studies being conducted at USSR, information about the severity and significance of the health effects of the accident has been often sparse and uneven, public opinion has been uncertain and.even many doctors were not sure as to how to interpret the information that did becoriie available. In order to mitigate this situation the Government of USSR has on occassion sought the help of international expert bodies for an assessment on the consequences of the accident and effectiveness of the measures taken. Following are the observations of the expert bodies.
World Health Organisation (WHO)
At the request of USSR Government, the WHO sent a team of experts in 1989. The major conclusions of the team are :
“The expert group was requested to assess the concept of a lifetime dose of 35 rem as a limit following the accident. They agreed that this was a conservative value which ensured that the risk to health from this exposure was very small compared with other risks over a lifetime. The experts volunteered the view that, had they been requested to set a level for the lifetime dose, they would have chosen a value of the order of two to three times higher than 35 rem.”
64 GOPINATH
“It became very clear in the meetings that the public and scientists who were not specialists in radiation protection did not fully understand the principles involved. Scientists who are not well versed in radiation effects have attributed various biological and health effects to radiation exposure. These changes cannot be attributed to radiation exposure, especially when the normal incidence is unknown, and are much more likely to be due to psychological factors and stress. Attributing these effects to radiation not only increases the psychological pressure in the population and provokes additional stress-related health problems, but also undermines confidence in the competence of the radiation specialists. This has in turn led to doubts over the proposed values. Urgent consideration should be given to the institution of an education program to overcome this mistrust by ensuring that the public and scientists in allied fields can properly appreciate the proposals to protect the population.
Considerable concern was expressed over the possible synergistic effects of radiation and other environmental agents. The experts stated unequivocally that at the dose limit proposed, no synergistic effect could result [1]”.
League of Red Cross and Red Crescent Societies (LORCS) Again at the request of the Government of USSR, the LORCS sent a team of experts in 1990 and one of their main conclusions is : “Among the health problems reported it was felt that many of these, though perceived as radiation effects both by the public and by some doctors, were unrelated to radiation exposure. Little recognition appears to have been given to factors such as improved screening of population and changed patterns of living and of dietary habits. In particular, psychological stress and anxiety, understandable in the current situation, cause physical symptoms and affect health in a variety of ways. We fell that there is a need for more objective information in order to allay many of the fears of the population [2)”.
International Advisory Committee (31
In October 1989 USSR Government requested IAEA for an indepth review of the work carried out by the USSR institutions and also make an independent assessment of the short term and long term impact in the light of protective measures taken by USSR. As a result, an international project was launched in the spring of 1990. An independent “International Advisory Committee” of 19 members was set up under the chairmanship of Dr. Itsuzo Shigematsu, Director of the Radiation Effects Research Foundation in Hiroshima. The other scientists on the committee came from ten countries and five international organizations. The expertise encompassed, medicine, radiopathology, radiation protection, nutrition radioepidemiology and psychology.
About 200 independent experts from 23 countries and 7 international organizations were involved in the study, and 50 scientific missions visited the USSR. Laboratories in several countries, including Austria, France and the USA, helped to analyze and evaluate the collected material.
The international project, carried out in co-operation with local authorities, selected 28 settlements in the contaminated areas of concern to perform the necessary surveys. Some of the settlements were located in areas with relatively high soil surface contamination while other settlements were chosen in areas of relatively low soil surface contamination but with potential for high radiation doses to the people living there. Seven settlements were selected outside the contaminated areas for purpose of comparison.
The project received the full support of the USSR Government and the Governments of the BSSR, the RSFSR and the UkrSSR. There were open and frank conversations with authorities, scientists, and particularly local citizens that added to the international expens understanding of the situation.
Scope of the Study
The operaöon concentrated on the direct study of the environment and of the population in those möre highly contaminated areas that were still inhabited (with suitable comparative studies in areas of low contamination). In parallel with thèse detailed independent sampling studies, a general evaluation was made of existing Soviet methodology and findings.
Radiation Exposure of the Population
Different physical data were used in calculating extemal dose. Additional important data are biological “transfer factors” influencing the movement of element from the ground through food into the human body. Finally, people’s living conditions are important for assessing doses, such as housing, time spent outdoors and diet. The project concentrated on examining data for significant radioactive elements that can affect human health such as caesium, strontium and iodine. Some 8000 personal dosimeters were distributed to residents of seven such settlements and were carried for two months.
Independent whole body counting of radiocaesium levels was done by
project teams on more than 9000 people in nine settlements. Comparisons between project and USSR whole body counting facilities data were made. Independent calculations for the past and the future doses were made for the surveyed settlements using intemationally accepted calculation methods and starling from average values for deposition of radioactivity in the soil. Doses to the thyroid were reported on the basis of some early direct thyroid measurements as well as assumptions about iodine intake. For the seven contaminated settlements specifically studied by the project team, average radioactive iodine thyroid doses for children reported by the USSR varied widely. The major conclusions of this study are :
— The extemal dose to people from radionuclides deposited on the ground is generally the most significant contribution to total dose, especially in the areas where food restrictions are in force.
— Of the dosimeters wom for two months bJ inhabitants of the highly contaminated areas, only 10 percent registered exposures above the
minimum detection limit of 20 mrem. This corresponds to 120 mrem per year which is a typical dose an individual would receive from the natural environment.
- Thewhole body counting of radiocaesium generally indicated lower contents of that element in people’s bodies than would be calculated using theoretical models based on transfer from the environment, intakes from food and
- Independent calculations of past and future doses, based on average deposition results for the surveyed settlements,gave doses lower by a factor of 2—3 than the USSR estimates for the same
Healf h Effects
The International studies set about reviewing the health situation reported by key medical centres and institutes in the Soviet Union. Subsequently, seven representative settlements of high contamination were selected for detailed independent health examination by the project medical teams. Six control settlements with the same socioeconomic structure, but with insignificant contamination, were similarly examined.
The individuals to be examined were selected according to a statistical sampling scheme giving a representative distribution of age groups. As many as 250 people were examined in each settlement. The examination focused on disorders that had been reported or that might be expected.
In any detailed clinical study of a particular population, some health disorders are bound to be detected due to the better documentation and closer scrutiny such a detailed study entails. This makes careful comparison with a similar population outside the contaminated area even more critical and rules out reference to pre-existing national or regional statistics. The conclusions of this study are:
- Therewere significant non-radiation related health disorders in the population of both the contaminated and the non-contaminated settlements studied, but no health disorders that could be attributed directly to
- Theaccident had, and continues to have, considerable psychological consequences such as anxiety and uncertainty, which extended beyond the contaminated These consequences were compounded by the socio-economic and political changes in the USSR.
- The USSR data that were examined did not indicate a substantial increase in incidence of leukemia or cancer or hereditary effects. However,the data were not detailed enough to exclude a slight increase in the incidence of some tumour types. The adequately performed USSR studies, for their part, have not substantiated any of the reported health effects alleged to be due to radiation.
- Thechildren examined (mostly 2, 5 and 10 years old) were found to be generally The field studies indicated that a considerable
number of adults in both the contaminated and the comparative settlements had substantial medical problems of a general nature.
— No detrimcntal effects on growth due to voluntary or official dietary restrictions imposed as a result of the accident were found. There was no evidence of differences in thyroid function between contami- nated and comparison regions.
— Blood lead levels were found to be well within the normal range. Thus, no support was found for believing that the large quantity of lead dropped on the reactor to help control the accident had been widely dispersed in the atmosphere in vapor form and created a health hazard for the population at large.
— Review of USSR data indicated that reposed cancer incidence had been rising for the last decade. The rise started before the Chernobyl accident and has continued at the same rate since the accident.
— The data did not show a marked increase in leukemia or thyroid tumours since the accident. However, owing to limitations in the statistical methods, the possibility of slight increase in the incidence of these disorders cannot be excluded. Nor can the later development of increased numbers of cases after longer latent periods be excluded.
— There was no evidence of radiation induccd cataracts from special eye examination in the general population.
— High blood pressure was common among adults, but no differences were observed between the contaminated and the comparable settlements. Both resembled figures available for Moscow and Leningrad.
- Review of USSR data for settlements in the contaminated areas of concern, as wellas for the three affected republics as a whole indicated relatively high infant and perinatal mortality levels. These levels existed before the accident and appeared to be
- Nostatistically significant evidence was found of an increase in the incidence of foetal anomalies as a result of radiation
While the data were not detailed enough to exclude the possibility of an increase in some tumour types, it is emphasized that, on the basis of the doses estimated by the project and using internationally accepted risk estimates, future increases over the natural incidence of all cancers or hereditary effects would be difficult to discern, even with well designed long-term epidemiological studies. There remains a possibility of a statistically detectable increase in the incidence of thyroid tumours at a later date.
Conclusions
It must be emphasized that presentation of the above summary is not to dismiss Chernobyl accident as inconsequential nor even to underestimate its overall impact. The accident has entailed psychological, social and economic burdens at an unprecedented level. A significant fraction of persons involved
in the rescue and clean up operations, called ‘Liquidators’ are reported to have several health problems. (This is still to be confirmed by International Studies.) The nuclear energy programme world over could be seriously undermined with another incident of the magnitude anywhere near the Chernobyl accident and every effert, at the design, construction and operation stages, should be made to prevent recurrence of such accidents. The point that is being made is, notwithstanding the seriousness of the accident and worldwide concern it generated the studies by International Expert Bodies have shown that the radio-biological risk to the population due to the accident has been marginal.
REFERENCES
- League of Red Cross and Red Crescent Report on Assessment Mission to the Areas Affected by the Chernobyl Accident, LORCS, Geneva, 1990.
- The InternationalChernobyl Project. Technical Report—Report by an International Advisory Committee,
- World Health Organisation.Report on the WHO Mission of Radiation Protection Experts to the USSR, 19—25 June 1989, WHO, Geneva
7. Investigation on Dose Assessment and Cancer Mortality in High Background Radiation Areas of Yangjiang, China
Wei Luxin, Zha Yongru, Tao Zufan, He Weihui, Chen Deqing and Yuan Yongling
High Background Radiation Research Group, Deshengmenwai, Beijing, China
Ionizing radiation is capable of adversely affecting the health of population exposed to high doses. Risk estimates of low dose and dose rate radiation have very often been extrapolated on the basis of data accrued from intermediate and high dose radiation. However, it is evident from literature that consequences of low dose and dose rate radiation are significantly different in that, the biological consequences following low doses are less detrimental. The controversy about the exact impact of low dose radiation has continued due to a very sparse population based human data for evaluation. High background areas of Yangjiang in China, where more than 7 generations of people have lived is an ideal model to evaluate the effects of radiation on man. Our group (High Background Radiation Research Group) started a health survey in these high background radiation areas (HBRA) and, neighbouring control areas (CA) to estimate the detrimental effects on health, if any.
A total of 540 sq km of HBRA from two neighbouring areas, as shown in Fig. 1, were included for the survey. Control areas with normal background radiation but not far from HBRA at same altitude were also included for the survey.
Particles of monazite are being perpetually washed down from surrounding rocky mountains rich in granite. A uniform sediment of monazite thus has been accumulating over the years in Yangjiang and, surrounding basin, leading to high background radiation. The area which was a wasteland for centuries started being inhabited 800 years ago by migrants from Jiangxi province.
Control areas with normal background radiation at comparable altitudes were chosen. Control areas are about 10 km apart from HBRA. Control areas were also chosen such that all confounding factors except high background radiation were similar.
Aims of the study were elucidated at the outset which are:
- Toidentify sources of ionising
- Toestimate radiation levels and their
tt 1” 30’ HE’D‘ JJ t‘3 ’
Fig. 1. Locations of the investigated areas.
)13‘ OO
- Assessmentof absorbed dose, accumulated dose in population of different age groups and, effective dose
- Comparisonof confounding factors between HBRA and CA- environmental, socio-economic and population
- Studyof cancer
- Toundertake the survey of demographic
- Prevalenceof disease due to genetic
- Frequencyof chromosomal abberation, comprehensive analysis and explanation of the accrued
Sources of radiation and distribution is shown in Table 1. It is evident that concentrations of natural radionuclides in soil of HBRA and CA are strikingly different. Thorium for example is 5.2 to 7.6 times more in HBRA
than in CA. Environmental radiation due to Rn2°2 and Rn°20 was higher in
HBRA. Thorium isotopes in human lung tissues, Ra226 and Ra22′ in human teeth and bone were also highcr in HBRA by a factor of 3 to 8. Distribution
of radon and decay products was similar in both areas [3].
Annual individual exposure to gamma radiation from 1975 ro 1982 is shown in Table 2. In faci effective dose equivalcnt of individual absorbed dose after accounting for cosmic and terrestrial radiation is in the range of 6 msv per year in HBRA while it is 2 msv in CA. A considerably higher absorbed dose in HBRA may partly be due to higher RBE attributed to alpha particles deposited in lungs. Nevertheless, it can be concluded that inhabitants of HBRA receive a dose of radiation higher than that by most of the radiation workers.
Dose Assessment and Cancer Mortality in HBRA 71
Table 1. Concentration ot natural radionuclides in soil based on field gamma spectrometry or sample analyses
Method of analysis
Uranium (10“ g/g)
Thorium (10“ g/g)
Radium (10“ g/g)
HBRA
| Field | NaI (TI) | 7.1 A 2.3 | 36.3 + 12.7 |
|
| Sample | NaI (TI) | 8.9 -£ 3.3 | 57.5 -h 25.1 | |
| Sample | Ge (Li) | 9.6 + 2.8 | 55.3 + 20.7 | 3.7+ L2 |
| Sample | Radiochemistry | 7.6 A 1.7 | 60.4 + 28.0 | 3.9 + 1.5 |
CA
| Field | NaI (TI) | 2.0 + 0.6 | 6.6 A | 2.6 | |
| Sample | NaI (TI) | 2.2 + 0.7 | 8.8 + | 3.3 | 0.78 0.24 |
| Sample | Ge (Li) | 2.4 + 0.9 | 8.9 + | 4.l | 8.8 + 0.3 |
| Sample | Radiochemistry | 1.7 + 0.7 | 7.9 + | 3.1 | 0.8 + 0.3 |
| Ratio | |||||
| Field | Nal (T1) | 3.6 | 5.5 | ||
| Sample | Nal (TI) | 4.0 | 6.6 | 4.23 | |
| Sample | Ge (Li) | 4.0 | 5.2 | 4.90 | |
| Sample | Radiochemistry | 4.S | 7.6 | 4.95 | |
Table 2. Annual individual gamma exposures (mR/yr) in HDRA and CA
based on results of various measurements
| Device or dosimeter | Annual gamm
HBRA |
a exposure
CA |
HBRAJCA |
Year of measurement |
| RSS-111* | 337.1 -£ 60.6 | 115.7 + 21.2 | 2.9 | 1982 |
| TLD | ||||
| CaSO (Tm) | 351.4 -b 56.8 | 120.3ñ 27.2 | 2.9 | 1978 |
| LiF (Mg. Ti)-M | 330.0 -£ 55.1 | 103.0 + 23.6 | 3.2 | 1978 |
| CaSO (Dy) | 330.0 -£ 52.8 | 119.0 + 29.4 | 2.8 | 1975 |
| RPL | 330.0 + 35.6 | 119.4 + 24.6 | 2.8 | 1975 |
*Results produced by Field Gamma-ray Spectrometry were similar to thosc obtained
with RSS-111 device.
An accumulated dose of 150 mGy (87—129 mGy) and an effective dose equivalent of 274 msv (range 248—369 msv) was documented in a group of peasants above 50 years. Accumulated dose is a function of age.
A substantial difference in environmental and accumulated radiation between HBRA and CA is enough for risk estimation due to high background
radiation.
Health Effects of Radiation—A survey of demographic data
The survey of demographic data which was started in 1975 laid down certain
criteria for both HBRA and CA [1]. Population of Han nationality who have been residents for at least more than 40 years in both HBRA and CA were studied. Migrants and their children from overseas, as also those who had moved into CA from high background areas or from areas of undetermined radiation levels were excluded from survey. Children of closely related parents or grand parents or those of foreign parents were also not included in this study. Radiation workers for obvious reasons were not taken as candidates for survey.
A survey for confounding factors known to influence the incidence of cancer and hereditary disease have been reported earlier. However, for this study 459 cases to control matched pairs are subjected for analysis. Primary occupation in both groups is farming. HBRA had 94.1% peasants while CA had 93.2’7 of 459 cases to matched controls. Table 3 shows confounding factors for both HBRA and CA. There is no significant difference in the listed factors.
Table 3. Comparison of factors known to affect diseases cnused by mutation process
Factors
Constituent ratio (‘No) HBRA CA p**
Odds ratio*
Matched Unmatched
analysis analysis
| Pesticide use Occupations | 62.3 | 62.3 | NS | v00 | 0.95 (0.72-1.24) |
| involving the use of poisonous | 1.5 | 2.0 | NS | 0.60 (0.15-2.47) | 0.77 (0.29-2.09) |
| and noxious substances | |||||
| Smoking | 37.9 | 37.6 | NS | 1.06 (0.71-1.58) | 1.01 (0.88-1.16) |
| Alcohol | 37.2 | 38.6 | NS | 0.85 (0.60-1.21) | 0.90 (0.64-1.27) |
| consumption Medical X-ray | 20.0 | 26.4 | NS | 0.62 (0.45-0.87) | 0.70 (0.51-0.95) |
| exposure |
*Figures in parentheses represent 95′ o confidence intervals.
**NS means no significance.
The end points of this survey are cancer mortality, incidence of leukaemia, thyroid nodularity and Down’s syndrome. Results of each will be discussed in the following pages.
Cancer Mortality
Cancer mortality was initially studied by calling out the data from a retrospective survey. However, since 1979 a cancer registry was set up with the participation of regional hospitals, county hospitals, health professionals and administrators. Patients diagnosed to have cancer—those who die of cancer are entered in cancer registry. An expert task force which meets twice a year has been reviewing the diagnosis and all the data collected in
Table 4. Site specific cancer mortality (per 10’ pyr) in HBRA and CA (1976-1986) for males*
| HBRA | CA | |||||||
| Site of | No. of | Mortality | No. of | Mortality | ||||
| cancer | deaths Avg. | Adjusted** | deaihs | Avg. Adjusted** value | ||||
| Nasopharynx | 59 | 11. l l | 11.58 | 72 | 14.27 | 13.79 | > 0.05 | |
| Esophagus | 10 | 1.88 | 1.97 | 7 | 1.39 | 1.32 | > 0.05 | |
| Stomach | 36 | 6.78 | 7.11 | 29 | 5.75 | 5.49 | > 0.05 | |
| Liver | 85 | 16.01 | 16.67 | 113 | 22.40 | 21.62 | > 0.05 | |
| Intestine | 10 | 1.88 | 1.96 | 14 | 2.78 | 2.69 | > 0.05 | |
| Lung | 17 | 3.20 | 3.36 | 18 | 3.57 | 3.39 | > 0.05 | |
| Breast | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Leukemia | 17 | 3.20 | 3.21 | 18 | 3.57 | 3.70 | > 0.05 | |
| Osteosarcoma | 4 | 0.75 | 0.78 | 4 | 0.79 | 0.77 | > 0.05 | |
| Other | 61 | 11.49 | 11.91 | 55 | 10.90 | 10.48 | > 0.05 | |
| Total | 299 | 56.31 | 58.55 | 330 | 65.42 | 63.25 | > 0.05 | |
*530,952 pyr observed in HBRA; 504,458 pyr in CA.
**Adjusted with the combined population of HBRA and CA.
74 LUXIN ET AL
cancer registry. Deaths due to other causes are also registered and analysed. At the end of 1956, 467 people had died of cancer out of 1008, 769 person years at risk in HBRA, with a crude mortality of 46.3, per 100,000 pyr. Table 4 shows the details of the break up according to site. As is evident from the table there is no statistical difference in the crude mortality rate due to cancer between HBRA and CA though deaths due to cancer in absolute number was higher in CA.
Thyroid Nodularity
A collaborative study with National Cancer Institute of USA was conducted to study thyroid disorders in the female population of both HBRA and CA. Thyroid nodularity is a harbinger of neoplasms of thyroid. Women aged from 50 to 65 (N — 1001 in HBRA and 1005 in CA) were evaluated as the accumulated radiation which increases with age would become significant by this age. Physicians specialised in thyroid diseases conducted interviews and physical examination of cohorts mentioned above. Randomly chosen women were evaluated for thyroid hormone levels, urinary iodine and chromosomal anomalies. Prevalence of nodular thyroid was 9.5’7o in HBRA as compared to 9.3% in CA. No appreciable difference between these two groups was rioted. It may be surmised that exposure to continuous low level radiations is unlikely to enhance the risk of thyroid diseases.
Down’s Syndrome
Initially children of 12 years age during 1975–1979 were evaluated. A pre valence rate of hereditary and congenital defects of 22.64 cases/1000 people in HBRA (N — 13,425) and 22.54 per 1000 in CA (N — 13,087) was recorded which is similar in both areas. However, pievalence of Down’s syndrome was more in HBRA than CA as shown in Table S. In 1985 the data was enlarged to verify the accuracy of this observation. The results are mentioned in Table 6. Incidence of Down’s syndrome in HBRA is more than in CA.
Table S. Prevalence of Down’s syndrome in HDRA and CA
| Year of examination |
No. of examinee |
HBRA
No. of cases |
Rate (10‘’) |
CA
No. of No. of Rate examinee cases (10 ‘) |
| 1975 | 3,504 | 7 | 2.00 | 3,170 0 0 |
| 1979 | 9,921 | 7 | 0.71 | 9,917 4 0.41 |
| 1985 | 25,258 | 22 | 0.87 | 21,837 4 0.18 |
| However, | there is a | wide | range of | spontaneous incidence of Down’s |
syndrome, 1–2 per 1000 newboms (UNSECAR 1982) [2]. Hence, a comparison of incidence was made with other provinces like Guandong province. No statistically significant difference was noted. Down’s syndrome
Dose Assessment and Cancer Mortality in HBRA 75
Table 6. Significance test
| Area of examination | Cases of Down’s syndrome | No. of examinee | Rate (10*’) |
| HBRA (1) | 22 | 25,258 | 0.87 |
| CA (2) | 4 | 21,837 | 0.18 |
| Zhanjiang and Foshan (3) | 16 | 26,504 | 0.60 |
of Guandong Province
(1) and (2) p < 0.01; (2) and (3) p = 0.01; (1) and (3) p > 0.05.
is also related to age at maternity. A study of the same in both areas showed that women who were parents at 35 years were more in HBRA. This also could be the reason for higher incidence of Down’s syndrome. But, as emphasised earlier the incidence has a large fluctuation and the incidence of Down’s syndrome in HBRA is comparable to other provinces.
It is unlikely that radiation has had a causative effect.
Summary
The High Background Radiation Research Group (HBRRG) began its epidemiological investigation in 1972 in the high background radiation areas of Yangjiang, Guandong Province near South China Sea. The purpose of this investigation is attempting to provide some information for evaluating whether any detrimental effect exists in a large population (about 80,000 people) whsse families have been continuously exposed to a low dose rate of ionizing radiation for many generations. After identifying the sources of the elevated natural ionizing radiation, measurements of gamma radiation levels indoor and outdoor by means of various devices and dosimeters and measurements of radionuclides in food, drinking water and in human tissues, the annual effective doses were estimated for the inhabitants in the high background radiation areas (HBRA, 6.4 msv in average) and for the inhabitants in the nearby control areas (CA, 2.4 msv in average). The cohort members were classified into four subgroups according to the annual dose rate of gamma exposure. Demographic survey was conducted in HBRA and CA for comparison of the age, sex distribution of the populations between HBRA ahd CA. Social, economic and environmental factors which might influence the cancer mortality were investigated, which showed that these factors were almost comparable in HBRA and CA. 1,008,769 person-years in HBRA, and 995,070 person-years in CA were observed for cancer mortality, with the mortality rate (adjusted by the combined population of HBRA and CA) of 48.81 per 100,000 pyr for all cancer in HBRA, 51.09 per 100,000 pyr in CA respectively.
The mortality rate of total cancers was higher in CA than that in HBRA
for male, female and both sexes. However, the difference was not large enough to be significant statistically. Age truncated analysis (40—70 years old) of mortality for all cancers except leukemia showed that the rate was
lower in HBRA than that in CA. Analyses of the data concerning the mortality rates of leukemia and the prevalence of thyroid nodularity revealed that there was no difference statistically of these rates between HBRA and CA. The excess relative risks estimated for different dose groups showed that the cancer mortalities were not dose-related in the range of this investigation.
This text has been prepared by Dr. Nagraj G. Huilgol on the basis of a lecture delivered by Prof. Wei Luxin and also on the basis of a synopsis kindly supplied by him.
REFERENCES
- Luxin, W. et al. Report of third stage(1982—1984) health survey in high background radiation area in Yangjiang, China. Chin. J. Radiol. Med. Prot. 1985; 5: 82.
- United Nations Scientific Committee On the Effects of Atomic Radiation. Reportto the General Annex I: Genetic effects of radiation. United Nations, New York.
- Zhang, Z.Annual effective dose equivalents arising from inhalation of Rn2^, Rn°“ and their decay products in high background radiation area in Chin. J. Radiol. Med. Prot. 1985; 5: 102.
8. Cancer Risk of the Indian Population from Low Level Radiation, Projected from the
Revised ICRP Risk Coefficients
Y.S. Mayya, P.V. Joshi and K.S.V. Nambi
Environmental Assessment Division Bhabha Atomic Research Centre, Bombay, India
Introduction
The projection of radiogenic cancer risks in exposed population groups of either occupational workers or of affected members of the public, constitutes an important aspect of their risk assessment. For acute exposures, the lifetime coefficient of expressed risk, referred to simply as the ‘risk coefficient’ provides an important measure of the index of harm in a given population. For chronic exposures, the 1990 report [l] of the International Commission on Radiological Protection, lists several additional risk attributes’ as the indices of harm. Unlike the primary risk coefficients, which are held universal while transferring risks across populations, the coefficient and the attributes of expressed risks depend strongly on the demographic data of the population in question. ICRP-d0 gives the values of the risk coefficients for acute exposure for several populations such as those of the US, Japan, UK, China and Puerto Rico, while, its new system of dose limitations for chronic exposures is based on the risk attributes computed using the cancer death rates of the Japanese, and the life table of the Swedish, populations. In this note, we estimate these quantities for the Indian population using the best available Indian cancer mortality and demographic data. These would be useful in predicting the excess mortality rates expected in chronically exposed Indian populations and also would provide suitable national criteria for deciding on the appropriate system of dose limitations both to occupational workers and the affected members of the public.
It may be noted that a similar risk estimate for the Indian population was also attempted recently [6]. In that approach, the unconditional excess death probabilities were computed by multiplying ICRP-60 tables of the conditional excess death probabilities (which refer to the Japanese population) with the survival probabilities taken from the Indian lifetable. On the other hand, here we have used the available Indian data for generating both the conditional and unconditional death probabilities. Besides, we compute certain additional risk parameters not considered by him.
Material and Method
The exposure situations considered are identical to those assumed by ICRP-
60: age 0 over lifetime for members of the public and age 18—64 years for occupational workers. (Although the Indian occupational worker retires at age 60 yr, we have retained the retirement age as 64 yr in order to be able to readily compare our values with the ICRP values. Lowering the upper age limit from 64 to 60 yr reduces the risk estimates by about 5%.) For chronic exposures, an individual is assumed to be uniformly irradiated between the above intervals for the two groups. For acute exposures, these represent the intervals between which an individual is assumed to have received an acute dose. The risk coefficients are then obtained by averaging with respect to the lifetable of the population between the respective intervals. For chronic exposures, the risk attributes computed are: (i) age-specific conditional and unconditional excess death probability rates, (ii) attributable lifetime excess death probabilities, (iii) loss of life expectancy at birth and (iv) loss of lifetime if cancer death occurs. For the definition of these quantities, the reader is referred to ICRP-60 (1991).
Separate computations are made for each sex based on the generally
accepted constant Relative Risk (RR) model. The revised primary risk coefficients (ICRP-60, Table C-1) given separately for leukaemia and for all other cancers combined, provide the basic data for the RR model. These have been used along with a dose and dose rate reduction factor (DDREF) of 2 for low dose exposures. The other input data required are: (i) the age and sex specific base-line cancer mortality rates and (ii) the lifetable of the Indian population. These have been obtained from the following sources.
A nation-wide data on the age-specific base-line cancer mortality rates does not exist in India. Population based registries for recording cancer incidences have started operating in a few major urban centers of the country only recently. Regarding mortality, the population-basecl data for the past decade are available only for 3 urban centres of a single state, viz., Bombay, Nagpur and Pune, for the periods 1983-89, 1980-89 and 1975-80, respectively and were published by the Indian Cancer Society [2, 3, 4]. The averages of these data, taken separately for leukaemia and for other cancers combined, and for each sex, are assumed as the Indian base-line rates for males (Fig. 1) and for females (Fig. 2). In the absence of a true national base-line, the above assumption would lead to risk attributes that are only indicative of the Indian values for the purposes of comparing with those of other countries. It may be noted that the data are available in the standard 5-year intervals only upto the age of 74 years. The age group above 75 years forms a single interval and has been retained as such. For comparison, the power-law forms based on the Japanese base-line data, employed in ICRP-60 in its Annexe C, have also been reproduced in Figs. 1 and 2. (There appears to be a printing error in the coefficient ‘a’ of the fitting function given in section (C47) of ICRP-60 for the non-leukaemia base-line rates. In order to reproduce the summary Tables C-2a, b, the factor 10°1 has to be replaced by 10*l° for both males and females.) It may be seen from Fig. 1 that above the age of 25 years, the base-line rates for Indian males are well below the corresponding ICRP values. On the other
0 25
50
AGE (Y)
75 100
Fig. 1. Base-links cancer mortality rate in males (Indian curves are based on data available on 3 cities only).
hand, in Indian females, the base-line rates for non-leukaemia are higher in the age group 10—60 years than the corresponding ICRP values and are lower above the age 65 years.
The data on the Indian lifetables based on the decennial censuses have been provided by the reports of the Indian Statistical Institute [5l. The lifetable for the period 1991—2001, which is most relevant for the present purpose, is provided by the Institute based on a projection from the earlier censuses. The expectation of life at birth in India has been rapidly rising over past several decades and is projected to be at 62.61 years for males and
63.15 yean for females during 1991—2001. It is still much lower than the respective approximate values of 72.46 and 78.58 years computed by us from the death rates given in ICRP—60 for the Swedish population.
The computations have been made using the lifetable for the period 1991—2001 following the procedure identical to that used in ICRP-60: same latent and cut-off periods have been used (leukaemia: 2 years latent period since the beginning of exposure and lasting for 40 years; for all other cancers, 10 years latent period lasting over lifetime). The expressed death probabilities were assessed at 18 years as in ICRP-60. The computer program developed has been tested by reproducing the summary Tables C-2a, b and
80 YYA ET AL
1000
10
0.5
25 50
AGE (Y)
75 100
Fig. 2. Base-line cancer mortality rate in females (Indian curves are based on data available on 3 eities only).
C-3a, b for the conditional and the unconditional death probabilities in Annex-C. (However, the numbers listed in Table C-.5 of ICRP-60 appear to be erroneous as they do not follow from Tables C-2a, b which they should.)
Results and Discussions
Figures 3 and 4 show the variations in the death probability rates with age for males and females respectively in the case of Indian public chronically exposed at a rate of 1 mSv/y from birth over lifetime. The corresponding quantities taken from the ICRP-60 report are also shown in the figures. The exposure category of the occupational workers also shows similar variations in the death probability rates and therefore is not shown. Essentially, the conditional as well as the unconditional excess death probability rates are lower at all ages in the Indian males than the corresponding ICRP values. In contrast, in females, Indian death probability rates are higher in the middle age group and fall below the corresponding ICRP values at higher ages. The unconditional death probabilities begin to differ from the conditional probabilities at an earlier age in the Indian case. These differences are a reflection of the differences in the base-line and lifetime patterns between
10*y
P: ICRP-60
– – – P: INDIAN
– O: ICRP-60
- •••••••••-Q:INDIAN
‘o 1o2
1.0 I
25 50 75
AGE (Y)
100
Fig. 3. Conditional (P) and unconditional (Q) (assured at age 18 yr) excess death probabilities for public (males: exposure rate = 1 mSv/y).
these population groups. The maximum values of the expressed risk (the peaks in the unconditional death probabilities), are lower by about a factor of 2 than the corresponding ICRP values.
Table l compares the values of the sex averaged risk coefficient for acute exposure obtained for the case of Indian public, with those given in ICRP-60 for the populations of Japan, USA, Peurto Rico, UK and China. The world average assumed by ICRP-60 is also given in the table. The Indian value (3.0% per Sv), which is less than 50% of that for UK and is 60% of the ICRP-60 average, is the lowest. It is closer to the value indicated for the Chinese population. Similarly, Table 2 gives a comparison
Table 1. Comparison of the Indian risk coefficient (10‘2 Sv°’) with that for other populations, for acute exposures
| India | Japan | USA | Peurto Rico | UK | China ICRP-60 average |
| 3.0 | 5.3 | 5.6 | 4.7 | 6.4 | 3.1 5.0 |
0 25 50
AGE (Y)
75 100
Fig. 4. Conditional (P) and unconditional (Q) (assessed at age l8yr) excess death probabilities for public (females: exposure rate = lmsv y ‘).
Table 2. Comparison of Indian rish attributes with ICRP-60 values, for chronic exposures at the risk of 1 msv per year
Exposed
group
Risk attribute
ICRP-60
Indian
| Public | Attributable lifetime probability of cancer | 0.4 | 0.22 |
| Worker | death (‘No)(Assessed at age 18 yr)
Attributable lifetime probability of cancer |
0.18 | 0.10 |
| death (‘No)(Assessed at age 18 yr) | |||
| Public | Annual extra probability of cancer death at age 70 yr (10“) | 135 | 69 |
| Worker | Annual extra probability of cancer death at age 70 yr (10“) | 65 | 34 |
| Public | Life expectancy lost at age 0 (yr) | 0.05 | 0.028 |
| Worker | Life expectancy lost at age 18 (yr) | 0.023 | 0.015 |
| Public | Life of lifetime if cancer death (yr) | 13.4 | 12.8 |
| Worker | Life of lifetime if cancer death (yr) | 12.6 | 11.5 |
20 30 40 50 60 70 80 90 100
LIFE EXPECTATION AGE (Y)
Fig. S. Variation of attributable death probability with life-expectation
of the Indian cohort during 1951—2001.
of the Indian and the ICRP-60 values of other risk attributes (sex averaged) for chronic exposures of both the public and the worker categories. These are about 50—609» of those indicated in ICRP-60. barring the case of ‘loss of lifetime if cancer death occurred’. The loss of lifetime is only marginally reduced to about 12.8 yr for public and 11.5 yr for worker
in the Indian case from the respective ICRP-60 values of 13.4 yr and
12.6 yr. This quantity, which, by its de*fin‘it’ion, is independent of the exposure
rate, appears to be somewhat robust against the lifetable of the study population.
It is interesting to examine the sensitivity of the risk attributes to the changes in the lifetable of the Indian population over the past decades. We do this by studying the variation of the ‘lifetime probability of attributable death’ with respect to the increasing expectation of life at birth over 5 decades upto year 2001 (Fig. 5). As may be seen, the increase in risk is almost linear in both the public and the worker groups of both the sexes; a linear regression analysis shows that the slopes in the case of public are about twice those for the workers. A linear extrapolation indicates that the female risks approach the ICRP-60 values while the male risks continue to remain lower even when the life expectations at birth match. This will be so provided that the base-line cancer risks of the Indian population will remain essentially unchanged in future.
Conclusion
This analysi5 indicates that the adoption of the ICRP-60 dose limits in India entails an extra safety margin by at least a factor of two. Similar conclusion was also arrived at in the study by Mehta (Mehta, 1992). The Indian demographic data point at a steady lengthening of the life-span; in which case, the safety margin will reduce in future years providing the ICRP dose limits will not be changed.
Acknowledgement
We thank Dr. S.K. Mehta for helpful discussion.
REFERENCES
ICRP-60 ( 1991): Recommendations of the International Commission on Radiological Protection. Ed. Oxford Pergamon Press 1990.
- Jussawala,J., Yeole, B.B. and Natekar, M.V. (t986, 1989, 1990 and 1991)
Cancer morbidity and mortality in Greater Bombay 1983, 1985, 1986—1987,
1988 and 1989. Report of the Indian Cancer Society, Bombay, 1984.
- Jussawala,, Deshmukh, N.K., Gandhe, M.L., Yeole, B.B. and Natekar,
M.V. Cancer morbidity and mortality in Nagpur city agglomeration, t980—1984 and 1985-1989. Reports of the Indian Cancer Society, Bombay, 1987, 1991.
- Jassawala, D.J., Shah U.,Yeole, B.B., Natekar, M.V. and Rajagopalan, R. Cancer morbidity and mortality in Poona city agglomeration, 1975—1977 and 1978-1980.
- Malaker,R. and Guha, Roy S. Reconstruction of the Indian lifetables for
1901—81 and projections for 1981—2001. Indian Statistical Institute, Calcutta
1986.
- Mehta, S.K.‘Assessment of the New ICRP dose Limits in the Indian context’, Radiation Protection, 1992; 15: 6—7.
9. Study of Population Exposed to Natural Background Radiation in Kerala, India
- KrishnanNair, N. Sreedevi Amma, P. Gangadharan,
- Sankaranarayanan,T.P. Ramachandran,
- Jayalekshmyand K.S. Mani
Regional Cancer Centre, Trivandrum, India
Man has lived with one or other forms of radiation and the health effects of ionising radiation in human beings continues to be an active research topic. Exposure to large doses of ionising radiation is known to increase the incidence of many forms of cancer and cause chromosomal aberrations [13]. However, such large doses are mostly used in therapy where benefit is the main concern. Patients treated with radiation for ankylosing spondylitis [3], tinea capitis [10], thymus enlargement f6l and cancer of various sites are observed to be at higher risk of developing cancer [2]. Although a higher risk among employees of nuclear power plant and those who are exposed to weapon testing etc. [4) is yet to be universally demonstrated, the high incidence of bone cancer observed among radium dial painters [11] is recognised as due to exposure. Life span studies of atom bomb exposed population have shown an increased incidence of cancer of various types, beginning with leukaemia incidence 5 to 7 years after bombing [1, 12].
Conclusive evidence of carcinogenesis due to low level radiation is yet to come [9, 14]. The difficulties in obtaining such evidence include estimation of absorbed doses for various body organs, interplay of confounding factors, lack of large population groups exposed for sufficiently long period of time, migration of population exposed and the long periods of surveillance needed for observing any statistical excess of cases.
Of the very few areas of the world having high natural background radiation, the Kerala coast in South-Western India provides a unique opportunity to study the effects of such radiation in human beings. In the district of Quilon, the coastal areas have high natural radioactive sands. High concentrations of monazite, definable geographical land marks, a high density of population, which was more or less stable are the special features of the area. The monazite sand deposited in the beach area contains ilmenite, rutile, zircon and other rare earths. It is known that thorium and its radioactive decay products are the main sources for background radiation. The people living in the Neendakara-Chavara coast in Karanagappally Taluk in this district have the maximum exposure to natural background radiation in the coast. There is no other place with such high density population living in such a high level natural background radiation area.
This population exposed to natural radiation has been the subject of investigation in the past. A population dosimetry as well as demographic data was done in 1970 [5]. It was observed in this study that the monazite deposits were not homogeneous. There were patches of areas with normal background radiation amidst areas of high background radiation. When a 100 mR/year was considered as normal background radiation it was observed that about 1.ldc of households received more than 20 times normal levels,
8.89c received 10 times the normal and 24.6% received 5 times the normal level. On the basis of the sample survey it was stated that per capita dose for the entire population resident in the area was around 397 mR/yr [5). There has been no statistically significant difference observed between the population groups receiving different doses of radiation in fertility index, sex ratio, abortion, still birth, infant mortality and congenital abnormalities. However, another study [8] presented data to demonstrate a higher prevalence of Downs Syndrome and certain forms of mental retardation. These initial studies in the area have been observed to have statistical drawbacks, besides poor recognition of confounding factors like tobacco use, age at marriage and pregnancy etc.
We have initiated a study (since September 1990) in the area to address the following questions:
- Whatis the cancer incidence in the area?
- Isit different from other population groups?
- Whatis the pattern of cancer in the area?
- Isit different from other areas?
- Arethe incidence and pattern related to background radiation and is it dose related?
The Study Area and the People
Karunagappally Taluk which borders Quilon Taluk.is about 80 km north of Trivandrum. The taluk spreads over an area of 212 sq kms with a coastline in the west of about 25 km long. The farthest eastern point from the coast is about 20 km. The estimated population of the district, according to 1981 census was approximately 5,00,000; however the 1991 census figures indicate that the population is around 4,00,000. This was primarily due to an overall reduction in growth rate in the state as well as deletion of some remote areas from the taluk. Agriculture, coir making, cashew nut industry and fishing are the main occupation of the people. The Indian Rare Earth Factory which processes the rare earth and the Kerala metals and minerals factory are the only two major industries in the area. The area is largely rural. There is no dedicated cancer centre in the area; however modem medical facilities are available through the governmental and private medical centres. A high literacy rate (above 75%) and a sex ratio favouring females are special features of Kerala population. An expectation of life around 70 years is also observed in the State which is almost 15 years more than the Indian average. Maintenance of a cancer registry demands a mechanism to identify and
record all cancer cases arising in the population as well as the necessary data on the base population. In the area under study, the medical records of the hospitals are far below the required standard for registry operations and the death registration system maintained by the governmental bodies also does not provide the data required for such medical research. Thus the standard methods of cancer registry operations are not feasible in the area. The study also required accurate base information on the socio-demographic characteristics. of the population, including the facilities of housing, life style factors and disease episodes in the family. Such information was not readily available from any existing documents. There are many life style factors like tobacco habits which are associated with the occurrence of cancer. It was felt ih the beginning that any attempt to correlate radiation exposure to health effects should recognise the confounding factors.
Taking into consideration all the above mentioned factors, it was decided that a total survey of the population enrolling every individual and a surveillance follow-up would be necessary. Such a study is now proceeding in the area. Every individual living in the area is enumerated, the radiation levels inside and outside the house are measured. Soil samples collected from each square kilometer grid in the area are analyzed for their thorium and other radioactive contents. TLD measurements are also undertaken in selected houses.
Individual enumeration of above 60,000 individuals have been undertaken,
so far. Apart from health related medical information, habits of smoking, chewing, alcohol consumption are recorded along with years of stay in the locality, occupation etc.
Field clinics are held in the rural area where individuals suspected to have cancer (on the basis of enumerators proforma returns) are examined to rule out prevalent cancer cases. A constant surveillance of the population is mounted through hospital visits, hospital records, contacts with village leaders and scrutiny of the records of the cancer centre at Trivandrum. The reported
Table 1. Tobacco-alcohol habituees Chavara Panchayat: 1942 males
Age
Below 15
15-19
20—24
25—29
30—34
35—39
40-44
45—49
5H-54
55-59
65—69
to with habit
0.6
10.0
45.5
71.6
75.2
87.3
93.1
87.1
91.4
92.6
84.3
deaths in the Panchayats (local administrative bodies) are also enquired into. With the excellent cooperation received from the villagers and officials, the methodology seems to be working well. Base line data collected so far indicate a high prevalence, of tobacco habit in the population. Among males above the age of 15 years, 58% were tobacco smokers, 38% were tobacco chewers and 46% were addicted to alcohol. Almost 90’7o of the male population above the age of 40 was tobacco alcohol habituees (Tables 1 and 2).
Table 2. Tobacco habituees Chavara Panchayat : 2106 females
Age No with habit
| 20—24 | 1.2 |
| 25—29 | 3.2 |
| 30—34 | 6.8 |
| 35—39 | 13.5 |
| 40—44 | 20.3 |
| 45—49 | 36.4 |
| SP-54 | 36.6 |
| 55—59 | 31.8 |
| 66—64 | 53.5 |
| 65—69 | 55.0 |
Even though the survey was started only in 1990, the data on cancer patients who attended the Regional Cancer Centre from Karunagappally since 1982 was analysed to study the distnbution of major forms of cancers. Comparison with the total hospital cases is also done in Tables 3 and 4.
The hospital attendance of cancer cases are influenced by several patient selection factors and comparisons between series should be cautiously attempted. Moreover, the hospital series provides only relative frequencies of various cancers and thus are highly unstable. Only the incidence rates, which are the absolute rates, should be used to study the patterns. Such data will be forthcoming from the current approaches. It will be erroneous to conclude, on the basis of such data, that certain forms of cancers are predominant in the radiation bclt area.
However, from an indepth analysis of patient records of Regional Cancer Centre for the period 1988—1991 and considering the cancer deaths recorded in the Panchayat records, an estimate of minimum average annual incidence rates was made. These are presented in Table S. It is felt that the true incidence will not differ by more than 5 to 10Vo of this estimated value. Further improvement of this estimate is possible only when all sources of cancer diagnosis in the study areas were tapped and information obtained. Suffice to comment that this estimate is at par with the reported urban rates. In fact the life style in rural Kerala is far different from the life style of rural people elsewhere in the country and is generally comparable with semi- urban life style.
Table 3. Distribution of cancer cases from Karunagapally Taluk 1982-1990 attending the Regional Cancer Centre, Trivandrum
Site
Number of cases and percent
Male Female
| No. | (to) | No. | (A) | ||
| Oral cavity, pharynx | 234 | 34.6 | 81 | 13.8 | |
| Oesophagus | 45 | 6.6 | 18 | 3.1 | |
| Stomach | 32 | 4.7 | 9 | 1.5 | |
| Colo-rectum | 12 | 1.8 | 8 | 1.4 | |
| Liver | 20 | 3.0 | 3 | 0.5 | |
| Other digest
Nose, sinuses |
6
13 |
0.9
1.9 |
7
2 |
1.2
0.3 |
|
| Larynx | 35 | 5.2 | 2 | 0.3 | |
| Lung | 100 | 14.8 | 10 | 1.7 | |
| Bone, conn. tissue, skin | 21 | 3.1 | 23 | 3.9 | |
| Breast | 107 | 18.3 | |||
| Cervix | 177 | 30.2 | |||
| Uterus | 10 | 1.7 | |||
| o*aW | 30 | 5.1 | |||
| Vulva, vagina | 9 | 1.5 | |||
| Prostate | 4 | 0.6 | |||
| Testis | 7 | 1.0 | |||
| Penis | 9 | 1.3 | |||
| Bladder | 9 | 1.3 | |||
| Kidney | 6 | 0.9 | 4 | 0.6 | |
| Eye | 2 | 0.3 | 1 | 0.2 | |
| Brain | 18 | 2.7 | 8 | 1.4 | |
| Thyroid | 12 | 1.8 | 18 | 3.1 | |
| Leukaemia, lymphoma | 54 | 8.4 | 28 | 4.8 | |
| All other and unknown | |||||
| primary | 38 | 5.6 | 28 | 4.8 | |
| 677 | 586 | ||||
The geographical area under study is unique because of high density of population. Further, in this same geographical area, there are places with high background radiation levels as well as normal levels and this provides opportunity for comparison. Near the sea coast there is a strip of land where the highest levels are observed. This strip which is separated from the mainland by backwaters is not more than 1 to 2 kms broad and about 18 kilometers long. The population in this strip is mainly engaged in fishing and the population comprises about 1,00,000.
There have not been many opportunities to study the health effects of natural background radiation in human populations. The reports of two smdies conducted in China where such high background radiation was present, did not demonstrate any significant excess of cancer risk or of Thyroid nodules (7, 15].
Table 4. Distribution of cancer cases (Regional Cancer Centre, Trivandrum, 1990)
Male
No.
Female
No.
| Oral cavity, pharynx | 23871 | 31.2 | 358 | 14.5 |
| Oesophagus | 136 | 4.9 | 52 | 2.1 |
| Stomach | 87 | 3.1 | 35 | 1.4 |
| Colo-rectum | 64 | 2.4 | 67 | 2.7 |
| Liver | 84 | 3.0 | 21 | 0.9 |
| Other digestive | 43 | 1.5 | 18 | 0.7 |
| Nasal cavity, sinus | 33 | 1.2 | 15 | 0.6 |
| Larynx | 119 | 4.3 | 4 | 0.2 |
| Lung | 346 | 12.4 | 42 | 1.8 |
| Bone, conn. tissue, skin | 130 | 4.6 | 90 | 3.6 |
| Breast | — | 597 | 24.2 | |
| Cervix | — | 492 | 20.0 | |
| Uterus | — | 32 | 1.3 | |
| Ovary | 129 | 5.2 | ||
| Vulva, vagina | — | — | 42 | 1.8 |
| Prostate | 58 | 2.1 | — | |
| Testis | 27 | 1.0 | ||
| Penis | 28 | 1.0 | — | — |
| Bladder | 53 | 1.9 | 6 | 0.2 |
| Kidney | 20 | 0.7 | 13 | 0.5 |
| Eye | 6 | 0.2 | 10 | 0.4 |
| Brain | 124 | 4.5 | 83 | 3.4 |
| Thyroid | 55 | 2.0 | 114 | 4.6 |
| Leukaemia, lymphoma | 320 | 11.5 | 148 | 6.0 |
| All other and unknown primary | 183 | 3.6 | 94 | 3.8 |
| Total Cases | 677 | 586 | ||
It may be appropriate here to mention the difficulties in arriving at definite conclusions on such low level exposure effects. These have been effectively mentioned by Modan “Unfortunately, the interpretation of the direct LDR findings is confounded by inadequate dosimetry, small sample sizes, lack of adequate controls, simultaneous exposure to extraneous carcinogenic factors and possibly erroneous measurement as well” [9). Our group which is making an enormous effort to evaluate consequence to health due to low levels radiation will perhaps, over the years elucidate the problem in a better perspective.
Population Exposed to Natural Background Radiation iii Kerala 91
Table S. Annual incidence rates of cancer per 1,00,000 population CR—Crude Rate : Age Standardised : To World Population
Place
Crude
Incidence rates
Age standardised
| M | F | M | F | |||
| Karunagappally Taluk** 1988—1991 | d4.9 | 48.0 | 102.8 | 73.5 | ||
| Barshi-Rural registry | ||||||
| Maharashtra | 1989 | 44.1 | 44.5 | 57.6 | 52.2 | |
| Bombay* | 1989 | 70.4 | 73.4 | 130.4 | 120.4 | |
| Bangalore* | 1989 | 65.6 | 76.7 | 112.2 | 124.7 | |
| Madras* | 1989 | 78.7 | 95.9 | 118.5 | 135.0 | |
| Delhi* | 1989 | 70.9 | 88.6 | 118.8 | 140.7 | |
Source: *National Cancer Registry Programme, Biennial Repon 1988-1989, Indian Council of Medical Research, New Delhi 1992.
**Minimum average annual incidence rate per 1,00,000 for 1988-1991.
REFERENCES
- Beebe,W., Kato, H. and Land, C.E. Studies of mortality of A Bomb survivors. Mortality and Radiation dose 1950—1974. Rad. Res. 1978; 75: 138.
- Boice,D. Jr., Day, N.E., Andersen, A. et al. Cancer risk following radiotherapy of cervical cancer: a preliminary report in Eds. John D. Boice Jr. and Joseph F. Fraumeni J. Radiation Carcinogenesis 1984; 161. Raven press.
- CourtBrown, M. and Doll, R. Mortality from cancer and other causes after radiotherapy for ankylosing. Br. Med. J. 1965; 2: 1327.
- Darby,C., Kendal, G.M., Fell, T.P. et al. A summary of mortality and incidence of cancer in men from the United Kingdom who participated in the U.K. atmospheric Nuclear Weapon tests and experimental programmes. Br. Med. J. 1988; 297: 332.
- GopalAyengar, R., Sundaram, K., Mistry, K.B., Sunta, C.Pvt., Nambi, K.S.V., Kathuria, S.P., Basu, A.S and David, M. 1971. Evaluation of the long term effects of’High Background Radiation on selected population groups of the Kerala Coast. United Nations Conference on Peaceful uses of Atomic Energy 11, 31, U.N. Vienna 1972.
- Hempelmann, L.H., Hall, W.J., Philips, M. and Ames, W.R.Neoplasms in persons treated with X-rays in Fourth Survey in 20 years. J. Nat. Can. Inst. 1975; 55: 519.
- HighBackground Radiation Research Group, Healthy Survey in High Background Radiation Areas in Science 1980; 209; 877.
- KochuPillai, , Thangavelu, M. and Ramalingaswamy, V. Nodular lesions of the Thyroid in an area of high background radiation in coastal Kerala, India. Indian J. Med. Research 1976; 64: 537.
- Modan, B. Low Dose Radiation Carcinogenesis. J. Cancer 1992; 28A: 6/7 1010.
- Ron, and Modan, B. Benign and malignant thyroid neoplasms after childhood irradiation for Tinea Capitis. J. Nat. Cancer. Inst. 1980; 65: 7.
- Rowland,E and Lucas, H.F. Radium Dial Workers: in Eds. John D. Boice Jr. and Joseph F. Fraumeni Jr. Radiation Carcinogenesis 1984; 231. Raven Press, New York.
- Schull,I Atomic Bomb Survivors : Patterns of Cancer Risk in Eds. John D. Boice Jr. and Joseph F. Fraumeni Jr. Radiation Carcinogenesis 1984; 21: Raven Press, New York.
- UnitedNations Scientific Committee on the effects of Atomic Radiation. Sources, effects and risks of Ionising Radiation, New York,
- Upton, A.C. Carcinogenic Effects of Low Level Ionising Radiation. J. Nat. Cancer 1990; 82: 448.
- Wang, Z.,Boice, J.D., Boice, Jr., Luxin, Wei, Gilbert, W. Beebe, Yongru Zha, Kaplan, M.M., Zufan Tao, Maxon, H.R. III, Sowzhi Zhang, Schneider, A.B., Bingde Tan, Wesselar, R., Dequing Chen, Ershow, A.G., Kleinerman, R.A., Gayle, L., Littlefield and Dale Preston. Thyroid Nodularity and Chromosome Abberrations among women in areas of high background radiation in China.
- Nat.Can. Inst. 1990; 82, 478.
10. Effects of Low Dose X-ray Irradiation on Autologous Tumor Killing System
Atsushi Uchida, Yashitaka Kariya, Taga Takashi and Katsuji Sugie
Department of Late Effect Studies, Radiation Biology Center
Kyoto University, Kyoto, Japan
Introduction
In vitro cell-mediated cytotoxicity is considered to be one expression of host immune defense mechanisms against tumor. Most studies on cytotoxicity against tumor were performed by the use of tumor cell lines as targets. Tumor cells, however, alter their susceptibility to cell-mediated lysis when cultured in vitro [11]. For a better evaluation of cytotoxicity of lymphocytes against tumor cells in cancer patients, autologous combinations of fresh effector and target cells have bcen used. In these studies, peripheral blood lymphocytes from 10 to 50% of cancer patients, depending on tumor types and metastases, expressed lysis of tumor cells freshly isolated from the same patients [14]. The results we have obtained from the studies of cell populations and single cells indicate that CD3-CDI6+ large granular lymphocytes (LGL] from the blood and tumor tissues of cancer patient lyse autologous, freshly isolated tumor cells [7] and release a novel cytotoxic factor with lytic effects on autologous and allogeneic fresh human tumor cells [12]. In addition, results of a previous study showed that autologous tumor cell killing [ATK] was also mediated by CD3+ T lymphocytes; this mediation was performed by T lymphocytes in patients with localized neoplasms and primarily by LGL in patients with metastatic tumors [14]. Cancer patients have been treated with some combination of surgery, chemotherapeutic agents, and radiation. While some patients responded in varying degrees to these treatment modalities, others are unresponsive. Immunotherapy has been proposed as a fourth modality of cancer treatment. Clinical studies with a variety of biological response modifiers (BRM), however, demonstrated response rates of 5—25’7», depending on BRM used and the type of cancer treated. The response rates for most protocols have not been markedly elevated by alterations in doses and schedules of treatment. Thus, it remains unclear why a subset of patients responds to a given treatment modality, while the majority remain unresponsive. It is important to search for some immunological parameter or tumor characteristic that could be used prospectively to predict patient response to a given treatment modality or to provide definitive answers regarding the mechanisms responsible for
an antitumor response.
Many of the anti-cancer chemothcrapeutic agents, including antinomycin D, abrogated the lytic activity of NK cells. In addition, actinomycin D rendered some tumor cells resistant to the lytic effect of NK cells [1, 8). The other treatment regimen, X-ray irradiation, was also demonstrated to affect the cytolytic function of NK cells and cytotoxic T lymphocytes (CTL) [3]. Although the radiation biology of lymphocytes in general has been studied, nothing is known whether X-ray irradiation could modulate the sensitivity of tumor cells to lysis by autologous lymphocytes. In as much as exclusive interaction of X-rays with tumor cells is unlikely to occur under physiologic conditions, it is important for one to identify the modulating effcct of X-ray irradiation on both effector cells and target cells.
Here we describe the biological and clinical significance of blood ATK activity in human cancer patients and the effect of low dose X-ray irradiation on the ATK system.
Clinical Significance of Autologous Tumor Killing (ATK)
We have studied the recognition and killing of freshly isolated human tumor cells by autologous lymphocytes in more than 2,000 patients with various types of neoplasms, including adenocarcinoma or squamous cell carcinoma of the lung, breast, stomach, liver, colon, ovary, and uterus. Blood lymphocytes of patients with a variety of primary localized solid neoplasms exhibited varying levels of cytotoxicity against autologous, freshly isolated tumour cells in 6-hour “Cr-release assay. Significant levels of cytotoxicity above base line were observed in 5—h0% patients, depending on tumor types. The frequency of positive results in ATK tests is comparable to the results of other studies. Although the number of ATK-positive samples decreased when patients developed metastases, the ATK test was not indicated by the clinical and pathological TNM stage while the primary tumor was localized. In patients with localized neoplasms, CD3-LGL and/or CD3+T-lymphocytes exprcsscd A)“K activity, while it was mediated primarily by LGL whcn patients had metastases [l l]. LGL of patients with metastatic cancer suppressed the induction of ATK potential in the autologous mixed lymphocyte-tumor culture, which may explain the low frequency of T-cells with ATK activity in these patients [14].
The patients who has received curative surgery were retrospectively evaluated for the postoperative clinical course [l 3]. Pathological examination showed no tumor cclls in the margins of tumor resections in all patients. No adjuvant anticancer therapy was performed after surgery. When local or distant recurrence developed, patients received chemotherapy, radiation therapy, or biological therapy. The patients who are alive have had follow- up for 5 years. More than 80’f of paticnts who were positive for ATK tests at the time of surgery remained tumor-free and are alive more than 5 years after the operation (Fig. la, b). The other patients with ATK, however, developed metastases and died by 4 years. In contrast, all patients who had no ATK activity relapsed and died within 4 years. When the disease-free
6JeCfs of Low Dose X-ray Irradiation 95
0 12
24 36 48 60
a Postoperative Period (m)
1
0 12 24 36 48 60
- PostoperativePeriod (m)
Fig. la, b Curves for postoperative disease-free interval (a) and total survival
(b) of patients with positive and negative test results for tests for autologous tumor killing (ATK) activity. Blood lymphocytes from patients with localized tumors were tested for ATK activity in a 6-hr “Cr-release assay. Patients were retrospectively evaluated for disease-free period and total survival after 60 months of follow-up, and survival was estimated by Kaplan-Meier analysis.
interval and total survival time were estimated by Kaplan-Meier analysis, the differences observed in curves for postoperative survival (disease-free interval and total survival) for patients with or without ATK activity were
statistically highly significant. The data strongly indicate that the potential of blood lymphocytes to kill autologous fresh tumor cells, tested at the time of surgery, may predict good prognosis of patients with primary localized tumors. The results also suggest that the measurement of ATK function at the time of surgery in cancer patients will provide valuable information for the probability of disease recurrence.
While all patients received curative operation, only those with ATK activity are free from tumor and are alive after more than 5 years. There were no differences in performance status, TNM classification, age or sex between ATK-positive and -negative groups, suggesting that the measurement of ATK activity may represent an independent prognostic parameter. All patients without ATK activity developed recurrence and died within 5 years, showing that a negative result in ATK tests indicates a poor prognosis. It seems thus likely that ATK lymphocytes may be the main effectors in the immunological defense against growth and metastasis of tumor. The test, however, has no absolute prognostic value, since some patients with short disease-free interval and short total survival showed positive results.
The possibility that patients with lymphocytes having ATK activity have better general immune status and thus survive longer than those without the activity could be ruled out for the following reasons [13]. Concomitant tests for other immunological functions revealed that ATK activity did not parallel with T cell proliferation induced by autologous tumor cells in mixed cultures, NK cell activity against the NK prototype target K562, mitogenic response, autologous and allogeneic mixed lymphocyte reactions, and production of interferon, interleukin (IL)-1 and IL-2. It is thus evident that the absence of ATK activity does not reflect general low immunity of the patients. In addition, there were no statistical differences in postoperative clinical curses for patients who were positive in these immunological tests and those who were negative in these test.
A recent report has shown correlation between survival and amplification of oncogene-coamplification units in breast cancer patients. Other reports have implicated the role of tumor suppressor genes in clinical outcome of cancer patients. However, in our study, ATK was independent of abnormal expression of oncogenes and repressor genes of tumor cells. Also, the result
of ATK tests was not associated with DNA patterns (aneuploidy or diploidy) of tumor target cells. There is controversy in the prognostic significance of NK cells: Previous studies supported the prognostic value of NK activity in patients with head and neck cancer. Other studies, however, showed no correlation between blood NK cell activity and postoperative prognosis in melanoma patients. Our data has supported the latter.
Effects of X-ray on ATK Lymphocytes
For conventional radiotherapy regimen cancer patients received X-rays at high doses which alone caused tumor cell death. It was also shown that NK cells are relatively radioresistant, while T lymphocytes and B lymphocytes
are radiosensitive t6]. In fact, low-dose X-ray irradiation (less than 10 Gy) was shown to augment NK cell activity by eradicating radiosensitive suppressor cells for NK cells both in humans and mice [5, 15). To know whether X- ray irradiation affects the human ATK system, we first studied the in vitro effect of X-ray irradiation on human ATK lymphocytes. When blood lymphocytes were irradiated with X-rays at 5 to 20 Gy, they showed a transient increase in cytotoxicity against untreated autologous tumor cells (Fig. 2). This ATK augmentation was dependent on the dose of X-rays, 50 Gy or more abolished ATK acövity. When the X-ray irradiated effector cells were cultured, they completely lost their lytic function. There was no difference in the viable cell recovery between X-ray irradiated and unirradiated lymphocytes, indicating the abrogation of ATK function is not a result of effector cell death. Similar results have been observed with human NK cells [10]. Since ovemight in vitro culture of X-ray-irradiated ATK lymphocytes was required for the abrogation of ATK activity, it appears the X-ray irradiation inhibits the maintenance of functional ATK cells. The inhibitory effect of X-rays was also observed in the induction phase of cytotoxic T lymphocytes, while the effector phase was radioresistant [2]. An inhibition of NK activity caused by irradiation and ultraviolet (UV) radiation was suggested to be attributable to adenosine diphosphoribosyl polymerase, which is known to be activated by DNA strand breakage [4]. Similar mechanisms could be operative for X-ray-induced suppression of ATK activity. Thèse data indicate
% CytOtOXiCitÿ
20
10
10 20
X-rays (Gy)
Fig. 2. kffects of X-ray irradiation of lymphocytes on ATK activity. Blood lymphocytes were irradiated with X-rays at different doses, washed and cultured for 1 h or 18 h at 37°C. They were then washed and tested for lysis of autologous tumor cells.
that X-ray irradiation induces the transient increase and subsequent abolition of ATK cell activity and thus this may be due to the direct effect of X-rays on ATK effector cells and is not attributable to a selective enrichment or loss of ATK cell subsets.
Biological response modifiers (BRM) such as the 1,3-D-glucan sizofiran and streptococcal preparation OK432 have been used in clinical trials to reduce the toxic effect of X-rays. Therefore, it seems of importance to determine whether the X-ray-induced abrogation of ATK activity could be protected by the application of BRM. When blood lymphocytes were stimulated with OK432 immediately after exposure to X-rays, however, they maintained the augmented ATK Activity, the level of which was comparable or even higher than that of control untreated cells (Fig.3). Similar protecting effects were observed with other BRM. Although the mechanism responsible for the protecting effect of BRM is not understood, the findings are nevertheless of clinical importance for the following reasons: First, exclusive interaction of X-rays with tumor cells is unlikely to occur under physiologic conditions. Second, conventional radiotherapy caused suppression of blood ATK cell activity.
%GytOtOXÎCÏt}f
None X-Ray BRM X-Ray + BRM
Fig. 3. Effects of X-ray and BRM on ATK lymphocytes. Blood lymphocytes were irradiated with 5 Gy X-rays, washed and cultured alone or with BRM for 1 h or 18 h at 37°C. They were then washed and tested for lysis of autologous tumor cells.
Effects of X-ray Irradiation on Sensitivity of
Tumor Cells on ATK
Exclusive interaction of X-rays with blood lymphocytes may not be expected to occur in vivo under physiologic conditions. X-rays may also affect tumor
cells. We therefore examined the effect of X-ray irradiation of target cells on their susceptibility to lysis by autologous lymphocytes. When freshly isolated human tumor cells were irradiated with X-rays and cultured overnight, they exhibited increased sensitivity to lysis by lymphocytes in a 6-hour “‘Cr release assay (Fig. 4). X- ray irradiation induced ATK sensitivity in previously resistant tumor cells. The X-ray-induced ATK sensitivity was observed following as little as I Gy X-ray irradiation, reaching a maximum at 5 Gy, the level of which was comparable to that seen at higher doses of up to 20 Gy. Exposure to over 50 Cly X-rays was less effective, and no augmentation was seen at 100 Gy. The induction of ATK sensitivity by X-rays is unlikely to be a result of toxicity, since the dose of X-rays did not influence the viability of target cells. We have previously reported that X-ray irradiation of tumor cells elevates their sensitivity to lysis by NK cells (12, 17]. Our present study has thus extended previous studies to demonstrate that X-ray irradiation enhances the susceptibility of tumor cells without affecting their viability.
9â CytOtOXiClt#’
X-raye (Gy)
Fig. 4. Effects of X-ray irradiation of tumor cells on ATK sensitivity. Fresh tumor cells were irradiated with X-rays at different.doses, washed, cultured for 18 h at 37°C. They were then washed and tested for the sensitivity to lysis of autologous lymphocytes.
We have previously shown that ATK activity was mediated by CD3- large granular lymphocytes (LGL] and/or CD3+ T lymphocytes. In the present study X-ray irradiation of tumor cells is found to induce or elevate their sensitivity to lysls by both effector cells. Tumor-associated macrophages have also been shown to express ATK activity [2]. The X-ray-irradiated
tumor cells exhibited also elevated susceptibility to the cytotoxic effect of autologous monocytes/macrophages in preliminary experiments.
The mechanism involved in the X-ray-induced induction of ATK sensitivity is not understood yet. X-ray irradiation of tumor cells induced or augmented MHC (major histocompatibility complex) class I antigens and adhesion molecules on their surface, which is in part responsible for the observed enhancement of target susceptibility. For conventional radiotherapy regimen cancer patients received X-rays at doses which when alone were toxic to tumor cells. X-ray irradiation is known to produce two distinct types of cell death; programmed death (interphase death) that may be caused by lysosomal enzymes, and reproductive death that is attributable to DNA damage. However, none of these mechanisms appear to be involved in the increased ATK sensitivity observed with irradiated target cells since the doses of X-rays per se did not kill fresh human tumor cells.
Conclusion
The data presented in this communication clearly indicates that the potential of blood lymphocytes to kill autologous, freshly isolated tumor cells, at the time of surgery, may predict a favorable clinical course of patients with localized nćoplasms treated by conventional approaches. In addition, irradiation with relatively low-doses of X-rays of fresh human tumor cells induces or elevates their susceptibility to lysis by autologous T cells and NK cells. Furthermore, the irradiation augments the lytic activity of ATK lymphocytes, the activity of which can be maintained by subsequent stimulation with BRM. Although the mechanism involved in the X-ray-induced modulation of the human ATK system is not fully understood yct, the present data may indicate the possible use of relatively low doses of X-ray irradiation in combination with biological therapy for treatment of human cancer. In fact, preliminary data indicate that a combination therapy with low-dose X-rays and BRM resulted in prolongation of survival time in cancer patients.
REFERENCES
- Chamella, L.J.,Myer, C., Thompson G.E. and Dimitrov, N.V. Chemotherapeutic agents and modulation of natural killer cell activity in J. Immunopharmacol. 1985; 7: 53.
- Gcrber, Radiosensitivity of murine T-lymphocyte cytotoxicity. Radiat. Res.
1984; 100: 365.
- Kadish,S. and Ghossein, N.A. Natural cytotoxicity in patients undergoing
radiation therapy. Am. J. Clin. Oncol. 1983; 6: 53.
- Schacte,, Hansal, S., Anno, J. and Levine, M.J. Polymorphic radiation sensitivity of human natural killer activity: possible role of DNA strand breakage. Hum. Immunol. 1985; 14: 49.
- Schen,N., Homback, N.B., Shidnia, H., Lu, L., Montebello, J.F. and Brahmi,
- Acomparison of lung metastases and natural killer cell activity in daily
fractions and weekly fracüons of radiation therapy on murine B 16a melanoma. Radiat. Res. 1988; 114: 354.
- Stewart, C.C., Stevenson, A.P. and Habbersett, R.C. The effect of low-dose irradiation on unstimulatedand PHA-stimulated human lymphocyte subsets. Int.
- Radiat.Biol. 1988; 53: 77.
- Uchida, and Micksche, M. Lysis of fresh human tumor cells by autologous large granular lymphocytes from peripheral blood and pleural effusions. Int. J. Cancer 1983; 32: 37.
- Uchida and Klein, E. Natural cytotoxicity of human blood monocytes and natural killer cells and their cytotoxic factors: Discriminating effects of actinomycin
- Int.J. Cancer 1985 35: 691—699.
- Uchida, A., Mizutani, Y., Nagamuta, M. and Ikenaga, M. Effects of X-ray irradiation to natural killer cytotohic factor (NKCF). Immunopharmacol Immunotoxicol1989; 11: 521—534.
- Uchida, A., Mizutani, Y., Nagamuta, M. and Ikenaga, M. Effects of X-ray irradiation onnatural killer (NK) cell system. I. Elevation of Sensitivity of tumor cells and lytic function of NK Immunopharmacol immunotoxicol 1989; 11: 507—519.
- Uchida, A. and Mizutani, Y. Autologous tumor killing activity in human: Mechanisms and biological significance. In: New Horizon in Tumor Immunotherapy, Torisu, and T. Yoshida, ed., Elsevier Biomedical Press, Amsterdam, pp. 201—213, 1989.
l2. Uchida, A., Fujimoto, T. and Mizutani, Y. Lysing of fresh human tumor by a cytotoxic factor derived from autologous large granular lymphocytes independently of other known cytokines. Cancer Immunol. Immunother. 1990; 31: 6Œ-64.
- Uchida, A., Kariya, Y., Okamoto, N., Sugie, K., Fujimoto, T. and Yagita, M. Predictionof postoperative clinical course by autologous tumor killing activi:y in human lung cancer J. Natl. Cancer Inst. 1990; 82:1697—1701.
- Uchida,, Kariya, Y., Okamoto, N., Kihara, T., Inoue, N. and Sugie, K. Biological significance of autologous tumor killing activity in human cancer patients and its modulation by biological therapy. In: Chemoimmuno Prevention of Cancer,
- Pastorinoand W.I. Hong, eds., Thime Medical Publishers, New York, pp. 9— 13, 1991.
- Waer,, Vanrenterghem, Y., Ang, K.K., Van der Schueren, E., Michielsen, P. and Vandeputte, M. Comparison of the immunosuppressive effect of fractionated total lymphoid irradiation (TLI) vs. conventional immunosuppression (CI) in renal cadeveric allotransplantation. J. Immunol. 1984; 132: 1041.
11. Changes in Subpopulations of Lymphocytes after Exposure to Ionizing Radiation
in vitro and in vivo
- Dehos,R. Kriehuber, H. Czempiel, I. Baumgartner,
- Egblomasseand W. Burkart
Institut für Strahlenhygiene des Bundesamtes für Strahlenschutz Neuherberg Krankenhaus Manchen-Schwabing, Abt. F. Strahlentherapie, Germany
Introduction
Immunological parameters were assessed in view of their potential use as biological indicators for radiation exposure. Since it is known that individual lymphocyte subpopulations show different sensitivities to ionizing radiation, one of the investigated parameters is the change in the relative and absolute number of several subpopulations of lymphocytes in peripheral blood. The aim of the project is to investigate if the currently applied rough estimation of large doses after accidental exposure [3] using changes in the absolute number of peripheral lymphocytes could be refined by using lymphocyte subpopulations. For this purpose, changes in lymphocyte subpopulations were examined after in vitro irradiation of peripheral blood mononuclear cells as well as in the blood of radiotherapy patients during treatment.
Material and Methods
Determination of Lymphocytes Subpopulations and Dead Cells
Peripheral blood mononuclear cells were separated by gradient centrifugation and incubated for three days in MEM containing 10‘7o human serum and antibiotics. Analysis of lymphocyte subpopulations and dead cell in irradiated and control samples was done shortly after irradiation and on each of the following three days. For determining the lymphocyte subpopulations, cells were incubated with they appropriate fluorochrome-conjugated antibodies and analysed by flow cytometry. The percentage of dead lymphocytes was determined by using propidium iodine and flow cytometry.
in vitro Irradiation
Isolated mononuclear cells from peripheral blood of healthy donors were irradiated with 250 kV X-rays at a dose of 0.4 Gy/min.
Radiotherapy Patients
A total of 65 radiotherapy patients with various diseases and irradiation conditions were examined. Blood was taken before, during and at the end of therapy. 20 of the patients could be classified into groups according to disease or type and size of the irradiated field.
Changes in SubpOpulations ofL ymphoc ytes 103
Results
iii vitro irradiation
Figure 1(a), (b) (c) and (d) show the dose-response-relationships for changes in the relative proportion of T and B cells and CD4+ Leu 8 –, CD4 — Leu8+
% of lymphocytes
100
80
60
40
20
(a)
0
dose (Gy)
CD4-I-Leu8-
% of lymphocytes
(b)
do (GY)
Fig. 1. (Contd.)
CD4-Leu8+
96 of lymphocytes
25
20
15
10
5 (c)
0
dose (Gy)
CD4+ Leu8+
’K of lymphocytes
30
20
10
0
0 0.5 1 2 3 5
dose (Gy)
(d)
Fig. 1. (a) Dose-response-relationship for changes in the relative number of T (CD3) and B (CD20) lymphocytes; (b) CD4+Leu8—; (c) CD4+Leu8+ and (d) CD4— Leu8+ subpopulations after irradiation of mononuclear cells izi viiro for several days after irradiation.
and CD4+ Leu8+ subpopulations of T helperinducer cells [2] in irradiated mononuclear cells for the day irradiation occurred and the three following days. Figure 2 shows time-related changes for the T suppressor/cytotoxic
- Aoflymphocytes
time (days)
Fig. 2. Time dependency of changes in T suppressor/cytotoxie (CD8) cells after irradiation with various doses.
(CD8) cells and contains results from one representative experiment out
of 4.
Radiotherapy Patients
Figure 3(a) shows changes in the relative proportion of the same subjx›pulations as in Fig. 1(b), (c) and (d) during radiation treatment in one group of patients who were irradiated in the thorax region. The pre-irradiation values for each patient are l0O9o. Each point represents the result from one individual patient for the tumour dose value indicated on the x-axis. Regression lines calculated for each subpopulation are drawn into graphs to indicate the trend.
Discussion
Our previous experiments have shown that lymphocyte subpopulations do not show definite changes after in vitro irradiation when analysed on the day of irradiation [l]. On the contrary remarkable changes in lymphocyte subpopulations could be observed in radiotherapy patients [l] (Fig.3(a)). Moreover it is known that lymphocytes may die from interphase death. Therefore we examined the changes in subpopulations of lymphocytes occurring in irradiated mononuclear cells during several days of culturing. As can be seen from Figs. 1(a) and 2 the differences between the relative numbers of various subpopulations and of dead lymphocytes are increasing in dependence of time and irradiation dose. Whereas for B cells (CD20) no significant differences between irradiated and control cells can be observed
CD4-i- Leu8-, CD4-r Leu8-r and CD4- Leu8-I-
CD4+ Leu8—
— CD4+ Leu8+
—- CD4 – Leu8+
- CD4+Leu8-
- CD4+Leu8+
° CD4 – Leu84
0
(a)
1
1
(b)
20 40
tumour dose (Gy)
20 40 60
tumour dose (Gy)
Fig. 3. Changes in the relative number of various lymphocyte subpopulations in the course of radiotherapy.
even for day 3 and the high dose values, the percentage of T cell decreases in dependence of dose at least on day 3. In contrast B cells are more sensitive than T cells in radiotherapy patients as can be seen in Fig. 3(b). This is well known from the literature. Results for the lymphocytes bearing the CD4 and the Leu8 antigen, however, are rather similar for in vitro and in vivo irradiation. After the in vitro exposure of cell the number of CD4+Leu8+ and of CD4— Leu8+ cells decreases in dependence on dose
DEAD CELLS
9L of lymphocytes
time (days)
Fig. 4. Dead cells (CD8) after irradiation with various doses.
during culturing time (Figs. 1(c) and (d)). The percentage of CD+Leu8- cells, on the contrary, increases with dose on all three days following irradiation with the only exception of the dose value 5 Gy on day 3 (Fig.1(b)). Therefore the relative number of T helper/inducer cells as a whole remains rather constant (approx. 56% of lymphocytes) until day 3 where a dose dependent decrease can be observed.
In radiotherapy patients (Fig. 3(a)) CD4+Leu8— cells do not change in a definite way during treatment whereas the percentages of CD4+Leu8+ and CD4—Leu8+ cells remaining in peripheral blood are reduced to approx. 20% of pre-irradiation values at the end of therapy for the patients who were irradiated in thorax region.
As can be seen in Fig. 2, T suppressor/cytotoxic cells (CD8) also decrease in dose dependence during culturing time. This again is in contrast to radiotherapy patients (data not shown) where CD8 cclls did not show definite changes during treatment.
Figure 4 also shows the time and dose dependency of the number of dead cells. Since those dead cells were not separated from living cells before analysis of lymphocyte subpopulations it can be concluded that dead cells still bcar surface antigens, but obviously some antigens, above all the Leu8 antigen, disappear from cell surface after radiation exposure. Therefore the situation is different between ifl vitro irradiation and radiotherapy patients where no dead cells are found in peripheral blood. Since, however, cells that bear the Leu8 antigen are reduced after in vitro irradiation as well as in radiotherapy patients, it would be interesting to examine if the Leu8+ cells are equally distributed between living and dead cells.
In conclusion it can be stated that there are still many effects occurring in the living organism after irradiation that can not be observed in vitro. On the other hand the time delay between irradiation and analysis of blood cells also plays an important role for in vivo irradiation. Therefore analysis of lymphocyte subpopulations several days after irradiation iit vitro results in changes of lymphocyte subpopulations that are closer to the effccts after iii vivo irradiation.
Moreover, it can be stated that lymphocytes bearing the Leu8 antigen are comparably sensitive to in vivo as iii vitro irradiation. Therefore it should be further investigated, if they can be used as biological indicators.
Acknowledgement
The research project was supported by the Federal Minister of Environment, Nature Conservation and Nuclear Safety.
REFERENCES
- Dehos,, Beaufort, F., Erzberger, A., Czempiel, H., Egblomasse, C. and Hinz,
- In: Advances in Analytical Cellular Pathology. Proceeding of the FirstConference of theEuropean Society for Analytical Cellular Pathology, SchloQ Elmau, F.R.G., 12—17 November 1989.
Eds.: Burger, G., Oberholzer, M., Vooijs, G.P. Excerpta Medica, Amsterdam— New York-Oxford 1990; 279—280.
- Lanier, L.L., Engleman, E.G., Gatenby, P.,Babcock, G.F., Warner, N.L. and Herzenberg, A. Immun Rev 1983; 74: 143—150.
3 UNSCEAR Report: Sources, effects and risks of ionizing radiation, United Nations Scientific Committee on the Effects of Atomic Radiation, 1988 Report to the General Assembly, with annexes, United Nations, New York, 1988; pp 554— 559.
- AdaptiveResponse Studies in Diploid Yeast Saccharomyces cerevisiae Exposed
to HTO Beta and Gamma Radiation
B.S. Rao, K.B. Anjaria and N. Sankara Naiayanan
Division of Radiological Protection
Bhabha Atomic Research Centre, Bombay, India
Introduction
It has been observed in a number of studies that cells exposed to low levels of radiation or chemicals become less susceptible to damage induced by high doses of same or other agents [3, 6, 7, 8, 9, 11]. This type of adaptive response has been reported in bacteria, plants, CHO cells, human skin fibroblasts or in human peripheral lymphocytes for alkylating agents or radiations. In this paper, we have studied the effect of low doses of gamma radiation using a diploid yeast strain. Possibility of human exposure simultaneously to a variety of genotoxic agents is quite high. Hence, it is very important to have a clear understanding of the mode of interaction between these agents for evaluation of risk to human beings. In this paper, we have also investigated in details, the interaction between very low energy
beta radiation from tritium (HTO) and gamma radiation from 60Co, using
the induction of gene conversion in diploid yeast as the end point.
Material and Methods
Yeast strains Saccharomyces cerevisiae BZ34 and D7 Revert to arginine and tryptophan independence respectively upon exposure to genotoxic agents (2, 13). The genotypes of these strains are:
BZ34:
D7:
a + arg 4—4 + thr 1 + tf]3 5—48
a petl + arg 4-17 + leu I—12 trp 5—48
+ ura 3 his 5—2 lys 1—1 ade 2—1
metl + his 5—2 lys 1—1 ade 2—1
a ade 2—40 W 5—12 ilv l— 92
a ade 2—119 trp 5—27 ilv 1—92
Gamma irradiation was performed in a 60Co gamma cell with a dose rate of 10 Gy/min. Beta irradiation was performed by selecting an appropriate activity of tritiated water (HTO) in the cell suspension. A concentration of
37 MBq/ml corresponds to a dose of 84.6 mGy/hr. Adaptive response was studied by irradiating the cells to 1 Gy of beta radiation under either lag or log phase followed by an acute exposure to graded doses of gamma radiation. Interaction between beta and gamma radiations was investigated under acute as well as low dose rate irradiation conditions.
Results
Adaptive response studies—Log phase
Adaptive response was studied by exposing cells to priming dose of 1 Gy of HTO beta rays during log phase. Cells were exposed to 24 hours in the presence of 18..5 MBq/ml of HTO. Subsequently the cultures were harvested and the cells washed repeatedly with sterile distilled water to remove the traces of HTO. These cells were exposed to acute gamma radiation doses of 20 Gy, 40 Gy and 60 Gy. Cultures grown in the absence of HTO were also exposed to gamma radiation to serve as positive controls. Results of these experiments are summarized in Table 1. Results suggest a purely additive interaction.
Table 1. Gene conversion frequency in 5accharomyces cerevisiae BZ34 strain exposed to gamma radiation alone or to combination of beta and gamma radiation
Conversion frequency per 10 cells
| Treatment details | Observed* | Expected |
| Gamma dose 20 Gy (acute) | 435 A 45 | |
| Gamma dose 40 Gy (acute)
Gamma dose 60 Gy (acute) |
778 -£ 60
962 d- 6l |
|
| Beta dose 1 Gy (18.5 MBq/ml) | 82 -b 14 | |
| Beta dose 1 Gy + gamma dose 20 Gy | 565 -b 63 | 517 |
| Bcta dose I Gy + gamma dose 40 Gy | 1002 A 127 | 860 |
| Beta dose 1 Gy + dose 60 Gy | 1122 -k 124 | 1044 |
*Conversion frcqucncy corresponds to the mean value derivcd from seven experiments. Background frequency of 15—20 convcrtants per million cells have been subtracted and the net values presented. Errors represent the standard deviation of the mean.
In another experiment, cells were exposed in YEPD broth containing HTO to l Gy beta dose during the 2 h corresponding to lag phase. This resulted in the induction of 31 convertants/106 viable cclls. Cells were then washed repeatedly and exposed to 20 Gy of gamma radiation. This resulted in the induction of 440 convertants/106 survivors following combined treatment. Cells not exposed to HTO beta rays were also exposed to 20 Gy of gamma radiation which yielded the conversion frequency of 425 per 106 survivors. Since the observed value of 440 convertants per 10 survivors following combined treatment was not significantly different from the expected
value of 455 convertants per 106 survivors, the experiment indicated the absence of adaptive response.
Interaction between HTO Beta and “Co Gamma Rays
Acute Exposure
Interaction between HTO beta and 60Co gamma rays was investigated by irradiating yeast Saccharomyces cerevisiae BZ34 to 12.5 HTO beta during the lag phase of growth, subsequently, these cells were washed and resuspended in distillGd water and irradiated with 12.5 Gy of 60Co gamma rays. Results of these experiments also showed an additive response (Table 2).
Table 2. Interaction between beta and gamma radlation for the lnductlon of gene conversion In Saccharomycec cereviciae DZ34 under acute exposure conditions
Treatment details
Conversion frequency per 106 survivors
Log phase
12.5 Gy beta
12.5 Gy gamma
12.5 Gy beta +
12.5 Gy gamma
Observed
339 A 6
169 A 4
524 -k 8
Expected
508 A 7
Low Dose Rate
Interaction studies under chronic conditions were performed at 0-4°C using D7strain of yeast. In these experiments yeast cells were exposed simultaneously to HTO beta and 60Co gamma rays at a dose rate of 250 mGy per hour. Cells were also exposed to gamma radiation alone or beta radiation alone under similar conditions. Results obtained in 3 independent experiments suggest a subadditive response under low dose rate conditions (Table 3).
Table 3. Interaction between HTO beta and ^Co gamma under low dose rate cond ltlon
(0.25 Gy. h ’, 0-4°C) D7
Conversion frequency per 106 survivors
Expected.
Discussion
The results obtained in the adaptation studies indicate that HTO beta dose of 1 Gy given in either lag or log phase does not induce adaptive response in yeast cells on subsequent exposure to challenging doses of gamma radiation. Studies with bacteria and mammalian cells with alkylating agents have indicated the induction of adaptation enzymes viz. DNA glycosylase and DNA alkyltransferase which leads to adaptive response [1, 4, 7]. Adaptive response has also been reported in human lymphocyte exposed to low levels of ionizing radiation and was reported to be due to induction of some specific proteins [10, 12]. However, results obtained in the present study indicate a total absence of adaptive response in our system. In other words, pretreatment with HTO beta rays does not influence the subsequent response to gamma rays. This suggests that HTO treatment fails to induce any error- free repair processes which could in turn reduce the response to subsequent gamma irradiation.
The results obtained in the interaction studies indicate that acute irradiation to HTO beta and 60Co gamma radiation under metabolically active conditions (in lag phase of growth) yielded an additive response suggesting the absence of induction of repair enzymes.
Simultaneous exposure of cells to beta and gamma radiation at low dose rate, at 0—4°C, exhibited subadditive response (see Table 3). Since the irradiations were performed under metabolically inert conditions, the possibility of interference from any repair processes cannot be invoked to explain the results. At present it is unclear if such an antagonism could arise by pure physical interaction between damage induced by the two radiations under metabolically inactive state.
The phenomenon of adaptive response seems to be influenced by a variety of factors such as the experimental system, end point scored and mutagen used. Hence, this phenomenon can not be considered as universal. Both in bacteria and mammalian cell system there is some evidence for the induction of DNA enzymes as a result of exposure to low concentrations of alkylating agents. This could certainly elicit a reduced response to a challenging treatment. Quite a few reports suggest the adaptive response for the induction of chromosomal damage in human lymphocytes irradiated in vitro, but then, it is not consistently seen in all individuals tested. A lot of experimental work is necessary to explain this discrepancy and to have a clearer understanding of the mechanism of adaptive response.
nrFrRENCES
- Evenscn, and Scebcrg, G. Adaptation to alkylation resistance involves the
induction of DNA glycosylasc. Nature, 1982; 296: 773—775.
- Fogcl, S.and Mortimer, K. Informational transfer in meiotic gene conversion. Proceedings of the National Academy of Sciences, USA, 1969; 62: 96—103.
- Ikushima, Radio-adaptive response: A novel chromosomal response in Chinese
hamster cells in vitro. Chromosomal aberrations, edited by G. Obc and A.T. Natarajan (Springer-Verlag Berlin Heidelberg New York), 1990; pp. 151—162.
- Karran, P., Lindahl, T.and Griffin, B. Adaptive response to alkylating agents involves alteration in situ of O -methylguanine residues in Nature, 1979; 280: 76—77.
- Rieger, R., Michaelis, A.and Nicoloff, H. Inducible repair processes in plant root tip “Below additivity effects” of unequally fractionated clastogen concentrations, Biology, 1982; 101 125—138.
- Samson, and Cairns, J. A new pathway for DNA repair in Escherichia coli,
Natures(Lond.), 1977; 267: 281-283.
- Samson, L.and Schwartz, J.L. Evidence for an adaptive DNA repair pathway in CHO and human skin fibroblast cell Nature, 1980; 287: 861—863.
- Sanderson,J.S. and Morley, A.A. Exposure of human lymphocytes to ionizing radiation reduces mutagenesis by subsequent ionizing radiation, Mutation Research, 1986; 164, 347—351.
- Sankaranarayanan,, Duyn, A.V., Loos, M.J. and Natrajan, T. Adaptive response of human lymphocytes to low-level radiation from radioisotopes or X-rays. Mutation Research 1989; 211, 7-12.
- Wolff,, Wiencke, J.K., Afzal, V., Youngblom, J. and Cones, F., The adaptive response of human lymphocytes to very low doses of ionizing radiation: a case of induced chromosomal repair with the induction of specific proteins. Low dose radiatiom: biological basis of risk assessment, edited by K.F. Baverstock and J.W. Stather. (Taylor and Francis, London) 1989; pp. 446-454.
- Wolff,, Olivieri, G. and Afzal, V. Adaptation of human lymphocytes to radiation or chemical mutagens: Differences in cytogenetic repair. Chromosomal aberrations, edited by G. Obe and A.T. Natarajan (Springer-Verlag Berlin Heidelberg New York) 1990; pp. 140—150.
- Youngblom, ITI.,Wiencke, J.K. and Wolff, S. Inhibition of the adaptive response of human lymphocytes to very low doses of ionizing radiation by the protein synthesis inhibitor cycloheximide Mutation Research, 1989; 227: 257—261.
- Zimmermann,K., Kem, R. and Rasenberger. A yeast strain for simultaneous detection of induced mitotic crossing over, mitotic gene conversion and reverse mutation. Mutation research, 1975; 28: 381—388.
13. Biological Basis of Radiation Protection Standards’ (ICRP-60)
S.D. Soman and B.S. Rao*
Chairman, Atomic Energy Regulatory Board, Bombay, India
*Division of Radiological Protection, Bhabha Atomic Research Centre, Bombay, India
Introduction
Man has always been exposed to ionising radiation from various natural sources. The cxtent of this natural background exposure varies with the locations. However no ill effects have been uniquely correlated with these variations. Either no deleterious effects are produced at these levels of exposure or their frequency is too low to be statistically observable.
The harards of ionising radiation became apparent in connection with exposures from man made source.s The discovery of X-rays and separation of radioactive substances at the end of last century, brought in very great benefits, albeit associated with unforseen hazards. Within five years, 170 cases of radiation injuries were recorded and by 1922 about 100 radiologists had died from effects of radiation over-exposure. These observations of radiation injuries in man and results of radiobiological experiments improved the knowledge of health effect of radiaion and thereby the need for protection became obvious. The International Commission on Radiological Protection (ICRP) was established in 1928 and it published its first recommendation in 1931. The recommendations of ICRP issued from time to time deal with basic principles of radiation protection on the basis of available knowledge from radiobiological and epidemiological findings.
Radiobiological and Epidemiological Basis of Radiation Protection
Direct source ot information on radiation hazards in man is obviously the follow-up of population groups exposed to certain levels of radiation. Use of statistical techniques and adequate identification and diagnosis of observed effects is covered by the term “Epidemiology”. Epidemiological information are mostly available for situations involving high doses and dose rates. On the other hand, in radiation work, in normal operation we encounter low
{This paper is based on an eariier publication in AMPI Medical Physics Bulletin on this subject by the same authors.
doses an dose rates, which therefore become the area of interest for assessment or radiation risks for purpose of setting up protection standards.
Epidemiological studies do not provide an understanding as to how radiation produces cancer and are therefore not useful in extrapolating the results from high doses and dose rates to low dose region. The mechanisms of induction of radiation effects and relationship between dose and response can be obtained from the radiobiological experiments with microorganisms, mammalian cells and experimental animals. The radiobiological information, thus provides conceptual basis for interpreting epidemiological results using the dose response relationship based on mechanistic model(s).
Epidemiology: At present three principal sources of information on stochastic effects are the epidemiological studies on survivors of Hiroshima and Nagasaki, patients exposed to radiation for medical treatment or diagonsis and some groups of workers exposed to radiation or radioactive substances at work. Among these, the survivors of Hiroshima and Nagasaki provide the single largest group being scientifically followed-up for more than four decades and these results provide the most important data base on radiation effects in man. These are then coupled with other eipidemiological results from the remaining two sources.
Radiobiology : in vitro cellular research clearly shows that relevant effects of radiation result primarily from damage to cellular DNA. Two mechanisms are involved:
- Directeffect of ionisation in the DNA
- Indirecteffects from production of reactive chemical radicals in the vicinity of DNA followed by their diffusion to DNA, inducing chemical
Both these are probabilistic in nature, probability being proportional to dose and target volume. With low LET radiation at low doses (<0.02 Gy), the probability of these two effects is vanishingly small. AT high doses the probability of “double event” becomes more significant and dose rate dictates the time elapsing between two events. Radiation damage in DNA is subjected to very efficient repair mechanism. The repair is highly probable in case of strand breaks but is not totally error-free. This small probability of damage not being repaired correctly, is also then proportional to dose.
There is strong evidence to suggest that majority of cancers originate from damage to single cells. Cancer initiation starts from deregulation of cell growth, and the loss of control of cell proliferation. Although the cells may have initiating changes they will not lead to malignancy until they are stimulated or “promoted” to proliferate due to presence of chemicals, hormones present in their environment. Radiation exposure would contribute to one relevant mutation step among the many needed for induction of a cancer
cell. Thus the age at which radiation induced cancer is expressed is not very different than that for spontaneous cancer. In this sense radiation replaces time, advancing slightly the occurencce of malignancy, having the same effect on reaching a higher age.
Background
Follow up of the A-bomb survivors from Hiroshima and Nagasaki constitutes the most important human data-base for the evaluation of radiation risk. Recent revision of dosimetry for survivors and the information on cancer morality from the longer follow-up by Radiation Effects Research Foundation (RERF) led to the revision of risk estimate by the United Nations Scientific Committee on the Effects of Atomic Radiations (UNSCEAR) [l] and National Academy of Sciences Committee on the Biological Effects of Ionizing Radiations (BEIR) [2]. Subsequently, it was realized that the risk of radiation had been underestimated at least by a factor of about 3. In the light of the above information, Inter-national Commission on Radiological Protection (ICRP) updated the recommendations of 1977 (ICRP 26) [3]. The Commission has now recommended a reduction in the dose limits for occupational exposure as well as for exposure of public. Adequate degree of flexibility is maintained to enable the practical implementation of hte new recommendations. These recommendations have been made with the objective to provide necessary guidelines for regulatory bodies to ensure adequate radiological protection. Annexure ‘A’ (ICRP-60) (4) provides information on revised concepts regarding the quantities and units mostly obtained from the reports of International Commission on Radiation Units and Measurements (ICRUM, 1980; 1985). Radiation weighting factor +R is introduced in the place of
quality factor Q, to account for the higher biological effectiveness of more
densely ionizing radiations. Photons of all energies, electrons and muons have been assigned a +R •alue of unity, whereas, neutrons in the enrgy, range of 100 kcV < np < 2 MeV, alpha particles, fission fragments and
heavy nuclei have been assigned a +R value of 20 (see Table 1). The commission now defines the term ‘equivalent dose’ (JfT) (formerly the term dose equivalent’ was used) which is the product of absorbed dose in a
tissue and the radiation weighting factor +R In the light of new information regarding the sensitivity of individual organs, tissue weighting factor It, has been assigned to different organs (see Table 4). The sum of these weighting factors is unity. Equivalent dose weighted by IV, is now defined
as E, the effective dose (formerly called the effective dose equivalent). Unit of effective dose is Sievert (Sv). Annexures B and C (ICRP 60) deal with the dose response relationships for both non-stochastic and stochastic effects, which provide a sound biological rationale for the revision of risk estimates. Since killing of a significant fraction of cells in a tissue or organ is essential to elicit a clinically detectable response, the non-stochastic effects occur only above a certain threshold dose. Even though the inital events at cellular
Table 1. Radiation weighting factors•
Type artd energy range
Photons, all energies
Electrons and muons, all energies**
Neutrons, energy < 10 KeV
10 KeV to 100 KeV
- 100KeV to 2 MeV
- 2MeV to 20 MeV
- 20MeV
Protons other than recoil protons, energy > 2 MeV
Alpha particles, fission fragments, heavy nuclei
Radiation weighting factor
1
1
5
10
20
10
5
5
20
* All values relate the radiation incident on the body or, for internal sources, emitted from the source.
**Excluding Auger electrons emitted from nuclei bound to DNA.
level are essentially random, response at tissue level has a deterministic character. Hence, the Commission has now replaced the term ‘non-stochastic’ effects by ‘deterministic’ effccts. Stochastic effects namely carcinogenesis and genetic effects, arise from non-lethal damage at cellular level and hence can occur even after exposure to low dose.
Revision of Cancer Risk Estimates
Dosimetry: Risk estmates for radiation carcinogenesis is largely based on the human data derived from survivors of Hiroshima and Nagasaki. Risk estimates were based on the tentative 1965 dosimetry (T 65 DR). Subsequently, the dosimetry was revised in 1986 (DS 86) to correct for building and organ transmission factors, bomb yield and reduced levels of neutron component due to humidity conditions that prevailed at Hiroshima and Nagasaki. Revised dosimetry revealed an increased risk of 48—95% for leukaemia and 5—38% for solid tumours. Among the solid cancers stomach, brcast, lung and colon showed large excess risk.
Risk Projection Model: Appearance of many new solid tumours among the A-bomb survivors during the last two decades, suggested a relationship between age and the expression of excess cancers. Furthermore, it was observed that the number of excess cancers continued to increase even after 40 years of exposure. It was clear from these observation that the absolute risk model which suggests a fixed level of cancer risk for a given level of exposure does not hold good for solid tumours. On the other hand, the excess cancer incidence was found to be a function of the base-line cancer incidence. Since the incidence of many cancers increases with age, radiogenic excess cancers also express in larger number during the older age (Relative risk model). Almost two third of the 94,000 A-bomb survivors are still
alive. Hence life-time risk has to be evaluated by projecting the existing risk over the entire life time using the relative risk model. Relative risk model (also called multiplicative risk model) predicts a much higher risk as compared to that by the absolute risk model used in ICRP-26 [3].
Nominal Fatality Probability Coefficients: Applying the multiplicative risk model, ICRP has calculated the life-time risk of mortality due to radiogenic cancer in five representative populations, namely, China, Japan, Puerto Rico, UK and USA. with different spontaneous cancer rates. The risk expressed as “nominal fatality probability coefficient”, ranged from 6 to 13% per Gy and an average of 9.4% Gy*’ for acute exposure. Similar calculations have been carried out by UNSCEAR and BEIR Committees, which are about 20% higher than that of ICRP. Taking these into account ICRP has estimated the fatality probability coefficient as 10’7o Gy*’. For chronic exposures, using DDREF of 2 (see next para) cancer fatality probability becomes 5% or 5 x 10 2 Gy*’. This can be compared with 1977 estimate of 1.25 x l0°2Gy*’. Such calculation have also been extended to different cancer sites in males and females of different age groups (see Table 2). In general, females show a higher risk (-20%) as compared to males. Age truncated risk analysis
Table 2. Nominal probability coefficient for individual tissues organs
Risk factors Probability of fatal cancer (ICRP-60)
Organ
Bladder
(ICRP 26) Workers Whole population (10-2 SV*’) (10*’ Sv*’)
0.24 0.30
Bone marrow Bone surface Breast
Colon Liver Lung
Oesophagus Ovary
Skin
2 x 10*3JSv
5 x 10“/Sv
2.5 x 10*3/Sv
2 x 10*3/Sv
1 x 10“/Sv
0.40 0.50
0.04 0.05
0.16 0.20
0.68 0.85
0.12 0.15
0.68 0.85
0.24 0.30
0.08 0.10
0.02 0.02
Stomach Thyroid Remainder Gonads
Total
— 0.88 1.10
5 x 10“/Sv 0.06 0.08
5 x 10‘’/Sv 0.40 0.50
4 x 10*’/Sv 0.60 1.00
(2 generations) (all generation i.e. at equilibrium)
1.66 x 10*2/Sv 4.60 6.0
shows a higher risk for children (a factor of 2) in the age range of 0—19 years. On the other hand, the working population in the age range 18—64
years have a risk coefficient 209» lower than that for the whole population (4 x 10°* Gy‘1).
Dose and Dose Rate Effectiveness Factors: Risk figures derived from the Japanese data pertain to high dose and acute exposure (high dose-rate conditions). Most of the radiobiological data for low LET radiations suggest a reduction in the effetiveness of radiation for cancer induction with the reduction in dose as well as dose rate. This is evident form the linear- quadratic dose response curves seen for many radiobiological end points such as induction of mutations and chromosomal aberrations in mammalian system. Furthermore, carcinogenesis data at low dose rates obtained from mice suggests a reduction factor in the range of 2—10 for different tissues and organs. In the absence of any reliable information from human epidemiological data from low dose-rate expoures. ICRP has adopted a dose and dose-rate effectiveness factor (DDREF) of 2. Commission has recommended that the probability coefficients be reduced by a factor of 2 for equivalent doses resulting from absorbed doses below 0.2 Gy and from all exposures at dose-rates less than 0.1 Gy h ‘. Revised information has enabled assignment of probability coefficients for many organs and tissues not included in ICRP-26. Further, the earlier risk coefficient have been modified in the light of new information.
Detriment
Aggregated Detriment: The commission realised that it is inadequate to consider the fatal cancer risk and serious genetic effects in two generations for assessing the harm associated with radiation exposure. The total harm that may be experiehced by an exposed group and its descendants is now assessed in ICRP-60 for estimating “Detriment” on the basis of: (a) fatal cancer risk, (b) morbidity associated with non-fatal cancers and (c) serious as well as other genetic disorders in all generations. Relative life lost due to each type of cancers is also taken into account while arriving at the aggregated detriment which is expressed as probability of death or its equivalent. Detriment due to nonfatal cancers is arrived at by multiplying the number of such cancers by lethality fraction (k) associated with each specific type of cancer. Lethality fraction (k) for different types of cancer is arrived at from the mortality data available for specific sites [5]. Allowance is made for the length of life lost by each type of cancer to assess the harm. This would depend upon the latent period for each of cancer and the corresponding frequency of age specifie spontaneous background incidence of solid tumours. Leukemia with a very short latent period of 2—3 years causes an average loss of life of 30 years. Genetic disorders of serious nature results in an average lost life of 20 years. Weighted average of life lost for radiation induced mortality is calculated as 15.7 years. Length of life lost for each specific cancer, relative to the weighted average value of
15.7 years yields the relative length of life lost for that cancer. Detriment due to each specific organ/tissue is calculated taking into consideration the
fatal cancers, non-fatal cancers and weighting by relative length of life lost. Further details of evaluating the contribution of each organ to the total detriment is shown in Table 3. This information forms the basis of new tissue weighting factors assigned by ICRP-60 (Table 4).
Total detriment of 725.3 (Table 3) refers to the whole population and includes the detriment from serious genetic disorders arising in all future
Table 3. Relative contribution* of organs to the total detriment
|
Organ |
Probability of fatal
cancer |
Lethality fraction
(i) |
Relative non-fatal
contribution |
Relative length of
life lost |
Total detriment
f.ilt{2—k) |
Relative contribution*
to detriment |
| (F.10“Sv*’) | (2—k) | ‹ilt) | (l0“Sv*’) | |||
| Bladder | 30 | 0.50 | 1.50 | 0.65 | 29.4 | 0.040 |
| Bone marrow | 50 | 0.99 | 1.01 | 2.06 | 104.0 | 0.143 |
| Bone surface | 5 | 0.77 | 1.30 | 1.00 | 6.5 | 0.009 |
| Breast | 20 | 0.50 | 1.50 | 1.21 | 36.4 | 0.05 |
| Colon | 85 | 0.55 | 1.45 | 0.83 | 102.7 | 0.141 |
| Liver | 15 | 0.95 | 1.05 | 1.00 | 15.8 | 0.022 |
| Lung | 85 | 0.95 | 1.05 | 0.90 | 80.3 | 0.111 |
| Oesophagus | 30 | 0.95 | 1.05 | 0.77 | 24.2 | 0.034 |
| Ovary | l0 | 0.70 | 1.30 | 1.12 | 14.6 | 0.020* |
| Skin | 2 | 0.002 | 2.00 | 1.00 | 4.0 | 0.006 |
| Stomach | 110 | 0.90 | 1.10 | 0.83 | 100.0 | 0.139 |
| Thyroid | 8 | 0.10 | 1.90 | 1.00 | 15.2 | 0.021 |
| Remainder | 50 | 0.71 | 1.29 | 0.91 | 58.9 | 0.081 |
| Gonads | 100** | 1.33 | 133.3 | 0.183** | ||
| Total | 600 | 725.3 | 1.000 |
*Cancer risk only.
**Genetic risk only.
*Relative contribution rcfers to the fractional contribution to the detriment from each organ or tissue, bone marrow, colon, stomach, lung and gonads are the first category; bladder, breast, liver, oesophagus and thyroid, second category; and bone surface, skin the last category (contributes very little).
k value based on mortality data for 5 and 20 years from US. DHHS 1989 [5] (Metler and Sinclair).
ift refers to the ration of life lost due to specific cancer to the weighted aveerage of the length of life lost (i).
generations (l 33. 10“ Sv‘’). Gnetic risk includes all types of genetic disorders namely, autosomal dominant, sex-linked recessive, autosomal recessive, chromosomal disorders and multifactorial disorders. Instead of considering the detriment arising from only two generations as in (ICRP-26, 1977), the present recommcndations cover the genetic risk for all future generations. For working population in the age range of 18—64 years the total detriment is 5.54’7o SV*1, i.e. 4.74’7o Sv*1 for cancers and 0.80% Sv*1 for genetic disorders), approximately 20% less than that for the whole population in the age range of 0—90 years.
Table 4. Tissue weighting factors (Wt)
| ICRP 60
(1990) (*) |
Organ | ICRP 26
(1977) (J‹) |
| 0.01 | Bone surface | 0.03 |
| 0.01 | Skin | |
| 0.05 | Bladder | |
| 0.05 | Breast | 0.15 |
| 0.05 | Liver | |
| 0.05 | Oesophagus | |
| 0.05 | Thyroid | 0.03 |
| 0.05’ | Remainder* | 0.30 |
| 0.12 | Colon | |
| 0.12 | Lung | 0. l2 |
| 0.12 | Red bone marrew | 0.12 |
| 0.12 | Stomach | |
| 0.20 | Gonads | 0.25 |
*Remainder organs in ICRP-60 include adrenals, brain, small intestine, kidney, muscle, pancreas, spiean, thymus and uterus.
In those exceptional cases in which one of the remainder organs receive an equivalent dose in excess of the highest dose in any of the twelve organs (with specific W, values) a If, of 0.025 should be applied to that tissue or organ and 0.025 to the rest of the remainder organs.
Information derived from those exposed iii utero at Hiroshima and Nagasaki
[6] brought to light the high radiosensitivity of the CNS during the 8—15th weeks of pregnancy. Risk of servere mental retardation during this period was estimated to be 40% Sv ‘ for high dose rate exposure. Period of pregnancy from 16 to 25 weeks has a risk of l0’7o Sv*1, whereas the risk of mental retardation does not exist for the rest of the pregnancy. Prenatal exposures are also associated with a risk of excess childhood cancer incidence. ICRP now accepts a risk estimate of 10°1 Sv*’ for childhood cancers. On the basis of the reanalysis of the mental retardation data and detailed IQ studies among the children exposed in utero, ICRP has assumed that the induction of severe mental retardation is a deterministic effect, with a threshold of about 0.1 Sv. At low doses/dose rates the effect is best described by an IQ shift of 30 points Sv*’. Human IQ distribution follows a Gaussian distribution with a sigma value of 15. As a result, for small doses, the shift of IQ by few points does not result in a significant increase in the number of mentally retarded. A summary of the risk cc›efficients for various effects under different exposure conditions are shown in Table 5.
Dose Limits
Dose limits of ICRP applies to occupational and public exposures. Dose
Table 5. Summary of estimates of probabilities of different effects
Effect
Population Exposure
exposed period/details
Probability
Fatal cancers
Genetic effects (Severe heriditary disorders including
Whole population Life time(low dose rate) 5 x 10‘’ Sv‘’ Workers Life time (high dose rate) 10 x 10‘’ Sv*’
Life time (low dose rate) 4 x 10‘° Sv‘’ Whole population Life time (high dose rate) 8 x 10*’ Sv‘’ (at equilibrium) 0—30 yrs. (low lose rate) 1 x 10*’ Sv*’
multifactorial disorders
Prenatal exposure effects several mental retardation
Workers (18—65 yrs) Fetus
(18—30 yrs) (low dose rate) 8—15 weeks
16—25 weeks
0.6 x 10*’ Sv‘’
40 x 10*2 Sv*’
10 x 10‘2 Sv*’
(high dose rate)
Reduction in IQ Childhood cancers Radon
Fatal lung cancers
Fetus 8—15 weeks 30 IQ Sv ‘ Embryo/Fetus (low dose/low dose rate) —10 x 10*’ Sv*’ Workers Life time (1—4) x 10“
WLM*
(WLM=7 msv/
month)
Detriment
General population Life time (low dose rate) 1.5 y-r Working 18—65 years 0.5 y-r
population (low dose rate)
Sv*’ Sv‘
limits do not apply to other important sources of exposure namely natural background exposure and the medical exposure for diagnostic and therapeutic reasons. The present ICRP recommendations regarding the dose limits are not aimed at maintaining the mortality rates comparable to those in many other safe industries as was done by ICRP 26. At present, the dose limits are based on the evaluation of detriment resulting from a continuous exposure over a working life time of 47 years. Limits are chosen such that the consequence of exposure are ‘just short of unacceptable’ or ‘just tolerable’. ICRP assumes a maximum risk figure of 10‘3 yr*1 mortalities in conventional industries as acceptable. Variation of the fatality rate with age among those occupationally exposed to 10, 20, 30 and 50 msv yr*’ are shown in Fig. 1(a). Among those exposed to 20 msv yr*’, maximum mortality rate marginally exceeds the value of 10*’ yr*’ when they attain the age of approximately 65 years. The various components of aggregated detriment arising from these exposures arc shown in Table 4. It can be seen that at 20 msv yr*1 the aggre- gated detriment compares with the detriment arising from an annual risk rate of 10*3. On the basis of these considerations ICRP set the annual dose limit at 20 msv for occupational workers. Table 6 also shows the detriment calculated on the basis of 1977 data. With the new tissue weighting factor
(+t). the limitation on the basis of effective dose automatically prevents any deterministic effects in all organs except the lens of the eye (which makes very little contribution to the effective dose) and skin which may be locally
Table 6. Attributes of detriment due to exposure of working population (18—64 yrs)
| Annual effective dose
(msv) |
Approximate lifetime dose (Sv) | Probability of attributable death (to) | Weighted contri- bution from non- fatal cancer
(9•) |
Weighted contri- button from hereditary effects (to) | Total detriment | Time lost due to an attributale death (yr) | Mean loss of life expectancy at age 18 yrs |
| 10 | 0.5 | 1.8 | 0.4 | 0.4 | 2.6 | 13 | 0.2 |
| 20 | 1.0 | 3.6 | 0.7 | 0.7 | 5.0 | 13 | 0.5 |
| 30 | 1.4 | 5.3 | 1.1 | 1.1 | 7.5 | 13 | 0.7 |
| 50 | 2.4 | 8.6 | 1.7 | 1.7 | 12.0 | 13 | 1. l |
| 50
(1977 data) |
2.4 | 2.9 | 1.2 | 4.J | 10.15 | 0.3—0.5 |
124 SOLN and Rno
exposed. Hence, the Commission has suggested an arinual equivalent dose limit of 150 msv to the lens of the eye and 500 msv to skin, hands and feet. In order to arrive at the dose limits to public aggregated detriment resulting from a continuous exposure (throughout the lifetime) to 1, 2, 3 and 5 msv yr°’ was assessed. Further details of time-distribution of fatal cancer risk are shown in Fig. l (b). An annual effective dose of l msv results in an aggregated
x
20
Annua risk
@ 1 ‘ 000’ ’ ’ ’
Annual risk
u- 0 J
0
l
30 50 BO 100
Age y
(a)
Fig. 1
Age y &)
detriment of 0.55%. On the basis of this risk level and the variation in natural background radiation levels, ICRP confirmed that the dose limits to the public should be I msv yr*’. The corresponding levels for the lens of the eye 15 msv yr*’, skin hands and feet 50 msv yr*1.
The basis for occupational exposure of women who are not pregnant is the same as that of men. However, in the light of new information regarding the high radiosensitivity of the human conceptus, if the woman is pregnant, the Commission has suggested a dose limit of 2 msv at the surface of the abdomen of the mother for the duration after the declaration of pregnancy to child birth, and intake of radionuclides during this period should not exceed 0.05 of ALI (Annual Limit Intake). The intent of these two limits for pregnant woman is to limit the dose to fetus to about l msv.
In order to provide some flexibility, the dose limits to both occupational and public exposures are permitted to be averaged over a period of 5 years, the new dose limits along with the explanatory notes are given in Table 7. It is the view of the Commission that the dose limits constitute a part of the system of protection with emphasis on the ALARA principle, and not a target to be achieved. If any individual exposure has crossed the limits, there need to no special restrictions to the exposure of the individual: instead, the regulatory body should look into the design and operational aspects of
protection in the installation.
These standards provide for such a high level of protection that the radiological aspect of working environment should have no influence on the administrative conditions of service of occupationally exposed persons.
Application
Table 7. Dose limits recommended by ICRP-60 (1990)
Dose limit
Effective dose
Annual equivalent
dose in lens of the eye Skin
Hands and feet
Notes:
Occupational
20 msv per year, averaged over defined period of 5 years. With no more than 50 msv in a single year
150 msv
500 msv*
500 msv
Public
1 msv in a year averaged over
5 years
15 msv
50 msv
- Forpregnant women, dose limit to the surface of abdoman of 2 msv, for the remainder of pregnancy and intake of radionuclides < 0.05
- Doselimit do not apply to medical exposures, to natural sources of radiation and under conditions resulting from accidents.
*Averaged over 1 cm2 when exposed area is small (<1 cm2) or actual area exposed.
**Averaged over areas of the skin.
REFERENCES
- Health Effects of Exposures to Low Levels of Ionising Radiations BEIR V Report. National Academy of Sciences, National Acadamy Press, Washington, DC, 1990.
- Recommendationsof ICRP. ICRP Publication 26, Annals of ICRP 1977; 1(3), Pergamon Press.
- Recommendationsof ICRP Publication 60, Annals of ICRP 1991; 21
(1—3), Pergamon Press.
- Schull, W.J., Otake, M. and Yeshimaru, H. Radiation related damages of the developing human brain in Low Dose Radiation: Biological Basis of Risk Assessment1989; 28—41 (Ed. KF Baverstock and JW Stather) Taylor and Francis, London.
- Sources, effects and Risks of Ionising Radiations. Report of UN Committee on Effects of Atomic Radiations, United Nations, NewYork,
1988.
- US(DHHS), Cancer Statistics Reviews 1973—1986. NIH Publication No. 89—2789. US Dept. of Health and Human Services PHS. NIH NCI, Bethesda, , 1989.
- BiomedicalEffects of Ionizing Radiation
K.S. Parthasarathy
Atomic Energy Regulatory Board, Bombay, India
Introduction
Irradiation at low doses and dose rates may cause several types of biomedical effects. The best documented effects are those on the alteration of the genes and chromosomes [2]. The alterations in different types of cells have been studied systematically. Consequently our knowledge of the relevant mechanisms has become very extensive. The dose-response relations are known reasonably well. The lesions that cause damage at molecular level are likely to be repairable to a considerable extent.
In its latest report the Committee on Biological Effects of Ionizing Radiations of the US National Academy of Sciences concluded that the epidemiological evidence thus far available does not contradict the following hypotheses [2]:
- Thefrequency of hereditary and cancer induction effects due to irradiation increases with low level
- Theeffects do not have a threshold. A statistical association of cancer risks with high radiation doses has been seen in a number of studies.
Persons who are “sensitized” about “the effects of radiation” always assert that there is no safe level of radiation. But it is only a prudent assumption. It is likely to remain so, in view of the difficulties to prove it conclusively.
Epidemiological Studies
The carcinogenic potential of ionizing radiation became evident from several epidemiological studies. These include exposed groups such as radium dial painters (1920), radiologists (1920), survivors of the atomic bomb explosions at Hiroshima and Nagasaki (1950), uranium miners (1950) and patients treated with radiation or radioactivity (1950).
The doses delivered in all these cases were very high. In some instances the dose rates were also high. It is difficult to demonstrate effects at low dose levels to which occupational workers are exposed. There are several reasons for this. First, the effects, if any, are very small compared with the rate at which they occur spontaneously. Secondly, in any group, there may be several confounding factors. Some of these are very powerful. The smoking habit and induction of lung cancer is one such instance. When the causes of cancer occurring among the general population were reviewed, only a maximum of one percent was attributable to radiation [32].
Ninetynine out of a hundred cases in the general population are attributable to food habits (33%), smoking (31%), viral infection (10’7»), sexuality (7’7»), industrial occupation (4′ o), etc.
Thirdly, the amount of statistical information necessary to get precise estimates of radiation risks from the radiation exposures to radiation workers is astronomical. Because of this the risks at low levels of doses are estimated on the basis of extrapolation of the risks demonstrated at high doses and dose rates and correcting for dose rates and other relevant factors. Any one who wants to do serious epidemiological studies must study the strengths and weaknesses of the methods. Joan M. Davies has reviewed the topic for the Committee on Radiation Protection and Public Health of the Organisation for Economic Cooperation and Development [10].
All epidemiological studies initially reported through news papers and television must be examined more objectively. This is because some scientists try to circumvent the traditional scientific peer review and publication process by exploiting newspapers and periodicals. This is a lamentable trend. In all studies we must look for the statistical design, the choice of control group and the efforts to reduce the influence of confounding factors.
Coggle noted that the major approach to the assessment of radiation risk has been through detailed epidemiology of about 2 million adults and children in over 50 population groups (7J. Most of the cases where the doses were much less than one Gray it was statistically difficult to detect any excess cancers. But in over 300,000 highly exposed individuals hundreds of “excess cancers” were recorded. But the interest in radiological protection is in the case of low levels of annual doses of a few centigray typical in occupational exposures.
Dr. Rosalyn S. Yalow cites [42) an interesting report. This report to the Congress of the United States indicated that upto 1981 US government alone had spent $2000 million on research on the health effects of radiation. The report afrmed that there was as yet no way to determine precisely the cancer risks of low doses of ionizing radiation and it was unlikely that this question wouid be resolved soon. A decade later the conclusion remains much the same. But the efforts should continue.
Radium Dial Painters
During 1920s several thousand people, mostly teenagers, were employed in about 50 dial painting studios. The job was to paint the dials of watches and even crucifixes with a mixture of radium and zinc sulphide. These radioluminous compounds were widely used in several industries.
The tips of the brushes with which they used to paint the dials must remain fine to execute the work accurately. The girls developed the habit of rolling the brush tips between their lips. Some yellow liquid laced with radium got into their mouths. Their clothes, hands and the studios were heavily contaminated. Many of the girls even painted their teeth with luminous
compound prior to an important “date” so that they would glow romantically when the lights were low [17]!
Several thousand people acquired measurable quantities of radium in their bodies. A group of dial painters were examined over the past several decades. As of 1971, 54 cases of bone sarcomas and 20 head sinus carcinomas had been identified in US subjects of known dosage [11]. It is also felt that radiation might have helped the induction of a few brain tumours and leukaemias among the dial painter. Since the natural occurrence of bone sarcomas is known to be very low, the carcinogenic potential of radium is fairly conclusive.
Leukaemia in US Radiologists
Dr. Shields Warren examined the mortality data of radiologists from 1930 to 1960 from the records of the American Medical Association. During this period, there were no radiologists certified below the age of 25 [34]. So the control group was US males aged 25 and above. Right from 1930, the average age at death of radiologists was five years less than that of US adult males aged 25 and above. This difference was maintained upto about 1950, when marked improvement began with no life shortening apparent from 1960 onwards. Dr. Warren suggested that this might be due to improved protection.
For the same period, when proportionate mortality due to specific causes was compared, it was found that deaths from leukaemia were six times higher among radiologists. Deaths from other cancers were about the same.
Cancer following Medical Irradiation
The study of the various groups of patients irradiated for medical reasons demonstrated the carcinogenic potential of radiation (Table 1). Boice reviewed in detail most of the publications and made the following general observations [4].
- Asingle exposure is sufficient to elevate cancer incidence many
years later.
- Radiationinduced cancer cannot be distinguished from natural
- All cancersappear to be increased after irradiation with the exception of chronic lymphatic leukaemia, cervical cancer and a few
- Breast,thyroid and bone marrow appear to be especially
- Leukaemiais the most prominent radiogenic
- Solidtumours have a minimum latent period of about 10
- Ageat exposure is perhaps the most important host
- Thepercentage increase per rad is not the same for all cancers. Also some cancers of high natural incidence, g. colon have low relative risks. Also some cancers of low natural incidence such as thyroid have high relative risks.
- Doseeffect relations are often linear but curvilinearity is also
Table 1 Medical irradiation for benlgn/malignant dlseases
| Disease | No. of patients | Average dose (gray) | No. of cancers | No. expected |
| Ankylosing | 14,558 | 3.7 | 52 leukaemia | 5.5 |
| spondylitis | cases | |||
| [8, 36)
Postpartum |
571 |
2.47 |
36 breast cancers |
* |
| mastitis [331 Thymus |
2,872 |
1.19 |
24 thyroid cancers |
0.29 |
| hypertrophy [15] | ||||
| Tinea | 10,842 | 0.09 | 23 thyroid cancers | 5 |
| capitis [30] | 6 brain cancers | l | ||
| Benign head and neck | 2,189 | 7.9 | 182 thyroid cases Low | |
| disease [12, 33] | ||||
| Radiation | 2,068 | 1.34 | 7 leukaemia cases 2.6 | |
castration [37]
Pneumothorax fluoroscopy [5, 6]
Pelvimetry [3]
1,047
1.5 41 breast cases 23.5
0.01-0. l Gy
50% increase
in leukaemia
Bone TB and anky- losing spondylitis wilh Ra-224 [22)
Polycythemia vera [23]
897 4.41
1,222
54 bone cancers Low
Excess
leukaemia
Thorotrast [9]
3,921
0.25 245 liver cancers
Gy/year to liver
*Number observed: 32 among 993 not irradiated.
**Findings disputed. Selection factor important?
Lung Cancer in Underground Miners
The metal miners in Schneeburg in the middle of the 16th century were known to die of a mysterious chest disease. Agricola a physician who studied medicine in Italy observed the following occupational conditions “the dust has corrosive qualities, it eats away the lungs and implants consumption in the body. Women are found who have married seven husbands all of them, this terrible consumption has carried off to a premature death”. Paracelsus, a contemporary physician attributed this “mountain sickness” to the inhalation of metallic vapours “which setfle on their lung in different ways. Agricola recommended the wearing of loose protective veils of fine netting which the miners women folk were trained to make”. He also understood the importance of ventilation in the mines.
Much later the mysterious disease was shown to be due to the airborne decay products of radon. Pirchan, Chief Physician at the Jachymove Radium Institute studied the problem systematically in 1926—1929. The minen had told him that the discovery of a rich uranium vein was always followed
some years later by a large number of deaths among the men concerned. Pirchan could prove the deadly nature of radon rather unequivocally.
Several epidemiological studies demonstrated the carcinogenic potential of elevated levels of radon. The results are available from US mines, Czech mines and Swedish iron mines.
The risk over 40 years at one rem/year appears to fall between l and
4 x 10 4/peryear affect the expression of disease.
Studies on Survivors of Atomic Bombs
Detailed epidemiological study on the survivors of the Atomic Bomb explosion at Hiroshima and Nagasaki is very illustrative. The study involved 75,991 persons. They were followed up for a period of 35 years (1950—1985) {28). The latest dosimetry is more accurate. It corrected for body shielding.
Also the latter study was extended for a longer follow up period.
Cause of deaths No. of
deaths
Leukaemia 202
All cancers 5734
The average dose to the group is 0.13 gray.
The final figure
Leukaemia
All cancers except leukaemia
No. attributed to radiation
79
262
80 deaths per 10‘ person gray
265 deaths per 10‘ person gray
Biological Effects of Prenatal Irradiation
The review of this topic has been published in 1986 by the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) in its report titled “Genetic and Somatic Effects of Ionising Radiation”.
Many new experiments on animals have confirmed earlier observations that embryos during the pre-implantation period get killed. Irradiation during organogenesis causes malformations. In man, unlike in other animals, most malformations occur in the central nervous system.
Studies on 1600 children who were irradiated in vivo during the Atomic Bomb explosions in Japan revealed that 30 of them suffered clinically severe mental retardation. Between 0 and 7 weeks post conception mental development was not affected. Between 8 and 15 weeks the sensitivity was maximum of the order of 0.4 Gy”. This is possibly because neuronal proliferation and cell migration in the cortex is most active during the period. From 16 weeks to 25 weeks the incidence of mental retardation is four times lower. Mental retardation was found to depend on dose. There was no detectable threshold, but a thrcshold of 100 mGy cannot be niled out.
The committee also reviewed information on the induction of tumours in man and animals irradiated prenatally. The evidence is conflicting. Two large retrospective studies in UK and in the USA on children exposed prenatally for medical reasons indicated an increase of 509c in the incidences of leukaemia and other tumours (3). But children who survived prenatal irradiation in Hiroshima and Nagasaki did not show any increase in cancer incidence. If anything, experiments with animals showed lower susceptibility
than after birth. As a prudent approach the UNSCEAR suggested a risk factor of 2 x 10*2 Sv ‘. This means that if 10,000 children are exposed in vivo to 10 msv of effective dose 2 extra cases of childhood cancer are likely.
The most recent recommendation of the International Commission on Radiological Protection on the dose limit for pregnant radiation workers was clearly influenced by the observed correlation between irradiation in vivo and mental retardation in the children of the survivors of the atomic explosions at Hiroshima and Nagasaki.
Once the pregnancy has been declared the conceptus must be protected by applying a supplementary equivalent dose limit to the surface of the woman’s abdomen (lower trunk) of 2 msv for the remainder of the pregnancy and by limiting intake of radionuclides to about 1/20th of the Annual Limit of Intake [16].
Studies on Workers in the Nuclear Industry
Workers at Hanford Plants in USA
A report by Mancuso, Stewart and Kneale has analysed the radiation exposure data of radiation workers at Hanford and claimed that sensitivity to the cancer induction effects of radiation is at a low ebb between 25 and 45 years of age [20]. This report claimed that at younger and older ages there is a cancer hazard associated with low level radiation which causes bone marrow cancers more than other neoplasms and cancers of the lung and pancreas more than other solid tumours.
Hanford Plants in USA is one of the largest atomic plants and most of the staff here were associated with the production of radioactive substances. The record of these workers contain sex, date of birth, date of hire, social security number, dates of entering and leaving specified occupations, external and internal radiation doses and date and cause of death. Since they wore radiation dosimeters mandatorily, their dose data were monitored periodically for compliance with appropriate dose limit which was 50 millisievert/year. Those workers who were involved in high risk occupations or were exposed in accidents or leaks were examined regularly for internal contamination and records for both internal and external exposures were readily available for the workers.
In 1977 this study was the only one of its kind available in literature and
has attracted critical appraisal.
Hanford works were commissioned in 1944. Mancuso and his co-workers
examined the records of all workers who have died since then. The records included the accumulated doses and causes of deaths. Of the 24939 males who worked at Hanford sometime between 1944 and 1971; 3733 are known to have died. Death certificates were available for 3520. From an unspecified number of female employees 412 death certificates were traceable. The male workers (3520) were classified into 2184 exposed people (life time dose greater than 0.01 rad) and rest as non-exposed. Their collective dose was 3756 person rads and mean working life time dose of 1.72 rads. Of the 670 cancer deaths 442 were in the exposed group. The number of females who died and who were exposed was 123 receiving a collective dose of 108 person rads or a mean working life time dose of 0.89 rad. 127 females died due to cancer, of whom 38 have been exposed. Most of the analysis of Mancuso et a1 was on the data of male workers.
In a comprehensive review, Reissland stated that the overall assessment is a rejection of the validity of the majority of the claims from the study [29]. He concluded that the only fatal malignancies at Hanford that may be associated with radiation are cancers of the pancreas and multiple myeloma. Since the possibility of the effects being due to other cancinogens was not eliminated in the study, further investigations are necessary.
Reissland’s review pertained to doses, ascertainment of the causes of death, methodology and interpretations. He highlighted the criticism by
- E.Land that any individual of 50 years receives 5 rad from background radiation (at a rate of nearly 0. l rad/year), a dose similar to the highest dose received by the workers. The confounding effect of background radiation and other non-occupational exposures such as medical x-rays must be kept in mind while drawing any firm conclusion from occupational doses smaller than the random variations in the total radiation dose to which the study population is exposed.
The choice of mean cumulative dose while the dose distribution of workers are highly skewed is also not appropriate. Yet another criticism is that the doubling dose estimated for lung cancer takes no account of smoking habits, the most dominant cause of lung cancer in any population. The deaths of workers were identified through the system of social security numbers. But this becomes operative only if a death benefit claim is made.
Six percent of deaths were missed because no claim was filed.
Reissland also stated that Mancuso report classified the expected deaths from various causes according to the 7th revision of the International classification of diseases while the observed deaths are classified by the 8th revision. For most cases this procedure makes no difference but it is important for leukaemias where the study differs most strikingly from other work.
The proportional mortality study basically employed by Mancuso et a1 has a clear disadvantage. Decrease in one cause of death causes an apparent increase in another.
Mancuso study was also flawed as their comparison was based on American
Biomedical Effects of Ionizing Radiation 133
death rates in 1960 which preceded most of the Hanford deaths. Cancer of the lung, pancreas and myeloma increased significantly during the period of study. The conclusions of Mancuso study differed substantially when analysis was done after adjusting for age and for years of death.
Mancuso report got wide media publicity. Dozens of scientific reports contesting the claims were published shortly in referred journals. But they did not get any media coverage.
Other Studies on Nuclear Workers
As on 1987, eight follow up studies covering 120,000 workers in nuclear industry have been published. In all the work forces, overall mortality rates were less than their respective natural rates, possibly because of the relatively high social classes and the initial selection of healthy workers. There were no major differences in mortality between workers where jobs did or did not bring them into contact with radiation [41].
Workers at Hanford in the USA and Sellafield, UK found that the deaths due to multiple myelomas were significantly related to cumulative radiation exposure. Despite the large number of workers studied, the data are too sparse to estimate cancer risks precisely.
Cancer Mortality Study of Nuclear Workers/Community in India
Nambi, Mayya, Rao and Soman have analysed the cancer deaths in the centralized health services of Department of Atomic Energy among its employees and families stationed in Bombay during 1975-1987 [25].
fn the data base of 702,000 person years the number of cancer deaths observed were 154. The analysis included employees and their families individually for important groups of cancer sites such as respiratory organs, breast; genito-urinary organs and lymphatic and haemotopoietic systems. Healthy worker effect is seen. The data base is very small. Of the 154 cancer deaths 50 occurred among employees of which only 9 were radiation workers. Their life time cumulative doses ranged between 0.10 and 95.75 msv.
The data are too small to give statistically meaningful association between radiation exposure and increased cancer risk.
Nambi and his coworkers examined the cancer mortality risks for workers at the nuclear facilities at Tarapur and for their family members [26]. There were 11 deaths from among the employees and this figure was too small to permit any trend analysis with radiation exposure. A much larger data base of person-years at risk would be required to reach definite conclusions. The authors also observed that the combined cancer risks for employees and families were similar to risks experienced by individuals in Bombay.
Major Conclusions [41]:
- Nomajor health hazard associated with working in nuclear industry is identified.
If the radiation to which workers are exposed is carcinogenic, the effect is so small and cannot be distinguished from normal incidence of cancer.
- Consistentincreased mortality due to multiple myeloma was noted in
the most highly exposed workers. In each work force there were probably no more than two or three extra deaths from this condition in the entire force.
- The finding looks reassuring. But the data are not enough tostate that the ICRP risk estimates are The results so far are consistent with there being no increased cancer risk at all and at the same time with a risk ten to fifteen times the ICRP figures. It is essential to pool results from more studies to reduce the uncertainty.
UK Study of National Registry of Radiation Workers
The National Radiological Protection Board has studied the records kept since 1976 in its National Registry of Radiation Workers. The histories of 95,000 individuals were analysed. Of these 6600 had died by the end of the study period. Fifty three of these deaths were due to leukaemia [18].
The death rates from most type of cancers were lower among the nuclear workers than in the general population. This is the healthy worker effect seen in similar epidemiological studies. The study showed statistical significance between all cancer deaths and radiation dose. According to NRPB there is no justification to amend the advice on occupational dose
limits yet. If the UK and USA stu-dies are pooled together, the overall
results fit ICRP estimates of risk more closely.
The UK study also reported more cases at one site of thyroid cancers than expected.. This was concluded to be due to chance.
Health Study of US Shipyard Workers
A study of Portsmouth Naval Shipyard nuclear workers by Najarian and Colton reported excesses of death from all cancers combined (especially leukaemia) (24]. Davies has detailed the steps in this study [10].
A team of persons (with unspecified qualifications) scanned all death certificates for the relevant local areas during 1959—1977. From over 100,000 certificates 1722 with recorded occupations indicating naval shipyard work were copied. Using telephone numbers 592 of the next of kin were contacted. Based on the strength of the telephone responses, they were classified as nuclear or non-nuclear workers. The only query was whether the deceased had worked with radiation or had wom a radiation badge.
Later a full study covering 24545 workers at the shipyard found normal fatality from leukemia in the whole group and in those exposed to radiation. Overall cancer deaths were slightly lower than normal as is expected in a healthy industrial population.
Results of a more systematic and extensive study in shipyard workers
indicated that there was no effect associated with radiation workers. The
Biomedical Effects of lonizing Rad.iation 135
number of radiation workers included were 107976 frum eight shipyards which employed 692612 workers. A brief summary of the study appeared in the December 1991 issue of “Nuclear Energy”
Broadly the following results were observed:
- No significantassociation of death with radiation exposure was
- Nuclearworkers showed the usual healthy worker
- Thereis no significant deviation for all cancers or for
- Therewas no indication of excess deaths from prostatic cancer among the radiation workers, an effect observed in UK
The extensive study of shipyard workers was the largest of the type and was carried out by an independent research team led by an international expert and was reviewed by a distinguished technical advisory panel.
Cancer in Populations Living Near Nuclear Facilities
Leukaemia clusters in Sellafield
In 1983 the Yorkshire Television telecast a programme titled “Windscale, the Nuclear Laundry” by a journalist, James Cutler. The programme claimed a ten-fold increase in leukaemia near Sellafield nuclear reprocessing plant. Five leukaemia cases under the age ten were found over a 25 year period in Seascale, the village near to Sellafield where only one case should be expected in 60 years. Within 48 hours the British Government authorised a special investigation under Sir Douglas Black, a former President of Royal College of Physicians of London.
The Black Report accepted that the observed number of leukaemias were highly unusual. The report also noted that the amount of radioactivity released from Sellafield was not high enough to account for the number of extra cancers observed. The report recommended further research into the association between radiation and leukaemia. Based on the recommendations of Black ‘ Report, the Department of Health and Social Security set up the Committee on Medical Aspects of Radiation in the Environment (COMARE).
Leukaemia due to parental irradiation
The study by Martin Gardner and colleagues of the Medical Research Council, Environmental Epidemiology Unit at Southampton University was a follow up of the Douglas Black Report. This study suggested that an accumulated dose of 100 msv or more to the workers prior to conception is associated with a six to eight fold increase in the risk of leukaemia in the offspring. A similar risk appears to be associated with much smaller exposures in the range of 10 to 30 msv if the dose is sustained in the six month prior to conception [21]. Gardner’s study also indicated that the workers in iron and steel, chemicals and farming industry also ran raised risks of fathering leukaemie children.
Gardner looked at all leukaemia and lymphoma cases in people under 25
born in West Cumbria (where Sellafield is located) and diagnosed between 1950 and 1985. There were 52 cases of leukaemia and 22 non-Hodgkins lymphoma (23 cases of Hodgkins disease were also studied but no links to Sellafield were found).
In ten of the cases (9 leukaemia and one lymphoma) the father had worked at Sellafield. Gardner’s paper depends mainly on these cases. The four highest exposures of fathers of leukaemia cases were 102 msv (over 7 years) 162 msv (6 years), 188 msv (7 years) and 370 msv (10 years). Five of the leukaemia cases in the study were in Seascale, a neighbouring village of Sellafield where the incidence of the disease is about ten times the expected level.
Is there any mechanism to explain the effect? Gardner’s team suggests that it is due to the effect of radiation exposure on sperm cells producing a leukaemogenic mutation. May be external dose is a surrogate to some other factor, internal dose or chemical exposure for instance.
In the Japanese study of the survivors of Hiroshima and Nagasaki the offsprings of 7387 men exposed to 492 msv on an average did not show any sign of excess of leukaemia. Children of men who received significant abdominal doses of x-rays also did not suffer from excess leukaemia. These doses were instantaneous, not chronic doses as with the Sellafield work force. Among the notable responses to Gardner’s paper was one from Dr. N.K. Notani, former Director of Biomedical Group, Bhabha Atomic Research Centre. He also highlighted the fact that “such genetic meltdown” is not observed in the survivors of Hiroshima and Nagasaki. He contended that Gardner’s explanation does not seem to be congruent with several biological and radio-biological principles [27].
Radioactive materials do not accumulate in testicular tissue. Sellafield workers did not suffer from excess of testicular cancer. It is well known that plutonium which is known to be an important pollutant from a reprocessing plant tends to accumulate in bone and liver. Only about 0.035 percent of ingested plutonium concentrates in the testicles.
Department of Health in UK sought the views of Britain’s Committee on the Medical Aspects of Radiation in the Environment (COMARE) on the conclusions of Gardner’s study. The Committee found the evidence for a link between childhood leukaemia near Sellafield and relatively high levels of radiation received by their fathers to be inconclusive (13). COMARE’s interim advice to the Government was the kind of study carried out by Gardner cannot prove evidence of a causal relationship. COMARE confirmed that the study was scientifically valid and agreed with the broad thrust of its conclusions [13J. Workers may have been exposed to other agents as well. The observed link may be due to a combination of factors. Based on the advice of COMARE the Government decided that there can be no fresh guidance for radiation workers till more conclusive research has been done [13].
Studies Carried Out in the USA
An ad hoc Committee for the study of cancer in populations living near nuclear facilities examined mortality from 16 types of cancers in 107 counties which host or adjoin 62 major US nuclear facilities which include 52 nuclear power stations. The Committee was constituted by the US National Cancer Institute. The committee has published a report titled “Cancer in populations living near nuclear facilities” in 1990 (NIH Publication 90-874).
From the data at hand there was no convincing evidence of any increased risks of death from any of the cancers due to living near nuclear facilities. The NCI methodology was subjected to rigorous peer review. The study had some limitations.
The NCI report summarises the conclusions from 19 studies carried out internationally. Some of them showed statistically significant effects while others did not.
Cancer near nuclear installations in England and Wales
David Forman and his colleagues have summarised the report entitled “Cancer incidence and mortality in the vicinity of nuclear installations in England and Wales 1959—80” [14].
All pre-1974 Local Authority Areas with at least one-third of their population living within 10 miles of 15 nuclear installations were included in the study. Twenty-five specific and combined cancer sites for three age groups 0—24, 25—74 and 75+ years were examined.
The report concluded that there has been no general increase in cancer mortality near nuclear installations in England and Wales during the period 1959—80. Leukaemia in young people may be an exception [14]. The reason for this remains unclear.
Radioactive discharges into the environment from nuclear installations are shown to be too small to account for the observed excess cancer. One theory that is often put forth is that the excess leukaemia are caused by viruses. Some authors have noted that excess of leukaemia may be ex{iected when isolated communities come together into areas of high population densities. Such excess has been observed in the new towns of 1940s.
Childhood leukaemia around nuclear facilities in Canada
The Atomic Energy Control Board Canada initiated study after repons of the UK noted an increase in the number of leukaemia cases in children living near certain nuclear facilities. AECB thought that exploratory research in the vicinity of Canadian nuclear facilities is prudent, though the leukaemia clusters in Sellafield could not be explained on the basis of low radiation dose to public, attributable to the plant [1].
The facilities selected for study were all in Ontario and have had a relatively long period of operation. They included the nuclear power stations at Pickering and Bruce (Douglas Point), the uranium mines and mills at Elliot Lake, the uranium refining facility at Port Hope and the nuclear
138 PARTTIASARATHY
research laboratories at Chalk River along with the small nuclear power plant near Rolphton.
Dr. E.A. Clarke and J. McLaughlin of the Ontario Cancer Treatment and Research Foundation, Toronto and Dr. T.W. Anderson of the University of British Columbia conducted the studies. An independent review panel of experts scrutinized the progress and the final report of the study.
Ontario Cancer Registry, one of the largest population based cancer registries in the World provided the information on leukaemia. Records of children upto 14 years of age were examined. The study identified children who died from leukaemia between 1950 and 1987 and children who were diagnoscd with the disease between 1964 and 1986. The number of observed cases around each facility was compared to the number expected in a population of equivalent size based on the Ontario average. Childhood leukaemia cases in the vicinity of the Pickering Nuclear generating plant both before and after the plant began operation in 1971 were noted.
The study concluded that while the rate of occurrence of childhood leukaemia around nuclear facilities may be higher or lower than the provincial average, there is no statistical evidence that the difference is due to anything but the natural variation in the incidence of the disease. The rate of occurrence of childhood leukaemia around the Pickering nuclear power station was slightly greater than the Ontario average both before and after the plant opened but this too could be due to natural variations.
Defective child births in the villages around
Rajasthan Atomic Power Station
The activists of some voluntary organisations have claimed that the defective births in some of the villages around the Rajasthan Atomic Power Station has increased after the commissioning of the reactors. The initial reports which appeared in all newspapers stated that i team which visited some of the villages found that children had a “weak nervous system”, and “tumors on bones”, while older people also developed similar symptoms. There were claims of abnormalities after birth and stomach upset; reduction of cattle fertility; paralysis and abortions among goats. The report also claimed the incidence of cancer among workers and high abortion rates among their women folk. No scientific study on the topic was available for scrutiny.
These reports were followed by the release of a clandestinely made film titled “The Price of Power” in England Channel IV TV during the first week of April 1991. The nlm dcpicted interviews with the “affected” people in the villages. The visuals were telling. This extract along with an interview in which I participated as Government spokesman was released by the Eyewitness HTV Video Magazine Programme in September 1991. The news story got wide publicity. The issues were raised in Parliament several times. I have contested the claim on the ground that the radiation dose, if any, to the villagers are too small to be measured.
The Environmental Survey Laboratory has been operating an extensive radiation monitoring programme ever since the reactors started operation. Thousands of samples of air, water, food, etc., were collected periodically from the area. The radiation increase is too small to be measured. The estimated value is of the order of 10 micro sievert. It is inconceivable that such a tiny amount of radiation dose can cause the alleged effects. The variation in the natural background radiation level itself at any location could be shown to be several tens to hundreds of microsieverts.
Studies carried out in the areas of high natural background radiation
There are several regions in the world where the gamma radiation levels due to natural background exceed the normal range. These regions are in Brazil, India, China, Italy, France, Iran, Madagascar and Nigeria [2].
Guarapari,’ a village in Brazil has nearly 12,000 inhabitants. The annual average dose to an inhabitant in this area is about 6.4 msv which is roughly six times the global average background radiation level excluding the dose due to radon progeny in the lung. An increase in chromosome aberration was reported in a study of 200 individuals from this area in comparison with a control group from a similar village.
Review of studies at high background radiation areas in Kerala
Because of the presence of monazite in the soil (thorium concentration 8 to 10.5′ o by weight) the average natural background radiation to about 70,000 people in the high background radiation area in Kerala is about
3.8 mGy/year. This value has been obtained in a study carried out by Bhabha Atomic Research Centre, by issuing 20% of the households on the strip with thermoluminescent dosimeters for two months.
The highest individual dose recorded was 22.5 mGy/year. The high background radiation areas in Kerala is of great interest since thousands of people have received for many gcnerations a dose level which far exceeds the ICRP recommended levels of one mSv/year for members of the public from human activities. Rose has published a detailed review of the studies in 1982 [31] and concluded as follows:
- Severalstudies of genetic traits and one of thyroid growths at Kerala have failed to produce any claims of a causal association with the high external radiation doses received in the area, with the exception of Down’s
- There has been no definitive study on the frequency of Down’s syndrome associatedwith irradiation. Other epidemiological studies of subjects exposed to medical X-rays, survivors of the A-bombs, and inhabitants of areas with high natural radioactivity do not produce a consistent pattern of
- The survey that revealed anexcess of Down’s syndrome cases was not conclusive and several explanations of the excess, e.g. differences in maternal age distribution, or parental care, could explain the finding
on the basis of the information published. It does not, therefore, provide good evidence of irradiation causing a genetic change in humans.
- No reliablestudy has revealed any adverse medical effects at
Genetic Effects
Human epidemiological data on genetic effects of ionising radiation are scanty. The children of the survivor of the irradiated population at Hiroshima and Nagasaki were studied extensively. The overall conclusion appears to suggest that humans may be somewhat less sensitive to radiation than mice (2].
Recent analysis included examining of 13,052 children of proximally exposed parents and 10,609 cNldren of control parents for rare electrophoretic variants of 30 blood proteins. Three mutations were detected in each group in 725,587 and 539,170 equivalent locus tests respectively. The mutation rate for control group is 0.6 per 100,000 per locus as against 0.4 per 100,000 per locus for the exposed group [40]. In a subset of 4983 children of exposed parents and 5026 control children who were studied for deficiency variants of nine erythrocyte enzymes one mutant was observed in the first group and none in the second [40].
Conclusions
The review of health effects of exposure to low levels of ionising radiation reveals that exposure at low levels is unlikely to cause unacceptable risks to radiation workers. The risk to members of the public will be significantly lower as the doses to which they are likely to be exposed are several orders lower. Recent studies show that it is prudent to limit the exposure to pregnant women because the foetal brain is likely to be very sensitive.
REFERENCES
- AECBInformation Bulletin, 91-2(Rev.1), 1991; June
- Beir, Health Effects of Exposure to Low Levels of Ionizing Radiation. Committee on the B iolog ical Effects of loni zing Radiation, Board of
Radiation Research Commission on Life Sciences, National Research Council, USA, 1990.
- Bithell,F. and Stewart, A.M. Pre-natal irradiation and childhood malignancy: a review of British data from the Oxford Survey. Br. J. Cancer, 1975; 31:
271—287.
- Boice,D. Jr. Cancer Following Medical Irradiation. Cancer 1981; 47: No. 5,
1081—1090.
- Boice, J.D. Jr. and Monson, R.R. Breast Cancer in Women after Repeated Fluoroscopic Examinations of the Chest. J. Natl. Cancer Inst. 1977; 59: 823-
- Boice, J.D. Jr., Advances inMedical Oncology, Research and Education Vol. 1, Carcinogenesis Ed Margison P Pergamon Press, 1979; 147—156.
Biomedical Effects of lonizing Radiation 141
- Coggle, J.E. Medical effects of low doses of ionizing radiation, Radiation ProtectionDosimetry 1990; 30: 5—12.
- CourtBrown, M. and Doll, R. Mortality from cancer and other causes after radiotherapy for ankylosing spondylitiis, Brit. Med. J. 1965; 2: 1327—1332.
- DaSilva, International Meeting on the toxicity of thorotrast and other alpha- emitting heavy elements, Environ. Res. 1979; l8: I—255.
- Davis,Joan ‘Epidemiological Studies of Groups with occupational Exposure to Radiation—A Review of the Committee on Radiation Protection and Public Health of the OECD Nuclear Energy Agency, 1985.
i 1. Evans, R.D. Health Physics 1974; 27: 497—510.
- Favas, M.J.et Thyroid cancer occurring as a late consequence of head-and- neck irradiation, Evaluation of 1056 patients, New Engl. J. Med. 1976; 294: 1019—25.
- FishlockDavid, Nuclear Europe World Scan May/June, 1990; 35.
- Forman, Paula Cook-Mozaffari, Darby Sarah, Davey Gwyneth, Stratton Irene, Doll Richard and Pike Malcolm, Cancer near nuclear installations, mature October 1987; 379: 499—505.
- Hempelmann, L.H. Hall, W.J. Philips, M., Cooper, R.A. and Annes, W.R. Neoplasmsin persons treated with x-rays in infancy: fourth survey in 20 years,
- Natl.Cancer Inst. 1975; 55: 519—530.
- ICRPpublication 60. 1990 Recommendations of the International Commission on Radiological Protection 1990.
- Jack Schubert andRalph Lapp. “Radiation: What It Is and How It Affects You”, William Heinemann Ltd., 1957.
- Kendall,M., Muirhead, C.R., MacGibbon, BJ-l., et al. Mortality and occupational exposure to radiation: First analysis of the National Registry for radiation workers, NRPB-R251, London, 1991.
- Luxin Wei, Zha Yongru, Tao Zufan, et al. Epidemiological investigation of radiologicaleffects in high background areas of Yangjiang, China, Radiat. Res. 1990; 31: 119—136.
- Mancuso, T.,Stewart, A. and Kneale G., Radiation Exposures of Hanford Workers Dying from Cancer and Other Causes, Health Physics 1977; 33: 369—385.
- Gardner,, Snee, M.P., Hall, A.J., Poweli, C.A., Downes S. and Terrell J.D.
(1) Results of case-control study of leukaemia and lymphoma among young people near Sellafield nuclear plant in West Cumbria, (2) Methods and basic data of case-control study of leukaemia and lymphoma among young people near Sellafield nuclear plant in West Cumbria, British Medical Journal 1990; 300: 423—434.
- Mays,W. and Spiess, H. “Biological Effects of Ra-224, Benefit and Risks of therapeutic application”. Eds. Muller, W.A., Ebert, H.G. 1978.
- Modan, and Lilienfeld, A.M., Polycythemia vera and leukaemia—the role of radiation treatment. A study of 1222 patients. Medicine 1965; 44: 305—344.
Najarian, T. and Colton, T. Mortality from leukaemia and cancer in shipyard nuclear workers, Lancet 1978; 1: 1018—1020.
- Nambi, K.S.V. A cancer mortality study in Bombay-based atomic energy community,Indian Journal of Cancer 1991; 28: 61—69.
- Nambi, S.V., Mayya, Y.S., Rao, D.D. and Soman, S.D. A study on cancer mortalityin Tarapur—based atomic energy community, Archives of Environmental Health 1992; 47: 155—157.
- Notani,K. British Med. Journal 1990; 300: 877—878.
- Preston,L. and Pierce, D.A. The effect of change in dosimetry on cancer mortality risk estimates in the atomic bomb survivors, TR 9-87 RERF 1987.
- Reissland,A. An Assessment of Mancuso Study-—National Radiological Protection Board, NRPB—R 79, 1978.
- Ron, E.and Modan, Benign and malignant thyroid neoplasms after childhood irradiation for tinea capitis, J. Natl. Cancer Inst. 1980; 65: 7—11.
- Rose,S.R. Review of health effects at Kerala, Nuclear Energy 1982; 21:
399-408.
- Rosenbaum,H. Can you prevent cancer? C.V. Mosby & Co., 1983.
- Schneider,B. et al. Incidence, prevalence and characteristics of radiation induced thyroid tumors, Am. J. Med. 1978; 64: 243—252.
- Shields Health Physics 1966; 12: 1737.
- Shore,E., Hempelmann, l..H., Kowaluk, E. et al. Breast neoplasms in women treated with X rays for acute post partum mastistis, J. Natl Cancer Inst. 1977; 59: 813—812.
- Smith,G. and Doll, R. “Late Biological Effects of Ionizing Radiation” IAEA, 1978; Vol. 1: 205—18.
- Smith, P.G.and Doll, R. Late effects of X-irradiation in patients treated for metropathia haemorrhagica, J. Radiol. 1976; 49: 224—232.
- Stalker,W. UK National Radiological Protection Board Bulletin, July 1987.
- UNSCEAR.Ionizing Radiation: Sources and Biological Effects, United Nations Scientific Committee on the Effects of Atomic Radiation, Repon to the General Assembly, with annexes, 1982.
- Sources, Effects and Risks of Ionizing Radiation, United Nations Scientific Committee on the Effects of Atomic Radiation, Report to the General Assembly, with annexes, 1988.
- ValerieBcral, et “Radiation and Health: the Biological Effects of Low Level Exposure to Ionization Radiation”, Eds. Rohl Russell Jones and Richard South Wood, John Wiley & Sons, 1987.
- Yalow Rosalyn, Radiation and Society, Interdisciplinary Science Reviews (The Institute of Metals) 1991; 16: 351—356.
15. Studies on Human Populations Living in Natural High Background Radiation
Areas of Kerala: A review
K.V. Aravindan and S.K. Mahajan
Molecular Biology & Agriculture Division Bhabha Atomic Research Centre, Bombay, India
Every living organism on this planet is exposed to low levels of background radiation. The assessment of radiation exposure to humans from natural sources is of particular importance because this contributes maximum to the collective dose of the world population [5]. The natural radiation levels are also used for comparison when judging the signircance of exposures from man-made sources. At some places, particularly those with radioactive rocks or soils, the background radiation levels are many times higher than average.
Although the health effects of radiation exposures from occupational sources had received attention earlier, it was not until the 1950s, when the atmospheric weapon tests had raised the level of environmental radioactivity, that the long term effects of low level radiation doses became a matter of major concern. The United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) was created and the World Health Organization (WHO) appointed an expert committee to provide advice concerning radiation and human health. In its first report (WHO, 1959) the committee identified several areas of natural high radiation where studies of the exposed population might provide information concerning the effects of chronic low level exposure. It was pointed out that there would be difficulties in conducting a study of the magnitude necessary to detect the small differences expected between the exposed and control populations.
Natural high background radiation areas (HBRA) include areas having higher than normal levels of radioactivity in the soil, e.g. Morro do Ferro, Guarapari and Meaipe in Brazil; Thekkumbhagam—Purakkadu strip of Kerala Coast in India; Yangjiang province in China and Ramsar in Iran. Several hot springs have also been found to contain higher levels of natural radioactivity and some of these, e.g., Badgastein in Austria and Tuwa in India have been used as spas. The other sources of elevated natural radiation ex¡›osure are radon and its decay products, especially in cold countries where people have to live in poorly ventilated closed houses. Sizable populations living in the states of Illinois and Iowa in the USA are also exposed to high levels of radium-226 in drinking water.
During the preparation for the first International Symposium on Areas of
144 ARAVINDAN and MAHAJAN
High Natural Radioactivity (Brazil, June 16—20, 1975) a general consensus on the working definition of HBRA was reached. Accordingly, “HBRAs” are those where at least one of the following conditions exist:
- exposurerate from external sources over extended areas is greater than 2 mSv/y.
- long lived alphaactivity ingested with the local diet (including water) is greater than 5 pCi/day.
- radon-222concentration in potable water is greater than 5000 pCi/l.
- radon-220and radon-222 concentration of the atmosphere is greater than 1 pCi/1.
Health effects in residents of HBRA have been reviewed by Hanson and Komarov (16]. A review of health studies pertaining to the HBRA in Kerala has been brought out by Rose [29). The findings of various studies reported to date from the Kerala HBRA, along with the current status of the ongoing investigations by BARC are presented below.
The imponance as well as the necessity to investigate the HBRA of Kerala Coast in India was emphasized by the WHO as early as 1959 [35]. Since then, the Bio-Medical Group and the Health Physics Division of BARC have undertaken a phased programme of studies aimed at obtaining data on the possible detrimental effects of continuous exposure to elevated background radiation on human and other biological systems. Historical records of this coastal strip of land (14] indicate that it has been inhabited for centuries.
The monazite bearing HBRA of Kerala (near Quilon town) is a coastal strip about 55 km long and roughly 0.5 km wide extending from Thekkumbhagam (Quilon Dist.) in the south to Purakkadu (Alleppey Dist.) in the north. This radioactive strip is effectively an island with the Arabian sea in the west, backwaters in the east and the Ashtamudi and the Kayamkulam lakes (which are connected to the sea) at its southern and northern ends. The beach along the western side of the strip contains monazite sand mixed in various proportions with ordinary sand. The radioactivity of the beach sands is due to monazite which contains mainly thorium and also traces of uranium. The thorium content of the monazite ranges from 8.0 to 10.5’7o (as against 5—6% in the Brazilian monazite deposits) and is among the highest reported in the world [8, 35].
The radiation intensity of the monazite belt is very non uniform. There
are two main features of the dose variation.
- Radiation ismaximum near the sea shore and gradually decreases towards the back
- Itincreases from a relatively low level in the north to a maximum near the village of Puthenthura in the Along the coast the high radiation belt is frequently interrupted by stretches with normal background radiation levels.
Human POpulations Living in Natural HBRA 145
The first phase of the programme was an assessment of the radiation dose received by the population (approximately 70,000 residing in about 13500 households—as estimated in 1968-69) living in the monazite belt (3] Dosimetric studies showed that the external radiation level was around 4 mGy/y [12, 13]. Of the 2374 households covered by the dosimetric survey, 24.6% recorded dose levels grater than 5 times the normal background; 8.8% of the household received more than 10 times the normal radiation level and about l to recorded more than 20 times the normal levels. With regard to individual exposure levels it was observed that 2020 of the 8513 individuals surveyed received exposures greater than 5 mGy/y. Of these 551 individuals received exposures greater than 10 mGy/y, while 57 individuals received a dose greater than 20 mGy/y. The amount of dose received by an individual was seen to depend on the place of residence, type of dwelling and occupation. The highest annual exposure encountered was 38.4 mGy for a dwelling and 32.6 mGy for an individual [33].
The monazite belt supports rich vegetation and thus offer opportunities
to study in situ the long term effects of chronic radiation exposure on plants. In the course of survey of the flora of this region, morphologically aberrant plants have been observed among species of Crotalaria and Lochnera which showed flower and leaf abnormalities. Accumulation of radionuclides of the thorium series in plants growing in these regions has also been demonstrated and significant positive correlations have been obtained between the radionuclide content of the plants and the soil activity for a large number of species [11, 21]. Cytological studies were carried out on a number of plant species from the radioactive areas. The freqency of occurrence of meiotic abnormalities and pollen sterility was found to be higher in plants growing in HBRA than in plants of the same species from adjoining areas with normal background radioactivity. The higher chromosomal damage was found to be correlated with the external radiation level to which the plant species were exposed as well as to the absorbed radionuclide content. Experiments with Tradescantia clone 02 have further demonstrated that the contribution from absorbed radionuclides is much more important than external radiation in the production of biological damage as measured in terms of the frequency of somatic mutation rates of staminal hair [23).
In addition to plants, investigations have also been carried out on rodents
inhabiting the HBRA of Kerala. In 1966, the UK Medical Research Council reported a negative result from a study made on the black rat, Rattus rattus [14, 15]. A total of 21 measurements, 6 dental and 15 skeletal, were made on each of the 450 rats trapped from the monazite belt. The rats in the study area received radiation doses 7.5 times greater than those in the control area. No statistically significant differences were found between the study and control populations.
Parida et al [25) and Mohapatra and Parida [22] from Utkal University in Orissa have investigated the occurrence of nuclear anomalies in the bone marrow cells of black rats from the HBRA of Kerala and also from Chatrapur
in Orissa (natural deposits of thorium containing monazite sand occur in Chatrapur although the mean radiation intensity of this locality is not available). Wild rats trapped from these two localities are claimed to show nuclear anomalies such as acentric fragments, micronuclei, karyohexis, double minutes, interchromosornal sticky connections, chromosome pulverization and anaphase bridges. No such abnormalities were observed in the control animals. Parida and Mohapatra [26] also studied the occurrence of micronucleatcd erythrocytes in the peripheral blood of rats trapped from these two areas and reported a significant radiaion associated increase. They also reported a higher frequency of double minutes (DMs) in the bone marrow cells of rats from Quilon and Chatrapur and interpreted this as a defence mechanism to counteract the detrimental effccts of elevatcd background radiation.
Chromosomal aberration studics on wild rats inhabiting the HBRA of
Kerala and adjoing control regions were carried out by Aravindan and Mahajan
[2] from BARC. Cytological analysis of over 8000 bone marrow metaphase spreads (from 42 animals) trapped from HBRA and 5267 cells (trom 24 animals) trapped from control areas revealed mostly chromatid type aberrations. Only a few cells had chromosome type aberrations. Cytogenetic data front this study did not indicate any significant difference between the study and control populations. Double minutes, which are implicated in gene amplification, were not observed in any of the animals studied from HBRA. These observations contradict the findings of Parida and Mohapatra but are in agreement with those of Jamali [17] who also reported negative results from chromosomal aberration studies on wild rats from the HBRA of Ramsar, Iran.
An ad hoc demographic survey was conducted by BARC with the assistance of the Bureau of Economics and Statistics, Kerala state and the results were published in 1972 [l 2]. Details of 13720 pregnancies were obtained by interviewing 2420 couples living in areas spread over the full length of the radioactive strip. The questions sought information relating to the following indicators of genetic change: sex ratio among the offspring; fertility index, infant mortality, pregnancy terminations, multiple births and gross abnormalities. The couples were grouped according to the personal exposures received by the female membcr, since in gcneral the maternal dose recorded was higher than the paternal dose.
The differenccs in ttie fertility index (number of pregnancies per couple) between groups receiving different levels of background radiation (1—5, 6—10, 1*.—20) and > 20 mGy/y) were not statistically significant although there was a tendency towards lower values as onc proceeded from Purakkadu in the north to Thekkumbhagam in the south. The couples in the group receiving exposures greater than 20 mGy/y had the lowest value of fenility index and the Ngflest value of infant mortality. However, the extremely small sample size of 22 couples make it difficult to draw any firm conclusions. The total loss of offspring was also significantly higher in this small group of couples but no pattern emerged for the three lower exposure levels
(1—5, d—10 and 11—20 mGy/y). With the exception of some parameters in the highest exposure group, none of the 6 indicators mentioned above showed statistically significant differences between the population groups exposed to different radiation levels. It was concluded in 1972 that the various parameters which were analyzed in the first phase of this study may not be sensitive enough to reveal any significant differences in the small population available for the study. Considering the fact that only about l % of the households surveyed in the HBRA recorded a dose in excess of 20 mGy per year, it is highly unlikely that, even if the investigators had covered the entire population falling in this category, it would have been possible to come up with enequivocal conclusions.
Human chromosomal studies on the inhabitants of HBRA and adjoining control regions were initiated by the Bio-Medical Group of BARC in 1975 and this work is still being continued. Blood samples from 435 adults comprising of 170 individuals (avg. age 27.7 yrs) from control areas and 265 (avg. age 30.7 yrs) from HBRA have been cytologically analyzed using conventional staining techniques. The frequency of dicentrics plus rings for the samples studied from control and HBRA was found to be 8.26 + 1.72 and 11.43 + 1, 59 per 10,000 cells respectively [8, 10]. The increase observed in HBRA samples is not statistically significant. However, the possibility of a marginal increase cannot be ruled out. It may be pointed out that values ranging from 5—10 per 10,000 cells have been reported for normal subjects studied from different population groups [19]. A variety of factors such as inherent variability, age of the subject, personal habits, exposure to environmental mutagens, viral infection, and the duration of culture time are known to contribute to the divergence among different studies [4].
Chromosome studies were also carried out on cord blood samples from newboms to estimate the spontaneous levels of chromosomal abnormalities and also to see whether foetel exposure resulted in enhanced aberration levels in samples from the HBRA. As compared to adults, the frequency of dicentrics plus rings observed among the new boms was relatively low. Moreover, the frequencies observed among newboms from control areas (1.93 + 0.18) and the HBRA (2.41 + 0.67) x 10 were comparable [10]. During mid 1980s monitoring of newboms was initiated to estimate the incidence of major congenital abnormalities and genetic disorders identifiable at birth. This is a collaborative study programme between BARC and the Health Department of Kerala State, and includes six hospital units covering inhabitants from control and radioactive areas. Monitoring the newboms for ahout 85 types of congenital anomalies had covered 19,561 babies at SAT Hospital, Trivandrum (a referal hospital which is located 90 kms to the south of HBRA), 38,109 babies from normal background areas and 5584 babies from HBRA upto Aug. 1990 [10]. The overall incidence of congenital anomalies was calculated to be 12.47 + 0.8 , 9.18 + 0.5, and 13.61 A 1.6 per 1000 newboms from referal, control and HBRA populations respectively. The frequency of anomalies in the HBRA, though significantly different
from control areas, was comparable with the referal control population (13.61 vs 12.47). However, two anomalies, namely hypospadias (urinogenital system) and calcaneo valgus (musculoskeletal system) showed excess among samples from the HBRA. Whether this increase is really attributable to radiation exposure or is due to some other aetiological factors is not clear at the moment. A possible source of error here is dosimetry. HBRA and NBRA have been demarketed based on the dosimetry data of 1971. The dose measurements then were not carried out at the specific addresses where the mothers of the newboms lived. Since the radiation levels can at times be significantly different in close by areas the possibility of some addresses which have been taken as NBRA having high radiation levels and vice versa cannot be ruled out. In view of this a programme to revise the dosimetry at the specific addresses where the concerned families lived has been initiated.
The frequency of Down’s Syndrome (DS) among the inhabitants of HBRA in Kerala has been a topic of controversy. Investigations so far carried out by BARC do not indicate any clear increase in DS cases in the HBRA. The data on the incidence of congenital anomalies showed 8 DS case among 19561 newboms at SAT Hospital, Trivandrum; 14 cases among 38109 newboms from control areas and only one case among 5584 newboms from the HBRA. Pooling the data from Trivandrum and the area control gives a figure of about l DS for 2500 newboms. Karyotype analysis on cord blood samples collected from control areas gives a frequency of approximately 1 in 800 which is comparable to the values that have been reported for other population groups. Thus, discrepancies in the data on DS based on clinical screening of newboms at the time of birth and that given by kaiyotype analysis on cord blood samples reflects diagnostic problems and underlines the necessity of cytogenetic monitoring for proper identincation of even such well known syndromes. It may be pointed out that the sample size covered, in particular from HBRA, is inadequate and only a larger data base can provide unambiguous conclusions.
The spontaneous level of karyotypic abnormalities among the cord blood samples from control areas was observed to be 0.68’7 (20/2934). The overall incidence of chromosome anomalies among newboms has been reported to be 0.64% by UNSCEAR [34] and the BARC data are in good agreement with this figure.
At the HBRA of Kerala two investigations were made by staff of the All India Institute of Medical Scien.ces, New Delhi. In 1°76, they published a study [18] that were prompted by a report that thyroid neoplasms were especially prevalent in Kerala State. As the thyroid gland is known to be highly sensitive to radiogenic cancer induction [28, 30], it was reasonable to suspect that the excess might be occurring among the inhabitants of the HBRA, but the study gave negative results thyroid swellings (benign and malignant) were found in humans from both the study and control populations, the rates being 13 and 14 x 10°3 respectively.
While conducting the above mentioned study Kochu Pillar and his associates observed 12 cases of DS among 13000 study population compared with none in the 6000 control population, a difference which appeared statistically significant. However, the validity of this study has been questioned by Edwards and Hamden [6] and Sundaram (32]. The difference seen by Kochu Pillai et a1 may be due to the abnormally low value among the control group. This has made an apparently normal value in the study population appear very high.
It has been suggested that dermatoglyphic studies, i.e. finger and palm prints, are easy to perform and might indicate genetic change. In 1973 a combined Indo-US team reported such a study [1]. Several traits were measured on hand prints obtained from 70 men inhabiting HBRA; the control group comprised of similar number of men living in a normal background region 120 km to the south of the monazite belt. No significant radiation related difference was seen.
In 1990 the results of a cross sectional survey among the inhabitants of HBRA in Kerala claimed [24, 25] higher prevalence rates for genetic disorders and congenital malformations including Down’s syndrome. These studies were not done on newboms and therefore represent prevalence rates in the existing population. Comparison between incidence rates and prevalence rates is not possible and these two estimates are bound to be different. Further, it is also important to rule out the effect of various confounding factors before attributing the observed effect to radiation exposure. While many factors (both environmental and host factors) have been implicated in the aetiology of DS, the influence of maternal age is perhaps the most important element [3 l]. It would have been very informative if Padmanabhan et al had provided the maternal age of those mothers who had given birth to DS babies.
While the data on the prevalence of DS in the HBRA of China [20, 36] reveals a significant increase of DS cases in the HBRA, the investigators associated with this study are of the opinion that the increased frequency of DS cases might be attributed to higher maternal age.
The assessment of the magnitude of biological effects in man following exposure to low dose rate chronic irradiation continues to be a highly debated issue. Low level radiation is potentially a cause of mutations in germ cells that may give rise to hereditary diseases in offspring and to mutations in somatic cells that may give rise to cancer in the exposed individual. With respect to the endpoints referred to above the information regarding low dose rate irradiation in man is almost entirely inferential and extrapolated from the effects observed at high doses. Though it is a universal belief among scientists that human beings should be susceptible to transmissible genetic damage from radiation exposure to the female egg cells or male sperm cells, such effects were not seen among the atomic bomb survivors despite a rigorous follow up for more than four decades. The only significant report which presented evidence for radiation induced transmittable genetic
damage to the progeny of the exposed individual was from Sellafield f7]. The findings of this study have not been widely accepted. Further work is needed either to confirm or to refute these observations. In view of the uncertainties and inherent confounding factors involved in studies of very small radiation doses, it is likely that no single population can provide reliable (or substantial) information to establish the absence or presence of small effects, if any.
REFERENCES
- Ahuja,R., Sharma, A., Nampoothiri, K.U.K., Ahuja, M.R. and Dempster,
E.R. Evaluation of effects of high natural background radiation on some genetic traits in the inhabitants of monazite belt in Kerala, India. Human Biology 1973; 45 : 167—179.
- Aravindan,V. and Mahalan, S.K. Cytogenetic studies on black rats, Rattus rattus from monazite bearing high background radiation areas of Kerala coast and adjoining regions. International conference on high levels of natural radiation, Ramsar, Nov. 3—7, Book of Abstracts 1990; l 10—111.
- Bharatwal,S. and Vaze, G.H., Radiation dose measurements in the monazite areas of Kerala State in India. Proc. Second U.N. International Conference on Peaceful Uses of Atomic Energy, Geneva 1958; 23: 156.
- Bender, M.A., Awa, A.A., Brooks, A.I., Evans, H.J., Groer, P.G., Littlefield,G., Caries Periera, Preston, R.J. and Wachholz, B.W. Current status of cytogenetic procedures to detect and quantity previous exposures to radiation Mutation Research 1988; 196: 103—159.
- Committeeon Biological Effects of Ionizing Radiations (BEIR III).The effects on populations of exposure to low levels of ionizing United States National Academy of Sciences, National Research Council. National Academy Press, Washington; 1980.
- Edwards J.and Harnden, D. Matters arising. Down’s Syndrome in Nature 1977; 267: 728.
- Gardn,er,, Snee, MW. and Hall, AJ. Results of case-control study of leukaemia and lymphoma among young people near Sellapeld nuclear plant in West Cumbria. British Medical Journal 1990; 300: 423—429.
- George,P., Sundaram, K., Mistry K.B. and Gopal-Ayengar, A.R. Investigations on human populations residing in high background radiation areas of Kerala and adjoining regions. Biological and environmental effects of low level radiation, 1976; II: 325—329, IAEA.
- George, K.P., Aravindan, K.V., Joseph, B.,Thampi, M.V., Cheriyan, V.D., Kurien, J., Shah, V.R. and Sundaram, K. Monitoring for unstable chromosomal aberraiions in populations exposed to ionizing radiations. In: lvtethodoiogical approaches for estimation of low dose effects of radiations and chemicals in the environment. Proceedings of an Indo-FRG Seminar, Bombay, March 15—19 1983; 25—34.
- George, K.P., Cheriyan, V.D., Kurien, C.J., Thampi, M.V., Joseph, B., Karuppasamy, C.V., Ramachandran, E.N., Sathy, N. and Sarvanandan, K.V. Monitoring human populations from high natural background radiation areas of Kerala Coast, Interim Status Report, BARC/1991/R/005.
Humay Populations Living in Natural HBRA 151
- Gopal-Ayengar, A.R., Nayar, G.G., George, K.P. and Mistry, K.B. Biological effects of high background radioactivity: Studics on plants growing in the monazite- bearing areas of Kerala Coast and adjoining Indian Journal of Experimental Biology 1970; 8: 313—318.
- Gopal-Ayengar, A.R.,Sundaram, K., Mistry, B., Sunta, C.M., Nambi, K.S.V., Kathuria, S.P., Basu, A.S. and David, M. Evaluation of the long term effects of high background radiation on selected population groups on the Kerala Coast. Peaceful Uses of Atomic Energy, 1972; 11: 31—5 1.
- Gopal-Ayengar, A.R., Sundaram, K.,Mistry, K.B. and George, K.P. Current status on investigations on biological effects of high background radioactivity in monazite bearing areas of Kerala coast in South International Symposium on areas of high natural radioactivity, Brazil, June 16—20, 1975; 19—27.
- Gruneberg, Genetical research in an area of high natural radioactivity in South India. Nature 1964; 204: 222—224.
- Gruneberg, H.,Bains, G.S., Berry, R.J., Linda, Riles, Smith, C.A.B. and Weiss R:A. A search for genetic effects of high natural radioactivity in South Medical Research Council, Special Report Series No. 307, London. 1966.
- Hanson,P. and Komarov, E. Health effects in residents of high background radiation regions. In: Biological effects of low level radiation. Proceedings of a Symposium, 11—15 April. 1983. IAEA/WHO, 211—220.
- Jamali, M.The study of high natural radioactivity effects on the bone marrow activities of Rattus rattus in Ramsar [chromosomal study). International Conference on high levels of natural radiation, Ramsar, Iran, 3—7, Book of Abstracts, 1990; pp 115.
- Kochupillai,, Verma, I.C., Grewal, M.S. and Ramalingaswamy. Down Syndrome and related abnormalities in an area of high background radiation in coastal Kerala, Nature 1976; 262: 60—61.
- Lloyd,C., Purrot, R.J. and Reeder, E.J. The incidence of unstable chromosome aberrations in peripheral blood lymphocytes from unirradiated and occupationally exposed people. Mutation Research 1980; 72 : 523—532.
- Luxin, W., Yongru, Z., Zufan, T., Weihui, H., Deqing, C. and Yongling, Y. Epidemiologicalinvestigation of radiological effects in high background radiation areas of Yangjiang, Journal of Radiation Research 1990; 31: 119—136.
- Mistry,B., Gopal-Ayengar, A.R. and Bharathan, K.G. On the radioactivity of plants from the high radiation areas of the Kerala coast and adjoining regions.
- Studieson the uptake of alpha and gamma emitters. Health Physics 1965; 11: 1459-1470.
- Mohapatra, and Parida, B.B. Cytogenetic evidence of gene amplification in natural populations of black rats from radioactive areas. Current Science 1988; 57: 468—471.
- Nayar, G.G.,George, K.P. and Gopal-Ayengar R., On the biological effects of high background radioactivity : Studies on Tradescantia grown in radioactive monazite sand, Radiation Biology 1970; 10: 287—292.
- Padmanabhan, V.T., Sugunam, A.P., Brahmaputran, C.K. and Suresan, A.P. Genetic effects in areas of high naturalbackground radiation in Kerala, Indo-US Symposium, Effects of environmental and genetic factors on pregnancy outcome, Delhi, April 3 May 3, (Abstract), 1990; pp 45.
- Padmanabhan,T., Sugunan, A.P., Brahmaputran, C.K. and Suresan, Ar. Genetic Effects of Ionizing Radiation, Information Unit on Militarization and Demilitarization in Asia (IUMDA) Newsletter 1990; 2 and 3; 154—163.
- Parida,B. and Mohapatra, G. Micronucleated erythrocytes in the peripheral
smear of Rattus rattus from radioactive areas. The nucleus 1986; 29 : 62—65.
- Parida,B., Mohapatra, G. and Parida, S.C. Occurrence of nuclear anomalies in the bone marrow cells of black rats inhabiting high background natural radioactive area. In: Perspectives in Cytology and Genetics. Eds. G.K. Manna and U. Sinha, 1986; 461—465
- Ron,, Modan, B. and Preston, D. Thyroid neoplasia following low-dose radiation in childhood. Radiation Research 1989; 120: 516—531.
- Rose, K.S.B. Review of health studies at Nuclear Energy 1982; 21: 399—408.
- Shore,E., Hildreth, N. and Woodard, E. Thyroid tumors following thymus irradiation. Journal of National Cancer Institute 1985; 74: 1177—1184.
- Staples, A.J., Sutherland, G.R., Haan, E.A. and Clisby, S. Epidemiology of DownSyndrome in South Australia, 1960—89, American Journal of Human Genetics 1991; 49:
- Sundaram, Matters arising. Down’s Syndrome in Kerala, Nature 1977; 267: 728—729.
- Sunta,M., David, M., Abani, M.C. and Nambi, K.S.V. Analysis of dosimetry data of high natural radioactivity areas of SW coast of India. Second Special Symposium on Natural Radiation Environment, BARC, January 19—23. Bombay. 1981; pp 35—42.
3J. United Nations Scientific Committee on Effects of Atomic Radiation, 1988.
- WorldHealth Effect of radiation on human heredity: Investigations of areas of high natural radiation. WHO Technical Repon Series No. 1ti6, WHO, Geneva. 1959; pp 1—47.
- Zufan, and Luxin, W. An epidemiological investigation of mutational diseases in the high background radiation areas of Yangjiiang, China. Journal of Radiation Research 1986; 27: 141—150.
16. Congenital Defects and Post-Implantation
Mortality in the Offspring of Gamma
Irradiated Male Mice
- Bhattacharjee
Radiation Biology and Biochemistry Division Bhabha Atomic Research Centre, Bombay, India
Introduction
Changes in the genetic material of germinal cells due to X-irradiation leading to congenital abnormalities in the offspring of mice have been reported for the first time by Nomura [12]. Subsequent elaborate studies confirmed that various types of birth defects are induced in the progeny of mice when either of their male or female parents was administered different doses of X-rays [5, 6]. So far as our knowledge of published literature is concerned, a detailed information is lacking on the aspects of genetic induction of malformations in the progeny following parental exposure to gamma rays. Both X-rays and gamma rays are electromagnetic radiation giving sparse ionization. The distribution of the ionizing events produced by these rays in the biological material is variable. Typical linear energy transfer (LET) valiies are 0.3 KeV/yen for cobalt-60 gamma rays and 2 Kev/pm for 250 kV
X-rays [4].
In view of the variation in LET values between these two types of ionizing radiation, we thought it is pertinent also to investigate the genetic effects of gamma radiation with particular reference to induction of malformation in the off-spring of mice.
Materials and Methods
Inbred strain Swiss mice from BARC animal house were used for all the experiments. They were housed in husk-bedded metallic cages and were maintained with standard laboratory diet and tap water for drinking ad libitum. Male mice aged 10—12 weeks were given whole body irradiation from cobalt-60 gamma source (BARC) at the rate of 0.55 Gy/min, for a total dose of 1, 2 and 4 Gy. Unexposed males were used as control. Each male was allowed to copulate at intervals of 1—7, 8—14, 15—21 and 64—80 days with a pair of 12 weeks old virgin females of the same strain. Copulation at these periods is corresponding to treated sperms, late spermatids, early spermatids and spermatogonial stem cells respectively at the time of radiation exposure. Therefore, the response of the germ cells of all stages to gamma radiation could be analysed. The identity of the females from all the groups
154 BHATTACHARIEE
kept secret by giving a code number for each mouse to avoid bias at the time of scoring. The presence of a vaginal plug was considered as day 1 of pregnancy and females were humanely killed by cervical dislocation on the gestation day of 19. The uterine contents were counted for live and dead fetuses and large and small moles (representing late and early deaths) and index of dominant lethality was calculated f6].
All live fetuses were removed from the uterus, weighed and kept fn a coded vial containing 109c formalin and later examined for gross morphological abnormalities. A fetus weighing less than 70’7 of the average of the rest of the litter was classified as a dwarf [11]. The frequency of abnormal fetuses was calculated as the number of fetuses with morphological defects divided by number of live normal embryos [11].
Standard errors were calculated for the data whenever thought necessary using a standard formula.
Results
The data on total number of pregnant mice and classification of various types of implantations in each experimental and concurrent control groups are shown in the Table 1. The total number of pregnant mice and their fetuses examined in this study were 218 and 1608 for irradiated groups and 94 and 779 for control groups respectively.
The relative radiation sensitivity to different germ cell stages in terms of dominant lethality is shown in Table 2. The percentage of pregnant females with one or more dead implantations is greaUy enhanced (70—100’7») when 3 stages of post-meiotic germ cells are exposed to radiation dose I—4 Gy compard to 42’7» observed in concurrent control group. However, when the stem cells are irradiated, the percentage of females with the same end point is comparable to the control group. Similarly the percentage of post- implantation survival as measured by the ratio of live fetuses by total implantation, decreased with increasing dose for all the stages of post- meiotic cell except stem cell spermatogonia. On the contrary, dead implantation per female increased with dose enhancement in mature sperm and late and early spermatids. For stem cells only at the highest dose rate of 4 Cry dead implantations are found to be more. The induction of post-implantation dominant lethality, thereby increased along with the dose. This lethality is primarily due to death of implants at mole stage. The dominant lethality is at its highest for each dose level for early spermatids indicating its greater sensitivity to radiation than matured sperm and late spermatids. However, dominant lethality was not induced at doses of 1 and 2 Gy for spermatogonial stem cells.
The results of the analysis on fetal abnormalities are shown in Table 3. Abnormal embryos are recorded in all three doses and four time intervals showing responses of all the germ cell stages to gamma radiation at a higher frequency. When the data from all the post-meiotic stages of germ cells are
Table 1. C lassification of implants from gamma irradiated male mice mated at varying intervals after irradiation
| Radiation dose (Gy) | Interval (days) | Number of pregnant females | Total implants | Normal fetuses | Abnormal living fetuses | Dead fetuses | Large moles | Small
moles |
| 1 | 1—7 | 20 | 199 | 172 | 3 | 2 | 13 | 9 |
| 2 | 1—7 | 15 | 136 | IOP | I |
|
20 | 5 |
| 4 | 1—7 | 23 | 209 | 116 | 6 | 3 | 52 | 32 |
| 1 | 8—14 | 10 | 78 | 63 | 3 | 2 | 8 | 2 |
| 2 | 8—14 | 7 | 63 | 44 | 4+1‘ | 1 | 11 | 2 |
| 4 | 8—14 | 20 | 156 | 85 | 10 | 1 | 59 | 1 |
| 1 | 15—21 | 11 | low | 82 | 2 | 3 | 11 | 8 |
| 2 | 15—21 | 10 | 79 | 49 | 3 | 0 | 27 | 0 |
| 4 | 15—21 | 10 | 68 | 25 | 1 | 2 | 36 | 4 |
| I | 64-80 | 44 | 430 | 385 | 18 | 1 | 12 | l l |
| 2 | 64-80 | 35 | 319 | 278 | 20 | 0 | 12 | 7 |
| 4 | 64-80 | 13 | 129 | 99 | 8+1‘ | 6 | 10 | 7 |
| Concurrent | ||||||||
| control | 1—80 | 94 | 832 | 769 | 5 | 5 | 42 | 11 |
Table 2. Frequencies of dominant lethality following exposure of gamma rays to male mice
| Radiation dose (Gy) | No. of pregnant mice | Interval time (days) | Total implants per female | Dead implants per female | Live fetuses/ total implants
{%j |
Induced domi- nant lethality (to) | Females with one or with dead
implants (to) |
| 1 | 20 | 1—7 | 9.95 + 0.33 | 1.20 + 0.18 | 88.38 | 4.03 + 1.54 | 70.00 |
|
|
15 | 1—7 | 9.06 + 0.43 | 1.73 + 0.25 | 75.52 | 13.33 + 3.17 | 86.70 |
| 4 | 23 | 1—7 | 9.09 + 0.29 | 3.78 + 0.54 | 64.35 | 30.12 + 3.44 | 95.65 |
| 1 | 10 | 8—14 | 7.80 + 0.71 | 1.20 + 0.32 | 82.50 | 10.41 + 5.04 | 70.00 |
| 2 | 7 | 8—14 | 9.00 + 0.57 | 2.00 + 0.43 | 77.97 | 15.32 + 4.63 | 85.70 |
| 4 | 20 | 8—14 | 7.70 + 0.47 | 3.05 + 0.44 | 65.91 | 28.45 + 4.24 | 95.00 |
| 1 | 11 | 15—21 | 9.60 + 0.27 | 2.00 + 0.23 | 78.81 | 14.42 + 3.32 | 90.90 |
| 2 | 10 | 15—21 | 7.90 + 0.75 | 2.70 + 0.71 | 67.47 | 26.74 + 4.91 | 90.0f1 |
| 4 | 10 | 15—21 | 6.90 + 0.34 | 4.30 + 0.51 | 37.86 | 58.88 + 7.03 | 100.00 |
| 1 | 44 | 64-80 | 9.77 + 0.25 | 0.54 + 0.09 | 94.43 | -2.57 + 0.25 | 40.90 |
| 2 | 35 | 64-80 | 9.11 + 0.48 | 0.54 + 0.13 | 94.52 | -2.64 + 8.81 | 37.14 |
| 4 | 13 | 0 | 9,92 + 0.50 | 1.77 + 0.31 | 82.69 | 10.16 + 3.11 | 76.92 |
| Concurrent control | 94 | 1—80 | 8.85+0.24 | 0.62+0.07 | 92.09 | – | 42.48 |
Table 3. Frequencies of abnormal fetuses derived from various stages of germ cells of male mice exposed to gamma rays
| Radiation dose | Time interval | Live fetuses | Abnormal | Frequency = |
| (Gy) | (days) | (A) | fetuses (B) | B/A x 100 |
| 1 | 1—7 | 175 | 3 | 1.71 |
| 2 | 1—7 | 110 | 1 | 0.90 |
| 4 | 1—7 | 122 | 6 | 4.92 |
| 1 | 8—14 | 66 | 3 | 4.55 |
| 2 | 8—14 | 48 | 4 | 8.33 |
| 4 | 8—14 | 95 | 10 | 10.53 |
| 1 | 15—24 | 84 | 2 | 2.35 |
| 2 | 15—24 | 52 | 3 | 5.77 |
| 4 | 15—24 | 26 | 1 | 3.85 |
| Total | 778 | 33 | 4.24 | |
| 1 | 64080 | 403 | 18 | 4.47 |
| 2 | 0 | 298 | 20 | 6.71 |
| 4 | 0 | 107 | 8 | 7.48 |
| Total | 808 | 46 | 5.69 | |
| Concurrent | 1—80 | 774 | 5 | 0.64 |
| control |
summed up, there are 33 fetuses with external abnormalities among 778 live fetuses giving a frequency of 4.24. From stem cell spermatogonia collectively 46 abnormal fetuses are recorded amongst 808 living embryos, the frequency being 5.69. In the control group only 5 abnormal out of 779 live fetuses are observed which gives a frequency of 0.64. Therefore, induction of abnormal embryos in the irradiated groups is clearly very much above the control mice. The frequency of abnormalities, of course, is found to be marginally higher for the stem cell spermatogonia. There are, however, no clear dose response relationship for both the post-meiotic and spermatogonial stCm cells. In addition, when the data from any one dose level are collectively taken over the 4 time intervals, the results from each dose are very much higher than the control. Similar result has been obtained from each week, summed over all doses.
The types of malformations observed in the fetuses are shown in Table 4. The most common type of defects recorded is dwarfism in the present investigation. Dwarfism alone is accounting for 54% and in combination with other abnormalities is another 6% of all the malformed fetuses. Bruising neck and exencephalia are found to be next common features at a relative frequency of 18.4 and l 1.1. In the fetuses of control mice, except dwarfism, no other malformations are recorded.
Table 4. Frequencies of abnormal types fetuses Identified from external morphology
Number (relative frequency) of abnormalities
| Type of abnormality | Irradiated‘ | Control |
| Dwarfismb | 44(54.3) | 5(100) |
| + exencephaly | 3(3.7) | – |
| + naval hernia | 1(1.2) | – |
| + bruising neck | l(1.2) | – |
| Exencephaly | 6(7.4) | – |
| Brain hernia | 2(2.5) | – |
| Naval hernia | 1(1.2) | – |
| Abdominal hernia | 2(2.5) | – |
| Syndactyly | 1(1.2) | – |
| Microophthalmus | 1(1.2) | – |
| Bruising neck | 10(12.3) | – |
| + limb and tail anomaly | 4(4.9) | – |
| Abnormality in limbs
Abnormality of dorsal surface |
3(3.7)
l(1.2) |
– |
| White skin (Leukaemia?) | l(l .2) | – |
| Total | 81(100) | 5(100) |
‘Data pooled from all irradiated groups.
bLess than 709c of average body weight of rest of litter.
Discussion
In the present studies, the potential of gamma rays to produce post-implantation lethality and congenital malformations was evaluated for different stages of germinal cell in male mice. Gamma rays administered in the dose range 1-4 Gy, caused dose-dependent post-implantation dominant lethality for post-meiotic germ cell stages. And among the post-meiotic germ cells, early spermatid cells are more sensitive to radiation. These results are in consistence with earlier two studies with X-rays [6,12]. The rate of induction of dominant lethal matutions, however, was reported to be over twice as high after acute X-irradiation of spermatids as after chronic gamma-irradiation [8,13]. Post- implantation lethality, however, was not apparently detectable for spermatogonial stem cells at the dose level of 1 and 2 Gy in comparison with control animals. It was suggested from the results of X-ray experiments that the great majority of dominant lethals induced in the spermatogonial stem cells are eliminated before spermatogenesis proceeds [6].
The results of these experiments further indicated that the gamma irradiation to male parents causes a high incidence of congenital anomalies in the surviving offspring of mice. The overall occurrence of 4.24’7o malformations in post-meiotic stages is more than double compared to 2% reported in the progeny induced by X-rays whose LET value is almost 7 times If gamma rays. A direct correlation with the doses administered or the types of germ cells exposed to radiation could not be obtained. Similar findings were
reported earlier with X-rays (6). Recently it was confirmed that the malformations seen in the first generation following X-ray exposure are of genetic origin and can be transmitted to later generations {9]. The incidence of congenital malformations by radiation exposure to spermatogonial stem cells are a matter of great concern from hazard point of view. Unlike post- meiotic germ cells, stem cell spermatogonia remain permanent throughout life in the testes. Any change in the genetic level of these cells, therefore, may be transmitted in the progeny irrespective of copulation time barring initial few weeks after radiation exposure. This fact is to be taken care of when genetic hazards of radiation in man is evaluated.
Amongst the types of abnormalities dwarfism and exencephaly were found to be more common. There is a possibility that the eKpressions of genes concerning these abnormalities are more susceptible to the radiation. In the control groups of mice except a few dwarfs, other anomalies were not detected. Exencephaly, although, is one of the common types of neural tube defects [1], it occurs as low as 0.2% in ICR strain of mice to as high as 6% in embryos of MT strain [10]. We have so far no data available in this regard of the strain used in the present work, neither we have observed any exencephalia in 779 embryos examined in the control mice. Besides, all the exencephalic fetuses were obtained from irradiated stem cells only in the present investigation. X-rays (5,6] and certain chemical teratogen [14) are known to yield a high incidence of exencephaly in mice. Direct radiation exposure at early stages of embryogenesis also causes higher incidences of exencephalia [3].
Genetic damage in the stem cell spermatogonia by gamma rays at dose range 1—2 Gy cannot be detected by dominant lethal method. Congenital defects, however, are identified at the same lower doses, suggesting supenority of the method to detect genetic lesions. It appears from the present work and previous work with X-rays {5, 6j that incidence of congenital malformations is a relatively easy and convenient end point to score and therefore, may be used simultaneously along with dominant lethality as an indicator to assess the comparative genetic damage to various radiation exposures {7).
REFERENCES
- Campbell, L.R., Dayton, D.H. and Sohal, G.S. A review of human and animal studieson the etiology of neural tube Taratology 1986; 34: 171—187.
- Friedberg, Faulkner, D.N., Neas B.R., Hanneman, G.D., Darden, E.B., Deal,
R.B. and Parker, D.E. Dose-incidence relationships for exencephalia, anophthalmia and pre-natal mortality in mouse embryos irradiated with fission neutrons or 25 kV X-rays. International Journal of Radiation Biology 1987; 52: 223—236.
- Darden E.B., Deal R.B. and Parkar, E. Dose-incidence relationships for exencephalia, anopthalmia and pre-natal mortality in mouse embryos irradiated with fissionneutrons or 25 kV X-rays. Int. J. Radiat. Biol., 1987; 52: 223—236.
- Hall,J., Radiobiology for the radiologist (J.B. Lippin cott, Philadelphia): 1988;
- PP.
- Kirk, M. and Lyon, M.F. Induction of congenital anomalies in offspring of femaleexposed to varying doses of X-rays. Mutation Research 1982; 106: 73—
- Kirk, K.M. and Lyon, M.F. Induction ofCongenital malformations in the offspring of male mice treated with X-rays at pre-meiotic and post-meiotic Mutation Research 1984; 125: 75—88.
- Knudsen,, Hansen, E.V., Meyer, D.A. and Poulsen, E. A proposed method for the simultaneous detection of germ cell mutations leading to fetal death (dominant lethality) and of malformations (male teratogenicity) in mammals. Mutation Research 1977; 48: 267—270.
- Leonard, A.Relation between the X-ray dose and the rate of dominant lethals induced by irradiation by mouse Mutation Research 1966; 3: 73—78.
- Lyon,F. and Renshaw, R. Induction of congenital malformation in mice by parental irradiation: transmission to later generations. Mutation Research 1988; 198: 277—283.
- Matsuda Comparison of the incidence of 5-azaeytidine induced exencephaly between MT/Hokldr and Slc: ICR mice. Teratology 1990; 41: 147—154.
- Nagao, Frequency of congenital defects and dominant lethals in the offspring of male mice treated with methylnitrosourea. Mutation Research 1987; 177: 171—178.
- Nomura, Prenatal exposure to X•rays and chemicals induces heritable tumors and anomalies in mice. Nature 1982; 296: 575—577.
- Searle, A.G.and Beeches, C.V. The effects of radiation dose rate and quality in the induction of dominant lethals in mouse spermatids. Mutation Research 1981; 81: 403-410.
- Takeuchi,K. and Takeuchi, Y.K. 5-azacytidine induced exencephaly in mice. Journal of Anatomy 1983 ; 140 : 403—412.
- DoseRate Effect for the Induction of Genetic Damage in Diploid Yeast Cells Exposed to Gamma or Beta-Radiation
B.S. Rao, N. Samara Narayanan and K.B. Anjaria
Division of Radiological Protection, Bhabha Atomic Research Centre, Bombay, India
Introduction
Dose rate effect has been investigated for a variety of end points such as cell killing, mutations, cell transformation, carcinogenesis and induction of genetic diseases. In general a reduction in the effectiveness has been observed for most of the end points. However, a lot of the information related to cell transformation and the induction of forward mutations in mammalian cell systems were not consistent [4, 7, 10, 20]. Particularly, negative dose rate effect was also demonstrated for the induction of transformation [2, 10]. As for the induction of forward mutations, dose rate effect seen in rodent cell lines were not seen in human cell lines [7, 8, 9]. Due to these differences in the results seen in different test systems, it has become necessary to investigate the phenomena of dose rate effects in as many systems and end points as possible to have a clearer view on the subject. Hence, we have undertaken dose rate effect studies extending to very low dose rates for the induction of gene conversion in diploid yeast cells BZ 34 and D7 both under growth and non growth conditions.
Materials and Methods
Yeast Strains
Diploid yeast strains Saccharomyces cerevisiae BZ34 and D7 used here, undergo reversion to prototrophy by the mechanism of gene conversion resulting in arginine and tryptophan independence respectively [3, 23]. Genotypes of these strains are described elsewhere [14].
Media, Plating and Scoring
Cells were routinely subcultured in YEPD broth (yeast extract I’d»; peptone 29 ; and dextrose 29») for 48 hours at 30 C. Cells were washed and resuspended in sterile distilled water and used for experiments. Treated cell suspensions were plated on omission media (arginine less for BZ34 and trp- for D7) to score the frequency of gene conversion. An appropriate dilution of the treated samples was plated on synthetic complete medium (SC) to score the number of viable cells. Colonies were counted after 3—4 days of
incubation of plates at 30’C. Gene conversion frequency is assessed by the ratio of the counts on omission plates to that on SC plates times the dilution factor, generally expressed as conversion frequency per million viable cells. Further details of these techniques are described elsewhere [13].
Irradiation
Cells were exposed to beta radiation using a suitable activity of HTO in the cell suspension. A final concentration of 3.7 x 107 Bq per ml of HTO results in a beta dose of 85 mGy h*’. Low dose rate gamma irradiation was performed using a 60Co gamma source of 1.2 GBq with a dose rate of approximately 10 mGy h°’. Acute gamma irradiations were performed in a gamma cell with a dose rate of approximately 10-Gy min*1.
Experimental
Low dose rate beta irradiation under non growth condition
Dose rate studies for HTO beta rays under non-growth condition were performed by suspending yeast cells Saccftaromyces cerevisiae D7 under different concentrations of HTO such that the beta radiation dose rates were
3.5 Gy h‘’ 0.42 Gy h*1, 0.06 Gy h ‘ and 0.014 Gy h‘’. Control samples were also incubated at 0-4°C throughout the duration of this experiment which lasted for 3 months. Periodically, aliquots were withdrawn and plated to check viability, back ground gene conversion frequency and gamma radiation response of preserved cells. Control cell suspension did not show any change in viability or conversion frequency under the experimental conditions. Gamma radiation response for the induction of gene conversion remained the same throughout the period of preservation indicating that cells do not undergo any changes in sensitivity during this period. Conversion frequencies were assessed after different durations of exposure to beta radiation to obtain the dose response.
Beta Irradiation Under Growth Conditions
Dose rate experiments under growth conditions using D7 strain, were performed for two dose rates, 0.423 Gy h*’ and 0.232 Gy h°’. The cells were incubated at 30°C in YEPD broth containing appropriate concentration of HTO. Throughout the irradiation cells were in exponential phase of growth. The number of cells inoculated in cultures were adjusted in such a manner that the cells did not reash. stationery pt:ase during t.he irradiation.
Gamma Irradiation
Low dose rate exposure to gamma rays under continuous growth condition was achieved by repeatedly sub-culturing the cells once in every 24 hours and growing them in an incubator placed in a radiation field of 10 mGy h*’ Cells were exposed for a period lasting over a month ensuring that the cells were in exponential phase of growth throughout the irradiation. Control
Dose Rate Effect for the Induction of Generic Damage 163
cultures were also maintained in a similar manner in a radiation free zone. During the period of growth aliquots were withdrawn at intervals of 2—3 days and the conversion frequency determined as described previously. The dose response obtained under chronic exposure condition was compared with that obtained for acute gamma irradiation performed in a gamma cell.
Results and Discussion
Dose rate studies with HTO beta rays under non-growth condition
Yeast strain D7 exposed to HTO beta rays at 0-4°C under non-growth condition in the dose rate range of 3.5 Gy h*’ to 0.014 Gy h°’ showed conversion frequencies in the range of 4 x 10“ Gy°’ to 5.5 x 10“ Gy‘’ (Fig. 1). Even though the frequency was slightly higher at lower dose rates studied, in the light of uncertainties which may arise due to very low HTO concentrations involved and irradiations lasting for several months, the differences observed cannot be considered as very significant. Our results are in agreement with those of Morrison ct al [11] but are not in agreement with those of Ito and Kobayashi [5].
0
Fig. 1. Variation of gene conversion frequency with HTO beta dose in yeast D7 strain incubated at different HTO concentrations. 0—3.5 Gy h ‘;
– 0.42 Gy h*’; 0.0.06 Gy h‘’, X—0.014 Gy h*’. Broken line represents
the least square fit derived by regression analysis.
Under exponential growth condition
Yeast strain D7 irradiated with beta radiations at a narrow range of dose rates i.e. 0.42 Gy h*’ and 0.232 Gy h*’ showed approximately similar response as seen by the results presented in Table 1. Even though the gene conversion frequency is slightly lower at lower dose rate the difference is statistically not significant. As compared to the response of stationary phase cells, the
Table 1. Dose rate studies In D7 strain durlng growth condition
Details of dose rate
Total dose
Convertants per
10 survivors
| (Gy) | Expt. I | Expt. II | |
| Control | 0 | 4.3 |
|
| Acute gamma | 10 | 48 | |
| Q: 0.423 Gy h‘1 | 10 | 96 | 98 |
| (23 h 40 min) Q: 0.232 Gy h‘’ | 10 | 67 | 82 |
| (43 h, 12 min) |
exponential cells appear to be nearly twice as sensitive to the induction of gene conversion. Since the dose rate range studied here is quite narrow, further experiments are currently in progress to explore the dose rate effect at lower dose rates.
Low Dose Rate Gamma Irradiation
Low dose rate exposure with gamma radiation under growth conditions were performed with both D7 and BZ34 strains. A remarkable enhancement in the conversion frequency was observed under low dose rate as compared to that observed for acute exposure. For D7 strain, convertants per 106 survivors per Gy were 5.4 A 0.5 and 35 5 for acute and low dose rate exposures respectively. Variation of conversion frequency with dose for D7 strain are summarized in Table 2. Analysis of this data suggests a remarkable difference in the induction of gene conversion under acute and chronic irradiation conditions.
Table 2. Induction of gene conversloti ln diplold yeast D7 Irradiated wtth gamma rays chronic or acute con0ltlon
Chronic exposure 10 mGy h*l
Acute exposure 10 mGy min‘’
| Dose (Gy) | Convertants per 106 survivors* | Dose (Gy) | Convertants per 10 survivors | |
| 0 | 10 | 0 | 10 | |
| 0.24 | 15 | 10 | 55 A 8 | |
| 0:72 | 20 | 20 | 134 -£ 22 | |
| 1.68 | 70 | 40 | 227 d- 38 | |
| 2.64 | 100 | 6£i | 306* 54 | |
| 3.\2 | 130 | 80 | 342 -k 19 | |
| 4.08 | 180 | 100 | 494:£ 22 | |
| 5.76 | 195 | |||
| 6.96 | 245 |
*Pooled data from 3 independent experiments.
**Values represent the mean from 2—3 experiments, errors represent the standard deviation
Dose Rate Effect for the Induction oyGenetic Damage 165
Similar results were obtained in BZ34 also. The response of acute and chronic irradiation conditions are presented in Figs. 2 and 3 respectively. The results have been pooled from several experiments. In spite of some scatter in the BZ 34 data, it is amply clear that the response is several fold higher than the cells exposed to acute irradiation. The gene conversion frequencies for BZ34 strain were 15 + 3 and 77.5 + 17.5 for acute and chronic exposures respectively. Our results are consistent with those reported by Kiefer et al [6] with continuous chemostat cultures. These results suggest that the cells fail to repair the radiation damage induced under very low dose rate condition. Further, high frequeficy of gene conversion observed under low dose rate condition suggests an amplification of radiation damage during the cell division process. Yeast cells are known to repair a significant fraction of potentially lethal and sublethal radiation damage induced at high doses and at high dose rates. It is not exactly known whether the mechanism of repair fails to recognize the damage induced at very low dose rates.
800
700
£i00
500
400
300
200
100
0 10 20 30 40 50 60
DOSE (Gy)
Fig. 2. Dose response curve for the induction of gene conversion of diploid yeast Saccharomyces cerevisiae. BZ34 acutely exposed to “Co gamma radiation. Dose rate was 10 Gy per minute. Different symbols represents the results of independent experiments.
The results of the above experiments clearly demonstrate the absence of dose rate effect for the induction of genetic damage in yeast cells. This is true for both gamma and beta radiations. On the contrary, observations here
166 Rno m w
700
600
500
300
200
100
0
230 460
DOSE cGy
690 920
Fig. 3. Variation of conversion frequency with accumulated dose in diploid yeast BZ34 continuously exposed under exponential growth conditions at a low dose rate of 10 mGy h*’. Symbols represents the results of two independent experiments.
suggest an increased sensitivity of cells to radiation under exponential growth condition.
Our results can be compared with those obtained in mammalian cell cultures for both cell transformation and induction of forward mutations. Some of the cell transformation experiments show a reduced effectiveness at lower dose rates [1, 4]. Few others repon a higher effectiveness at lower dose rates or in fractionated regimen [2,10]. Cell cycle sensitivity variations seem to interfere with the cell transformation process leading to contradictory results. As for the induction of forward mutations, rodent cell lines showed a reduction of mutation frequency at lower dose rates [20]. Similar studies with human cell lines however, failed to show a reduction in effectiveness at lower dose rates [7, 8, 9].
In radiation protection, ICRP-60 assumes a dose and dose rate reduction factor (DDREF) of 2 in extrapolating the risk derived from high dose rates to low dose rates [19]. This is mainly based on the carcinogenesis data [21] derived from mice exposed tO acute (0.45 Gy or protractcd exposures (0.06 mGy min*’). squans locus mutation experiments in mice also showed a three fold reduction in effectiveness when the dose rate is reduced from 9 Gy min*1to 8 mGy min°l, although no further reduction occurred at lower dose rates [15, 16, 17). At present a dose rate reduction factor of 3 is used in genetic risk analysis. Human epidemiological data is scanty to derive a reliable dose rate reduction factor. Data from breast and thyroid cancers do not suggest a dose rate effect [18, 22]. Further, the data from Hiroshima and Nagasaki survivors are not consistent with a DDREF higher than 1.5 on the basis of linear quadratic model [12].
Dose Rate Effect for the Induction of Genetic Damage 167
The above’ discussion clearly points out that our present knowledge of dose rate effect is restricted mostly to the high dose region related to the phenomenon of cell killing. Lower dose region leading to non lethal damage (cell modification) alone has importance in the evaluation of risk of stochastic effects such as radiation carcinogenesis and genetic effects. Due to the many limitations in the experimental studies carried out so far, renewed effort is necessary to clearly understand the phenomenon of dose rate effect.
REFERENCES
Balcer-Kubiczek, E.K., Harrison, G.H. and Thompson, B.W. Repair time for oncogenic transformation in C3H/10T2 cells. International Journal of Radiation Biology, 1987; 51: 219—226.
- Brenner, D.J. and Hall, E.J. The inverse dose-rate effect for oncogenic transformation by neutrons and charged particles: a plausible interpretation consistent with published data. International Journal of Radiation Biology 1990; 58: 745—758.
- Fogel, S. and Mortimer, R.K. Informational transfer in meiotic gene conversion. Proceedingsof the National Academy of Sciences, USA, 1969; 62: 76-103.
- Han, A., Hill, C.K. and Elkind, M.M. Repair processes and radiation quality in neoplastic transformation of mammalian cells. Radiation Research, 1984; 99: 249—261.
- Ito, and Kobayashi, K. Mutagenesis in yeast cells by storage in tritiated water. Radiation Research, 1968; 76: 130-144.
- Kiefer,, Muller, J. and Gotzen, J. Mitotic recombination in continuously gamma- irradiated diploid yeast. Radiation Research 1988; 113: 71—78.
- Koenig, F. and Kiffer, J. Level of dose-rate effect for mutation induction by gamma rays in human TK6 cells. International Journal of Radiation Biology,1988; 54: 891—897.
- Liber, H.L., Ozaki, V.H. and Little, J.B. Toxicity and mutagenicity of low dose rates of ionizing radiation from tritiated water in human lymphoblastoid cells. MutationResearch, 1985; 157: 77—86.
- Little,B. Low-dose radiation effects : Interactions and synergism. Health Physics, 1990; 59: 49—56.
- Miller,C. and Hall, E.J. X-ray dose fractionation and oncogenic transformations in cultural mouse embryo cells. Nature, 1978; 272: 58—60.
Morrison, D.P., Osborne, R.V. and Unrau, P. Q-ray induced gene conversion in
yeast. Radiation Research, 1981; 87: 50-58.
- Pierce, D.A. andVaeth, The shape of the cancer mortality dose-response curve for atomic bomb survivors. Radiation Effects Research Foundation Technical Report, 1989; 7—89.
- Rao,S., Murthy, M.S.S. , Raddy, N.M. S., Subrahmanyam, P. and
Madhvanath, U. Post-irradiation modification of radiation induced, heteroallelic reversion in diploid yeast. Effects of nutrient broth. Mutation Research, 1975; 28: 185—189.
- Rao,S., Anjatia, K.B. and Sankaranarayanan, N. Adaptive response studies
in diploid yeast Saccharomyces cerevisiae exposed to HTO beta and gamma radiation, Proceedings of International Symposium on Low Level Radiation and Living State, 1992.
168 RAO ET AL
- Russell,L., Russell, L.B. and Kelly, E.M. Radiation dose rate and mutation frequency. Science, 1958; 128: 1546—1550.
- Russell,L. and Kelly, E.M. Specific-locus mutation frequencies in mouse stem-cell spermatogonia at very low radiation dose rates. Proceedings of the National Academy of Sciences, U.S.A., 1982a; 79: 539—541.
- Russell, W.L. and Kelly, E.M. Mutation frequencies in male mice and the estimationof genetic hazards of radiation in Proceedings of the National Academy of Sciences, U.S.A., 1982b; 79: 542—544.
- Shore,E., Woodard, E., Hildreth, N. et al. Thyroid tumours following thymus irradiation. Journal of National Cancer Institute, 1985; 74: 1177—1184.
- Soman, S.D. and Rao, B.S. Biological Bases of ICRP-60 Recommendations MedicalPhysics Bulletin, 1992 17: 30-38.
- Thacker, and Stretch. Recovery from lethal and mutagenic damage during post-irradiation holding and low-dose-rate irradiation of cultured hamster cells. Radiation Research, 1983; 96: 380—392.
- Ullrich,C. & Storer, J.B. Influence of dose, dose rate and radiation quality on radiation carcinogenesis and life shortening in RFM and BALB/C mice p. 95 In: Late Effects of Ionising Radiation, IAEA, Vienna; 1978; Vol. 2.
- UNSCEAR,Genetic and Somatic Effects of Atomic Radiation, 1986 Report to the General Assembly, New York,
- Zimmermann,K., Kem, R. and Rasenberger. A yeast strain for simultaneous detection of induced mitotic crossing over, mitotic gene conversion and reverse mutation. Mutation Research, 1978; 28: 38 I—388.
18. Micronucleated Erythrocytes for Low-Dose Effects of Ionizing Radiation in the Bone
Marrow of Mice: Role of sample size
H.N. Bhilwade, R.C. Chaubey and P.S. Chauhan
Molecular Biology and Agriculture Division Bhabha Atomic Research Centre, Bombay, India
Introduction
A large number of environmental chemicals and ionizing radiation are capable of producing cytogenetic changes in laboratory animals. Analysis of chromosomes at metaphase has long been used to detect cytogenetic changes in laboratory animals and in human lymphocytes in vitro. Metaphase chromosome analysis provides an opportunity of detecting, both numerical and structural changes in chromosomes in various kinds of dividing cells. However, chromosome analysis is a time consuming process and requires suitable chromosome morphology and number for reliable scoring of chromosomal changes. Based on mouse bone marrow micronucleated erythrocytes (mn-Es), Schmid [26] and Heddle [13], working independently introduced an alternative system, providing an indirect measure of chromosomal damage. A number of investigators have shown bone marrow mn-Es to be a simple and rapid indicator of chromosomal damage induced by ionizing radiation and chemical mutagens [2, 5, 6, 8, 22, 24].
Formation of micronuclei are based on the principle that, during anaphase, acentric chromatid and chromosomal fragments lag behind [and sometimes due to spindle dysfunction even a whole chromosome), whereas the centric elements migrate to the spindle poles. In telophase, both the undamaged chromosomes and centric elements give rise to the daughter nuclei (Fig. 1). The lagging elements may be included in to the daughter cells too, but a considerable proportion is transformed into one or several secondary nuclei. These are, as a rule much smaller than the principal nucleus and are, therefore, called micronuclei [26]. Micronuclei have been observed in dividing cells of plants, insects and mammals, e.g. bone marrow [26], embryos [24], germinal cells [17], spleen [29], etc. Subsequent studies have shown an increase in the frequency of mn-Es in peripheral blood of mice exposed to chemical mutagens or ionizing radiation [7, 12, 25]. Micronuclei present in cytochalasin-B blocked binucleate peripheral lymphocytes also provide a reliable cytogenetic system for human monitoring. However, anucleated bone marrow polychromatic erythrocytes (PCEs) provide the most convenient cell type for rapid monitoring of environmental agents for clastogenic potential. In view of the wider acceptance and applicability of mn-PCEs as a rapid
S
” ” ” ” ” ” ” ” ” ” ” ” ” “ ” S-dependent ”
C‹
LI
! Interphase
S-lndpendent aqents
Prophase
Metaphase
Anaphase
Tel ophase
Fig. 1. A simple diagramatic representation of micronucleus formation in the bone marrow erythrocytes of mice.
measure of chromosomal damage, in recent years, a number of investigators have reported different protocols for treatment schedule and sampling time following mutagenic exposure of the animals [9, 11]. Likewise, several statistical models have been proposed to optimize sample size for adequate sensitivity and reliability of the micronucleus assay but this aspect remains experimentally unaddressed [1, 10, 23]. An attempt has been made to seek the influence of sample size viz., the number of animals, number of slides and number of PCEs per dose point on various parameters characterising the dose-response in mice exposed to relatively low doses of ionizing radiation.
Micronucleated Erythrocytes for Low-Dose Effects 171
Material and Methods
Foetal calf serum (SIGMA, USA), May-Grunwald powder (BDH, Poole, England), Giemsa (BDH, Poole, England), Euparal (Chroma Gesselschaft, GMBH and Co., West Germany) were purchased directly from the manufacturer or obtained through local dealers.
Inbred adult C57BI6 male mice, 10 to 12 weeks old reared in the animal house of this Research Centre were used. Animals, randomized into different groups based on parentage, age and body weight, had free access to food and water throughouttheexperimentandweremaintainedunderstandardconditions. Animals were irradiated with gamma rays using a Cobalt-60 teletherapy machine over a dose range of 0.0313, 0.0d25, 0.12•i0, 0.1875, 0.2500 and 0.3750 Gy at a dose-rate of 0.3214 Gy/min. Twenty four hours after irradiation
all the animals were processed for bone marrow smears.
Bone marrow smean were prepared and stained based on the initial method of Schmid (26] with minor modifications as reported elsewhere [6]. Animals were killed by cervical dislocation and both the femora removed and cleaned with cotton gauge from adhering muscle and tissues. Bone marrow was aspirated slowly into the foetal calf serum collected in 5 ml. centrifuge tubes with the help of 25 gauge needle. The bone marrow aspirate was centrifuged at 1000 rpm for 5 min. and the supernatant removed carefully with the help of Pasture pipette and mixed throughly. A small drop of the mixed bone martow aspirate was placed on the cleaned slide and smeared. Three slides were prepared from each animal.
Air dried bone marrow smears were stained in undiluted May-Grunwald for 5 min and subsequently, in diluted May-Grunwald (diluted with distilled water 1 : 1) for 3 min. Slides were rinsed in distilled water and stained in Giemsa solution (Giemsa stock solution one part and six parts distilled water) for 10 min. Slides were again rinsed in distilled water and blotted gently between the two layen of filter paper and cleared in xylene for 5 min and finally mounted in euparal.
Slides were coded and 500 or 1000 polychromatic erythrocytes (PCEs), which stain slightly bluish and the corresponding number of normochromatic erythrocytes (NCEs), staining golden yellow to pinkish in colour with or without micronuclei were counted at 1000X magnification under bright field in oil.
Data were analysed using a computer programme on NORSK DATA computer (Norway) for statitical significance. Mean values were calculated based on the frequency of micronuclei in individual mice. Regression analysis was carried out and the variability seen in observed and experimental values calculated for different sample size viz. number of cells scored, slides scored and number of animals per dose point. Differences were considered significant at 95% confidence limit.
Results and Discussion
5lides from two independent experiments were assigned to fourteen batches
based on the number of animals per group, number of slides and number of cells scored from each animal. The frequencies of micronucleated polychromatic erythrocytes (mn-PCEs) in fourteen different batches of control and irradiated mice are depicted in Table 1. The control frequency of mn-PCEs varied from 0.13 to 0.42’». The variaaon was seen among the batches where 500 PCEs per slide or 1000 PCEs per animal were counted. The variability was reduced when the number of cells was incresed to 1000 PCEs per slide or 9000 PCEs per dose point. Values of 0.259» in control were observed in batches XI, XII and XIV, which was very close to the overall mean of 0.247%. Similar to the controls, in the treated groups also there was more vańation in standard deviation (S.D.) when 500 PCEs perslides or 3000 total PCEs were scored for each dose point. However, when 1000 PCEs per slide or a total of 9000 PCEs per dose point were scored, the values of S.D. were quite low. In batches XI and XII which are based on 90Œ PCEs, the SDs were 0.04 and 0.02 respectively. In batch XIV, which is based on 18,000 PCEs the S.D. was found to be 0.03. Thus, it is evident that increasing the number of cells from 12000 to 18000 per dose point (6 mice, 3000 PCEs per mice] did not make any further noticeable difference. It is also, interesting to note that the lowest dose of 0.0313 Gy induced a significant increase in the frequency of mn-PCEs, an observation hitherto unreported.
The correlation-cœfficient, slope, intercept and standard error of estimates of the dose-response relationship of all the different batches are shown in Table 2. The correlation-coefficients vańed from a minimum of 0.941 in batch ł (3 animals, 500 cells per slide and a total of 3000 cells per dose point) to a maximum of 0.997 in batch XIII (six animals, 1000 cells per slides and a total number of 12,000 cells per dose point) and batch XIV (six animals, 1000 cells per slide and 18,000 cells per dose point). Similarly, standard error of the estimate was also minimum 0.041 for batches XIII and
XIV. Thus, the results show that scoring based on six animals, with 1000 PCEs each from two slides and a total of 12,000 cells per dose point as an adequate requirement to achieve reproducible and homogenous results, since scoring of cells beyond 12000 did not make any appreciable difference.
Because of its simplicity and versatality, mouse bone marrow micronucleus assay has been widely accepted and attempts continue to be made to improve the system based on biological and statistical considerations. Salamone et a1, have suggested steps towards improved micronucleus test. We have investigated the role of ôitferent moaes of treatment fi 1j. Studies have also been carried out to investigate the effect of single, double or tńple exposure on bone marrow and peripheral micronucleus assay [9]. MacGregor et al
[18] have also discussed guidelines for micronucleus assays in mammalian bone marrow erythrocytes. Like wise, several authors have discussed the statistical aspects of the micronucleus assay [10, 19, 21, 23, 28]. Very recently, Heddle et al [15] have reviewed the current status of micronuclei as an index of eytogenetic damage. Based on the guidelines recommended
Table 1. Effect of low-dose gamma radiation on the frequency of bone marrow micronucleated erythrocytes of C57Bl6 mice
Expt. No.
¥’CEs/ Total
MN/ 100 PCEs dose (Gy)
No. mice slide PCEs scored
| Cont. | 0.0313 | 0.0625 | 0.1250 | 0.1875 | 0.2500 | 0.3750 | ||||
| 1. | (3)‘ | 500(2)‘ | 3GD | 0.13 | 0.52* | 0.93*** | 1.01•* | 1.46*** | 1.76*** | 1.58** |
| S.D. | 0.11 | 0.15 | 0.07 | 0.35 | 0.22 | 0.35 | 0.44 | |||
| II. | (3) | 500(2) | 30fD | 0.26 | 0.52 | 0.73 | 1.04** | 1.04** | 1.40*** | 1.85*** |
| S.D. | 0.05 | 0.21 | 0.41 | 0.22 | 0.33 | 0.20 | 0.35 | |||
| flI. | (6) | 500(1) | 3OX1 | 0.42 | 0.68** | 0.72 | 1.01• | 0.97*** | 1.54** | 1.41** |
| S.D. | 0.07 | 0.11 | 0.46 | 0.38 | 0.12 | 0.46 | 0.54 | |||
| IV. | (3) | 500(3) | 45tXI | 0.24 | 0.54** | 0.87*** | 0.91** | 1.28*** | 1.62*** | 1.56*** |
| S.D. | 0.04 | 0.10 | 0.01 | 0.27 | 0.12 | 0.29 | 0.19 | |||
| V. | (3) | 500(3) | 45tXl | 0.28 | 0.61* | 0.72 | 1.13*** | 1.04** | l A2*** | 1.66*** |
| S.D. | 0.04 | 0.14 | 0.05 | 0.10 | 0.23 | 0.27 | 0.17 | |||
| VI. | (3) | 1000(2) | 6ØD | 0.21 | 0.46* | 0.83** | 1.07** | 1.30*** | 1.60*** | 1.54** |
| S.D. | 0.08 | 0.10 | 0.16 | 0.22 | 0.05 | 0.28 | 0.44 | |||
| VII. | (3) | 1000t2) | 6000 | 0.23 | 0.50*** | 0.66 | 0.94*** | 1.05*** | 1.32*** | 2.00*** |
| S.D. | 0.03 | 0.05 | 0.36 | 0.13 | 0.18 | 0.03 | 0.32 | |||
| VIII. | (6) | 1000(l) | 60£D | 0.32 | 0.54** | 0.69** | 1.04*** | 1.06*** | 1.42*** | 1.48*** |
| S.D. | 0.04 | 0.08 | 0.27 | 0.25 | 0.24 | 0.18 | 0.30 | |||
| IX. | (6) | 50 i(2) | 6000 | 0.18 | 0.52** | 0.83** | 1.03*** | 1.25*** | 1.58*** | 1.71*** |
| S.D. | 0.10 | 0.16 | 0.28 | 0.26 | 0.33 | 0.32 | 0.39 | |||
| X. | (6) | 500(3) | 9000 | 0.26 | 0.57** | 0.79** | 1.02*** | 1.16*** | 1.57*** | 1.61*** |
| S.D. | 0.04 | 0.12 | 0.30 | 0.22 | 0.21 | 0.26 | 0.17 | |||
| XI. | (3) | HD(3) | 9OOO | 0.25 | 0.49** | 0.77*** | 1.04** | 1.20*** | 1.56*** | 1.48*** |
| S.D. | 0.04 | 0.08 | 0.07 | 0.21 | 0.09 | 0.18 | 0.26 | |||
‹Contd)
Expt. No.
No. mice
PCEs/ Total slide PCEs scored
MN/ 100 PCEs dose (Gy)
Figures within parentheses denote ‘number of animals pér group and bnumber of slides scored per animal. Values marked with asterisk are significantly different from the control. *p < 0.5, **p < 0.01 and ***p < 0.001.
Table 2. Statistical parameters characterizing the dose-response based on different sample size
| Expt. No scored | No. mice | PCEs/ slide | Tout AEs | Corr. coeff. | Slope | Intercept | S.E. of esti. | |
| I. (3)‘ | 500 | (2) | 3000 | 0.941 | 0.235 | 0.256 | 0.178 | |
| II. (3) | 500 | (2) | 3000 | 0.972 | 0.247 | 0.011 | 0.124 | |
| III. (6) | 500 | (1) | 3000 | 0.919 | 0.173 | 0.028 | 0.156 | |
| IV. (3) | 500 | (3) | 4500 | 0.966 | 0.221 | 0.118 | 0.123 | |
| V. (3) | 500 | (3) | 4500 | 0.964 | 0.216 | 0.077 | 0.124 | |
| VI. (3) | 1000 | (2) | 6000 | 0.968 | 0.227 | 0.129 | 0.156 | |
| VII. (3) | 1000 | (2) | 6000 | 0.956 | 0.274 | — 0.111 | 0.176 | |
| VIII. (6) | 1000 | (l) | 6(J00 | 0.980 | 0.197 | 0.027 | 0.085 | |
| IX. (6) | 500 | (2) | 6000 | 0.995 | 0.241 | 0.131 | 0.048 | |
| X. (6) | 500 | (3) | 9000 | 0.987 | 0.219 | 0.092 | 0.076 | |
| XI. (3) | 1000 | (3) | 9000 | 0.970 | 0.214 | 0.092 | 0.113 | |
| XII. (3) | 1000 | (3) | 9000 | 0.971 | 0.252 | — 0.055 | 0.129 | |
| XIII. (6) | 1000 | (2) | 12000 | 0.997 | 0.251 | 0.008 | 0.041 | |
| XIV. (6) | 1000 | (3) | 18000 | 0.997 | 0.232 | 0.022 | 0.041 | |
Figures within parentheses denote the number of ‘animals per group and bslides scored per animal.
by different agencies in USA, Canada. EEC, OECD and Japan the authors have suggested to have an universally acceptable protocol on different aspects of micronucleus assay. Under this envisaged protocol, Heddle et a1 (15] have recommended the use of five animals of either sexes and at least 10,000 PCEs per dose point (alternatively 10IXl PCEs per animal). The present study designed to address the role of sample size with regard to the number of animals and the total number of cells per dose point, provide experimental evidence, interestingly in confirmity with the above attempts
[15] to universalize the protocol of the micronucleus assay.
Conclusions
The control frequency of mn-PCEs varied from 0.13 to 0.429o,respectively, in batch I (500 PCEs per slide, two slides each of 3 animals) and batch III (5tXI PCEs per slide, one slide each of 6 animals). Large variation was seen among batches when 500 PCEs were scored per animal. The results based on 500 PCEs per animal, even from 6 animals also exhibited large variability among control and treated groups. There was a significant increase in mn- PCEs even at 0.0313 Gy of gamma radiation. Performance of micronucleus test depends on sample size employed, results obtained during these investigations suggest the use of 6 mice and about 1000 PCEs per mouse for a reproducible and reliable bone marrow micronucleus assay in mice.
Acknowledgement
The authors wish to thank Shri B.L. Kulkami, M.R. Morwekar and S.P. Kamble for technical assistance.
REFERENCES
- Amphlett, G.E.and Delow, F. Statistical analysis of the micronucleus test, Mutation Res. 1984; 128: 161-166.
- Ashby, J., deserves, J.,Shelby, M.D., Margolin, B.H., lshidate, M. Jr. and Becking, G.C. Evaluation of shon-term tests for carcinogens, IPCS (UNEP/ ILO/WHO), Cambridge University, UK, 1988; pp. 1.145—1.230).
- Brodin, Statistical analysis of the micronucleus test—a modelling approach, Mutation Res.1989; 211: 259—269.
- Chaubey,C., Kavi, B.R., Chauhan, P.S. and Sundaram, K. Evaluation of the effect of ethanol on the frequency of micronuclei in the bone marrow of Swiss mice, Mutation Res. 1977; 43: 441—444.
- Chaubey,C., George, K.P. and Sundaram, K. X-ray induced micronuclei in the bone marrow erythrocytes of mice, Int. J. Radiat. B)ol.1978; 33: 507—510.
- Chaubcy, C., Kavi, B.R.. Chauhan, P.S. and Sundaram, K. The effect of hycanthone andmaleic hydrazide on the frequency of micronuclei in the bone marrow erythrocytes of mice, Mutation Res.1978; 57: 187—191.
- Chaubey, R.C., Bhilwade, H.N., Joshi, B. and Chauhan, P.S. Studies on the migration of micronucleatederythrocytes from bone marrow to the peripheral blood in irradiated Swiss mice, Int. J. Radiat. Biol.1992. (In press).
Micronucleated Erythrocytes for Low-Dose Effects 177
- Garriot, M. L. and Grahn, D. Neutron and gamma ray effects measured by micronucleustest, Mutation Lett.1982; 105 157—162.
- Gulati,K., Wajciechowski, J.P. and Kaur, P. Comparison of single, double or triple-exposure protocol for the rodent bone marrow/peripheral blood micronucleus assay using 4-aminobipheny1 and treosulphan, Mutation Res.1990; 234: 135— 139.
- Hart, J.W.and Engberg-Pedersen, Statistics of the mouse bone-marrow micronucleus test: Counting, distribution and evaluation of results, Mutation Res.1983; 111; 195—207.
- Hayashi, M., Sutou, S., Shimode, H., Sato, S., Sasaki, Y.F. and Wakata, A. Differencebetween intraperitonial and oral gavage application in the micronucleus test, The 3rd collaborative study by CSGMT/JEMS. MMS, Mutation 1989; 223: 329—344.
- Hayashi, Micronucleus test with mouse peripheral blood erythrocytes by acridine orange supravital staining: The summary report of the 5’th CSGMT/ JEMS. MMS. Mutation Res.1992; 278: 83—98.
- Heddle,A. A rapid in-vivo test for chromosomal damage, Mutation Res. 1973; 18: 187—190.
- Heddle,A., Hite, M., Kirkhart, B., Movoumin, K., MacGregor, J.T., Newell,
G.W. and Salamone, M.F. The induction of micronuclei as a measure of genotoxicity, A Repon of the U.S. Environmental Protection Agency Gene-Tox Program, Mutation Res.1983; 123: 61—118.
- Heddle, J.A., Cimino, M.C.,Hayashi, M., Romagne, F., Shelby, M.D., Tucker, D., Vanparys, P.H. and MacGreger, J.T. Micronuclei as an index of cytogenetic damage : Past, Present and Future, Environ. Molecular Mutagen 1991; 18: 277— 291.
- Kliesch,, Danford, N. and Adler, I.D. Micronucleus test and bone marrow chromosome analysis, A Comparision of two methods in vivo for evaluating chemically induced chromosomal alterations, Mutation Res.1981; 80: 321—332.
- Lahdetie, and Pervinen, M. Meiotic micronuclei induced by X-rays in early spermatids of rat, Mutation Res. 1981; 81: 103—115.
- MacGregor, J.T.,Heddle, J.A., Hite, M., Margolin, B. , Ramel, C., Salmone, M.F., Tice, R.R. and Wild, D. Guidelines for the conduct of micronucleus assay in mammalian bone marrow erythrocytes, Mutation Res.1981; 189: 103— 112.
- Mackey,E. and MacGregor, J.T. The micronucleus test: Statistical design and analysis, Mutation Res. 1979; 64: 195—204.
- Margolin,H., Collins, B.J. and Malson, J.M. Statistical analysis and sample size determination for mutagenicity experiments with binomial responses, Environ. Mutagen. 1983; 5: 705-716.
- Margolin, H. and Risko, K.J. The analysis ofin vivo genotoxicity data: Case studies of the rat hepatocytes, UDS and mouse bone marrow micronucleus assay in: J. Ashby, et al., (Eds.) Evaluation of short-term tests for carcinogens, Report of lPCS’s collaborative study on in vitro_assays, Cambridge University Press, Cambridge, 1987; pp. 1.29—1.42.
- Matter,E. and Wild, D. Mutagenicity of selected chemicals in the mammalian micronucleus test, in: FJ. deserves and MD. Shelby (Eds.) Comparative Chemical Mutagenesis, Plenum Publishing Corporation, New York, 1981; pp. 657—680.
- Mitchell, deG. and Brice, A. J. Investigations into parametric analysis of data in in vivo micronucleus assays by comparisions with non-parametric methods, Mutation Res. 1986; 159: 139—146.
- Molls, M., Streffer, C. and Zamboglou, Micronucleus formation in preimplanted mouse embryos cultured in vitro after irradiation with X-rays and neutrons, Int.
- Radiat.Biol. 1981; 39: 307—314.
- Schlegel, R.and MacGregor, J.T. A rapid screen for chromosomal damage in mice. Accumulation of circulating micronucleated Mutation Res. 1983; 113: 481—487.
- Schmid, W.Chemical mutagen testing on_in vivo somatic mammalian cells, Agents and Actions 1973; 3 (2): 77—85.
- Schmid, W. The micronucleus test for cytogenetic analysis in: A.Hollaender (Ed.) Chemical Mutagens, Vol. 4 Plenum Publishing Corporation, New York, 1976; pp 31—53.
- Snedecor G.W. and Cochran, W.C. Statistical methods, 6th Edn., Iowa State Press,Ames, I.O. 1967; pp 223—226.
- Tates, A.D., I. Neuteboom M., Hofker and den Engelse, L. A micronucleus techniquefor detecting clastogenic effects of mutagens/carcinogens (DEN,DMN) in hepatocytes of rat liver in vivo, Mutation 1980; 74: 11—20.
- A ComputerSoftware Package for Determining Statistical Significance of Radiation Effect in High Background
Radiation Area
M.B. Yadav, Satya Jyothi and Hari Singh
Nuclear Materials Accounting Section, Fuel Chemistry Division Bhabha Atomic Rescarch Centre, Bombay, India
In the study of the radiation effect in High Background Radiation Area (HBRA) one needs to deal with large sample sizes of N and N2 located respectively in HBRA and in Controlled Area (CA) with the observed numbers of abnormalities in the two samples being • and •2 These variables, x1 and x2. follow Poisson’s distribution. Two questions arise now:
- Whetherradiation effect in HBRA is significant?
- Howto test the statistical significance of the radiation effect in HBRA?
In order to answer these questions, a computer software package MUTAN in FORTRAN 77 language compatible with Personal Computers (PCs) has been developed. The algorithm used in MUTAN is simple, fast and based on exact calculations without any approximations being made.
Introduction
Radioactivity in the environment whether naturally occurring or otherwise may cause hazards to human lives. Study of radiation effcct in such areas termed as High Background Radiation Areas (HBRAs) is a subject of importance in radiobiology. Comparison of frequencies of occurrence of an ailment with or without a stress factor in this respect is of interest. Studies of this nature are carried out by scientists and doctors and a prominent example is the effect of smoking cigarettes on the incidence of cancer. It is important to know whether the effect caused by these sourccs is significant or not particularly with regard to its effect on human lives. The objective of this paper is to develop a simple procedure to test the statistical significance of these effects. One such test procedure has been given in the form of tables by [3] but it has limited scope with respect to the parameters of the test. The test procedure suggested in this paper does not have any restriction on the parameters of the test.
Description of the Problem
In the study of the radiation effect in HBRA one needs to deal with large sample sizes of N and >2 located in HBRA and in CA and observe the
number of abnormalities xt and z¿ in the two samples respectively. If we imagine large number of studies in this kind, the variables z; and •2 are expected to follow Poisson distribution with parameters and estimated
by x lN and zJN2 The problem reduces to testing of the null hypothesis Jo: Hi = , i.e. the rate of abnormalities in HBRA is the same as that in the CA against the alternative hypothesis ftp: 1; > , i.e., the rate of abnormalities in HBRA is more than that in CA. If we restrict to those samples of sizes N and N2 for which zt + •2 – n is constant then it can
be shown [4] that under and for the given value of n the conditional
probability mass function (p.m.f.) of the variable x is given by
where p = N f{N + N2)
This is a binomial probability mass function with parameters n and p. For the given values of n = x› • •2. and zt = c, the probability of observing c or more abnormalities from HBRA out of the total n abnormalities observed from both the populations is given by
The problem now reduces to one of testing the null hypothesis ftp: — against the alternative hypothesis Us:r >Phwithparameters a = total number of abnormalities observed in the two samples and p, the expected proportion of abnormalities from HBRA if we corribined the two populations together and Ph = N !/(Ni + N ) he preassigned level of significance is n, then the null hypothesis of equality of the abnormalities in the two populations will be rejected if xt > c, where c is the smallest integer such that
n
Z ‘r p’(I — PM”°” —
In this context c is called rejection number for the critical region of size not larger than a (level of significance).
Thus the problem now reduces to one of finding the smallest number c
of success (or failure) in n independent Bemoulli trials such that
r ——c
where p is the probability of success (or failure) in a trial. Standard tables of the cumulative binomial probabilities available in literature [5, 6, 7] are arranged in such a way that they can be of help in determining the values of alpha for specired values of c, (1 y c y n) and p, (0 y p y 1.0). A mani- pulation is required to get the value of c for specified values of n, p and a. Any mistake in the manipulation will lead to a wrong rejection or acceptance
of the hypothesis. Also these tables are available only upto n = 1000 in steps of 1, 2, 5, 10 and 50. Getting a value of c directly for given values of n, p and alpha would be of great help to persons working in this field. One such table available in literature [2, 3], is such that the values of c are available for n = I [1] 200 and p — 0.01 (0.01) 0.99 and alpha = 0.01, 0.05 and 0.10. For sample sizes larger than 200 no such table is available. In these cases one has to go for the normal distribution approximation [1] which may lead to a wrong or unsound conclusion (Examples 1 and 2). Exact values are always better than any approximation if they are easily available.
Development of a Computer Software Package
A computer software package MUTAN in FORTRAN 77 language compatible with Personal Computers (PCs) has been developed in order to find the minimum number c of success or (failure) in n independent Bemoulli trials such that
where p is the probability of success (OF failure) and 0 ñ o S 1 is a specified level of significance. c is called ’REJECTION NUMBER’ for a critical region of size not larger than alpha.
The software is applicable for any n k 1 and 0 p y 1 and ‹x = 0.01,
0.05 and 0.10. The necessary parameters for MUTAN are n (= x› + • ) and
(= N ytN + N2))•
The values of ii and p will be evaluated in the program itself. The user has only to supply the values of N , N2. l and 2 following the prompts appearing on the terminal. If the observed number of success (or failure} (number of abnormalities in the HBRA in the present problem) is less than c (the rejection number for a particular alpha), the hypothesis will not
be rejected and if it is greater than or equal to c, Hz will be rejected at that level of significance. The algorithm used in MUTAN is very fast and does not assume any approximation at any stage.
Advantages of Mutan Over Normal Approximation
- Supposewe wish to test the hypothpesis : –– . against H : p >
Let p be estimated from a large sample of size ii, where r number of success (or failure) were observed. To test this hypothesis, one performs ‘the following calculations in case of the normal approximation to the Binomial distribution. First find p — (r/n), the standard error with p is given by Se(p) = ? [p(1 — p)/n). In the normal approximation y is assumed to be normally distributed with mean = tnpfn) — p and variance = p(l — p)/n. The z (score) is given by (p – )/! ( ). Then
compare this z (score) with the z (score) of a normal distribution at
a given level of signir‹cance alphaa. If this z (score) is greater than or equal to the r (score) of the normal distribution, reject thc null hypothesis Off, otherwise Off is not rejected. Thus this approach involves calculations, lot of time and a need for trained personnel. The present software package requires no special training on the part of the user. It requires only the values of N , N2. <t and <2 to be typed.
- Normal approximationis just an approximation, whereas this software
package gives values based on exact methods of calculations.
- Normal approximation may notalways give the correct decision at all levels of significance as is evident from examples l and 2 of Sections S. The r (score) in example l is 1.33 and that in example 2 is 1.32. Both these values are greater than 1.28, the z (score) for the normal distribution at n= 10 and for one-tailed test. This suggests that the null hypotheses Hg in both the examples be rejected at 10% level of significance. However when MUTAN is applied to solve these problems, it is shown in examples l and 2 that the null hypotheses are not rejected at a = 0.10.
- Inth5 present age of computers it is preferable to use exact methods of calculation to approximate methods if the former are easily
Application of Mutan
Example 1. An experiment was conducted to study the effect of radiation in a HBRA on the new born babies in the surrounding population. Out of N children from HBRA • abnormalities were reponed, whereas out of N z
children from controlled area xc abnormalities were observed. The experimenter
wanted to know whether the effect due to the high background radiation is
significant or not. MUTAN is very useful in this type of problems where one has to type only the values of Nt. >2. • and •2 on the terminal to get the answers.
If No• 289, x — 144, N2 2996 and x2 – 1369, the values of n, p and c (rejection numbers) obtained by MUTAN for a 0.01, 0.05 and 0.10 are as follows:
The value of n = 1513 and the value of p — 0.088
At a = 0.01 Rejection number = 160
At n = 0.05 Rejection number = 152
At n = 0.10 Rejection number = 148
This Shows tfiat the null hypothesis <0 of equalities of rate of normalities in the two population (i.c. HBRA and CA) is not rejected at all the three levels of significance because the observed number of abnormalities in HBRA namely 144 is less than all thcse values of c viz., 160, 152 and 148.
Example 2. In an experiment involving induction of mutations in mice by two different types of radiation, let < be the mutations among a large number of offspring N of males subjected to radiation of types i (i = 1, 2),
and let and be the true and unknown mutation rates in the respective groups. The experimenter wishes to test the hypothesis : = i.e. the mutation rate is the same for both types of radiation, against the alternative Its : > that is the mutation rate is higher in group 1. If N — 262770, – 69,’N2 15,
the values of ri, p and c (rejection numbers) obtained by MUTAN for tr =
G.01, 0.05 and 0.10 will be displayed on the terminal as follows.
The value of n = 84 and the value of p — 0.76
At n = 0.01 Rejection number = 74
At e = 0.05 Rejection number = 71
At H = 0.10 Rejection number = 70
The observed value of mutations in group 1 is d9 and hence JJ is not rejected at n = 0.01, 0.05 and 0.10.
Other Possible Applications of Mutan
Other possible uses of MUTAN are in the studies of:
- Smokingcigarettes, chewing of betel nuts, tobacco , intake of drugs such as opium, hashish and heroin have deleterious effects on health affecting the heart, the circulation and respiration system, the central nervous system and local tissues causing cancer. In studying these effects one has to study large population of patients who have been smokers or tobacco users or non smokers and non users of tobacco.
- In the field of Medical Science where effects of two systems of treatmentsfor curing the same disease have to be
- In the studies of radiation effects inHiroshima and Nagasaki or Chernobyl types of accidents
- In thestudies of census data, such as rate of morialities of children of ages below S years, social and educational status of females in rural and urban
Acknowledgement
The authors warmly acknowledge the active support and help received from Dr. D.D. Sood, Head, Fuel Chemistry Division, during the course of this work and offer their grateful thanks to Dr. S. Vaidyanathan, Head, Nuclear Materials Accounting Section for going through this paper carefully and critically. Thanks are also due to Dr. S.K. Mahajan, Head, Microbiology and Agriculture Division, BARC who posed a similar problem to the authors and this paper is an outcome of their attempts to solve it.
REI•ERENCES
- Bland, An Introduction to Medical Statistics, Oxford Medical Publications,
1987.
- Kastenbaum,A. and Bowman, K.D. “The minimum number of successes
in a Binomial Sample.” ORNL-3909, Oak Ridge National Laboratories, Tenn. Feb. 1960.
- Kastenbaum, M.A.and Bowman, K.D. “Tables for determining the statistical significance of mutation frequencies”, Mutation Research 1970; 9: 527—549.
- Kastenbaum, M.A.and Bowman, K.O. The minimum number of successes in a Binomial Sample. ORNL-3909, Oak Ridge National Laboratories, Tenn. Feb. 1960.
- Kendall, and Stuart, A. The Advanced Theory of Statistics, Vol. 1, Charles Griffin and Company Limited, London & High Wycombe, 1977.
- NBS Tables of the Binomial Probability Distribution, National Bureau of standards, Applied Mathematics series 1950; 6: Washington 25, DC, US Government Printing Office.
- ORDP Tables of the Cumulative Binomial Probabilities, Ordinance Corps Pamphlets ORDP 20—1, US Government Printing Office Washington, Sept. 1952.
20. Health Impact Assessment of Indoor Radon Levels in Some High Background
Areas in India
M.C. Subba Ramu
Environmental Assessment Division, Bhabha Atomic Research Centre, Bombay, India
Introduction
Radioactive materials became part of the earth at its very formaion. There is radioactivity in the air we breathe, the food we eat and the dwelling we live in. Even man himself is slightly radioactive since all living tissues contain traces of radioactive substance. To this ubiquitous natural radiation, man has started adding his own, which arises out of the technological activities. It is therefore quite natural that we should assess the effect of the natural components of radioactivity for understanding the effect of man’s contribution which at present is a very small fraction of the total dose received by humans. For example an Indian on an average receives 2490 microSv/yr, out of which 97.7% is contributed by natural sources, 1.93 to by medical sources, 0.07’7o by miscellaneous sources and 0.3’7o by nuclear activities. The contribution from nuclear fuel cycle is 0.005% which is equivalent to the dose resulting from 30 minutes exposure to average natural background or 10 minutes exposure to the background areas in Kerala f7].
The total annual dose to an individual in India, from natural sources
works out to be 2433 microsv, out of which 734 microsv (30’7») is from external exposure and 1699 microsv (70%) is from internal exposure. Of the latter, 1370 microsv is from Rn, Tn and their daughters (progeny) and 15 microsv and 195 microsv are from cosmogenic and terrestñal radionuclides respectively. Thoron (Tn) contribution accounts for about 5 to 10’7» except in Kerala, where the monazite sands are expected to give rise to comparatively higher Tn and its short lived daughter products in some dwellings. Out of the dose of 1370 microsv from Rn and Tn and their short-lived daughters, 1210 usv is from Rn and its short lived daughters alone and even in this 1160 microsv is from Rn and its short lived daughters present in dwellings. Thus Rn and its short-lived daughters present in indoor atmosphere are mainly responsible for a major contribution to the total radiation dose to man.
Rn gas seeps out of the earth all over the world into both outdoor and indoor air. The dose from the inhalation of the indoor short-lived Rn daughters varies with time. It also depends on the building material used for constniction and the ventilation system. Natural ventilation in Indian dwelling can be
expected to reduce the indoor Rn and its progeny levels. However, if the emanation rates are very high, Rn and its progeny levels in dwellings can be expected to be higher. The level also depends on the building materials used, geographical locations and the geological conditions of the soil. Depending on the survey carried out by the Atomic Minerals Division, some of the areas in India have been outlined as high background areas as regards the soil uranium and radium content. Houses in these areas can be expected to have elevated levels of Rn and its progeny. This article presents the summary of the results of the measurements of indoor Rn and its progeny in the above high background areas and the associated health impact assessment.
Health Effects
In radiological protection, emphasis is placed on limiting the exposure to man made radiation. How one should deal with exposure to natural radiation is less clear. Direct risk information does not exist for environmental levels of Rn progeny. It is therefore nccessary either to extrapolate from data for occupational exposure or to infer the same from the incidence of lung cancer in the wider population. Linear extrapolation from the dose-response values of the uranium miners exposed to higher Rn levels suggests that majority of lung cancer amongst the public could occur due to Rn and its progeny present in their dwellings. The most conservative upper estimate of life-time risk to the general population is 100 deaths from lung cancer for a collective exposure of 10 WLM [2]. This factor is supported by indirect evidences of lung cancer incidence analysis and lung dosimetry in the United States of America.
“Emanatoria” is a place in Austria, where persons suffering from disease like arthritis, cardiovascular disease, disorder of locomotor system, diseases of nervous system etc. are made to breathe Rn and its short-lived daughters. The spring waters in these “Emanatoria” contain a very high concentration of radium in them, varying between 4,000 and 26,000 Bq/ltr, which is 106 times more than what is encountered normally in many of the spring waters.
“Stimulation of gro›vth” or “Hormesis” response of biological species such as plants and animals have been reported. Results of some published animal experiments indicate that the low level ionising radiation is beneficial for the growth and development, fecundity, resist,.nce 😮 subsequent I:arm5ui doses of radiation, resistance to infection, resistance to cancer induction and increased average life-span (5].
For health impact assessment one needs to know the concentrations of indoor Rn and its progeny levels and the data of risk estimates. Studies carried out elsewhere has shown that a substantial number of cancer cases have been found in relatively small segment of population exposed to levels higher than the averages [13]. Hence the results of the measurements carried
out by us in some high background areas have been considered for assessment in this paper.
Measurements
A passive detector technique using solid state nuclear track detectors (SSNTD), which gives the time integrated concentrations in a dwelling has been used for carrying out the measurements. This involves exposing small strips of
2.5 cm x 2.5 cm size LR-115 Type II detectors in the bare mode in the dwelling to be monitored [10] for a period of 90 days. After the period of exposure the detectors were retrieved and were etched in a suitable chemical etchant and the recorded tracks were scanned using an optical microscope. The recorded tracks were converted into potential alpha energy concentrations (PAEC) in WL units using a calibration curve generated by doing controlled experiments [9].
Results
Measurements were carried out in about 15 different locations outlined as high background areas by the Atomic Minerals Division of the DAE. About 335 individual exposures were made to measure the Rn daughters concentrations. Estimated PAEC varied fTOm 1.5 mWL at dwellings in Udaipur to 21.9 mWL at dwellings in Godhra, Gujarat state and the geometric mean was 9.4 mWL with a geometric standard deviation of 1.9. Measurements carried out in the high background areas do not include the monazite areas of Kerala due to the reason that the location consists of high deposits of thorium. For the measurement of Tn and Rn PAEC in WL units an SSNTD method is being developed in out laboratory although preliminary calibration studies using a somewhat elaborate system have been carried out. Measurements carried out in the normal background areas spread all over the country [11] gives the PAEC varying from 1 mWL to 15.0 mWL with a mean of 4.2 mWL.
Risk Estimates
So far no epidemiological study among the population group exposed to enhanced levels of Rn progeny indoors are available to evaluate directly the lung cancer risk associated with Rn progeny inhalation. Hence the best estimate by Evans et al [3] of 100 deaths from lung cancer for a collective exposure of 106 WLM is used for risk estimates. It should be mentioned here that this is an upper estimates of life-time risk.
It has been estimated that the population in the high back ground areas where the indoor Rn measurements were made is 13.4 million [1]. Using the above risk factor, the total number of lung cancer in this areas works out to be 150 cases per year. The Indian Council of Medical Research (ICMR) has made an estimate of 700 cancer cases per year per millions of the Indian population 8]. On the basis, it can be estimated that the cases
of lung cancer attributable due to the breathing of Rn and its daughter products in high background areas in India is 1.60% of the total cancer cases in those areas estimated by ICMR. Population exposed to indoor Rn and its daughters products in normal background areas is estimated to about 4 times higher. On this basis the fraction of lung cancer cases in India attributable to the breathing of Rn and its daughters of 4.2 mWL works out to be 0.72% of the total cancer cases.
Hormesis which suggests that ecologically realistic low levels of ionising radiations may be beneficial to humans, has not been scientifi“cally quantified, although some evaluations have been made in USA and Japan [4].
Studies made in Kerala where the average external radiation levels is 400 microsv per year with peak values of 32,500 microsv per year in some locations, have not shown any health hazard to human population (3]. It may be concluded that ionising radiation is not damaging at the dose levels recorded above.
Studies correlating the external natural radiation doses to the population with the cancer data available for five cities and 14 states in India shows an inverse correlation. Where the radiation level is greater, cancer risk is invariably less (6].
Comments
The measurement data of the Rn progeny levels in high background areas in India show that out of about 13.4 million persons living in this area, the total number of lung cancer cases attributable to Rn progeny inhalation is only 150. This is too small a figure to be considered for a statistically valid analysis. It is difficult to deduce any reasonable conclusion after taking into consideration the variations that occur in the Rn progeny levels from place to place. The confounding etiological factors further add to difficulty in asessing the exact role of environmental radon.
While the harmful effects of low level radiation cannot be proved beyond doubt, it is also not possible to say with confidence that low level radiation is beneficial to humans. ALARA principle should still be the guiding spirit in matters of radiation protection.
Acknowledgements
The author wishes to thank Dr. D.V. Gopinath and Dr. K.S.V. Nambi for their keen interest in the study and to Dr. T.V. Ramachandran and T.S. Muraleedharan for their help in the preparation of the manuscript.
REFERENCES
- Census,Provisional Population Totals: Paper-2 of 1991 Census of India 1991, Register General & Census Commissioner, Ministry of Home Affairs, New Delhi.
- Evans,D., Harley J.H., Jacobi, W., Mclean A.S., Miles, W.A. and Stewart,
C.G. Estimation of risk from the environmental exposures to radon-222 and its decay products, Nanire, 1981 290: l2th March, 99—101.
- Gopal Ayengar A.R., Sundaram K.,Mistry K.B., Sunt, C.M., Nambi K.S.V., Kathuria, P., Basu, A.S. and David, M. Evaluation of long term effects of high background radiation on selected population groups on the Kerala coast, In. Proc, of 4th lnte. Nat. Conf. on peaceful uses of Atomic Energy, Geneva (IAEA), 1971: Vol. 11, 31-51.
- Hickey,J., Clelland R.C., Bowers E.J. and Zemae, B.S. Possible human health benefits of radiation hormesis, In: Conference on Radiation Hormesis. 14-16th August 1985.
Luckey, T.D. Hormesis, Nuclear News, 1981; Dec. 48—55.
- Nambi,S.V. and Soman, S.D. Environmental Radiation and cancer in India. Hlth. Phys. 1987; 52: 653—657.
- Narayanan,K., Kirshnan, D. and Subba Ramu, M.C. Population exposure tp ionising radiation in India, ISRP (k)-BR-3, ISRP, Kalpakkam Chapter, 1991.
- National Cancer Registry, 1982, Annl. Report 1982, New Delhi, Indian Council of Medical Research.
- Subba Ramu, M.C.,Muraleedharan, S. and Ramachandran, T.V. Calibration of solid state nuclear track detectors for the measurements of indoor radon levels in dwellings, Sci. of Tot. Envir, 1988; 73: 245—255.
- SubbaRamu, C., Ramachandran, T.V., Muraleedharan, T.S. and Shaikh, A.N. Indoor levels of radon daughters in some high background areas in India. Rad. Prof. Dosim, 1990a; 30: 41M5.
- Subba Ramu, M.C., Ramachandran, T.Y., Muraleedharan, T.S. Shaikh, A.N. Measurements of indoor radon workinglevels in Indian dwellings using passive detector techniques, in 8th Sym. on Radiation Phys (NSRP-7), 1990b; Jan. 17—19: 143—147.
- Ujeno, Relation between cancer incidences or mortality and external natural background radiation in Japan. Biological effects of low level radiation. 1983; 253—262. Vienna, IAEA.
- United Nations Scientific Committee on the Effect of Atomic Radiation,Report to General Assembly, Ionising Radiation: Sources and Biological Effects., United Nations, 1988.












![Creating an Audience: Social Implication of Aesthetic Experience[i]](https://sciphiweb.com/wp-content/uploads/2025/03/Creating-an-Audience-Social-Implication-of-Aesthetic-Experience-100x100.jpg)



Leave a Comment
Your email address will not be published. Required fields are marked with *