Radiation Sensitizers A Contemporary Audit Other Books of Interest Adaptation Biology and Medicine (Volume 1: Subcellular Basis) B.K. Sharma et at
Radiation Sensitizers
A Contemporary Audit
Other Books of Interest
Adaptation Biology and Medicine (Volume 1: Subcellular Basis)
B.K. Sharma et at
Adaptation Biology and Medicine (Volume 2: Molecular Basis)
K.B. Pandolf et al
Auditory Evoked Responses in Clinical Practice
Anil Malhotra
Biomaterials
S.V. Bhat
Brain Protection and Neural Trauma Diarrheal Diseases: Research Perspectives
V.K. Khosla et at
N.K. Ganguly anal N. Appaji Rao
Essentials of Clinical Toxicology Immunomodulation
S.B. Lall
S.N. Upadhyay
Immunopharmacology
S.N. Upadhyay
Introduction to Rational Use of Drugs
R.R. Chaudhury and C.D. Tripathi
Liver and Environmental Xenobiotics Medical Diagnostic Techniques and Procedures
S.V.S. Rana ‹md K. Taketa
Medical Diagnostic Techniques and Procedures
Megha Singh et al
Multi-Drug Resistance in Emerging and Re-Emerging Diseases
R.C. Mahajan wid Amu Therwath
Myasthenia Gray is
P. Christndoss
Physiological Fluid Dynamics—III
N.V.C. Swimy turd M. Singh
Radiobiological Concepts in Radiotherapy
D. Bhattacharjee and B.B. Singh
Rapid Eye Movement Sleep
B.N. Mallick arid S. Inoué
Recombinant and Synthetic Vaccine
G.P. Talwar et al
Reproductive Immunology
Satish K. Gupta
Clinical Studies
The antitumor antibiotic MMC has shown cli nical activity in a number of cancers, including stomaCh Cilncer, breast cancer cervical cancer and cancers of the upper aerodigestive tract. Due to its bone marrow toxicity after daily treatments the drug was abandoned by many medical oncologists. After the introduction of an high-dose intermittent schedule the interest in this drug again was revived.
The interest of MMC as an adjunct to radiation therapy started in 1974 after the publication by Nigro et al. on combined treatment of squamous cell cancer of the anus (Nigro et al. 1974). MMC was combined with a continuous infusion of 5-Fluorouracil and proved to be highly effective when combined with radiotherapy in these cancers. It has completely changed the treatment standard in anal cancers predominantly in the United States. In Europe, treatment of anal cancers has always had major influence by the radiation oncologist (Papillon and Montbarbon 1987). Clinical studies have shown that MMC and 5-FU-chemotherapy should not be omitted from this treatment regimen. Both randomized and non- randomized studies have shown significantly higher local tumor control after treatment when MMC was given in combination with 5-Fluorouraci1 and used with simultaneous radiotherapy.
The UKCCCR Anal Cancer Trial published their results in 1996. The trial was designed to compare combined modality therapy with radiotherapy alone in patients with epidermoid anal cancer. In their study 585 patients were eligible for analysis. All patients were treated with 45 Gy radiotherapy in 20 or 25 fractions over 4—5 weeks. Chemotherapy consisting of MMC (12 mg/sqm on day 1) and 5-FU (1000 mg/sqm/24 hours for 4 days or 750 mg/sqm/24 hours for 5 days) by continuous infusion during the first and last weeks of radiotherapy was administered in 295 patients. Clinical iesponse was assessed 6 weeks after completion of therapy. Good responders went on to boost radiotherapy whereas poor responders were recommended to undergo salvage surgery. After a median follow-up of 42 months, the rate of local failure was 59% following radiotherapy alone, compared to 36% in patients after combined radio-chemotherapy. This represents a
28 DOBROWS KY
469c reduction in risk of local failure (p < 0.0001). The risk of death from anal cancer was also reduced following combined radio-chemotherapy (p — 0.02), overall survival was not statistically different in the two groups, early toxicity but not late toxicity was significantly higher after combined therapy. The authors conclude that MMC, 5-FU and radiotherapy should be used as standard therapy in epidermoid cancers of the anus (UKCCCR Anal Cancer Trial Working Party 1996).
An other trial, performed by the EORTC, evaluated the results from a randomized study in 110 patients treated with advanced anal cancer in the years 1987 to 1994. Patients were randomized to receive radiotherapy alone (45 Gy in 5 weeks) and 15–20 Gy boost after 6 weeks in case of complete or partial response, respectively versus the same radiotherapy with additional administration of MMC (15 mg/sqm on day 1) and 5-FU (750 mg/sqm/24 hours as continuous infusion days 1–5 and 29-33). This study also showed a benefit for patients who had received additional chemotherapy (complete remission after combined therapy 80% versus 54% after radiotherapy alone). This led to a statistical significant benefit regarding local tumor control and colostomy-free interval (p —- 0.02 and p — 0.002, respectively). The locoregional control rate improved by 18% and the colostomy-free interval increased by 32% at 5 years following combined radio-chemotherapy. No increase in severe side effects were seen comparing the two group. It was concluded that concomitant use of radiotherapy and chemotherapy with MMC and 5-FU resulted in a significant improvement for the patients regarding locoregional control and reduction in the need for colostomy, without a significant increase in late side effects (Bartelink et al. 1977).
At the Princess Margaret Hospital a series of 110 patients with epidermoid cancer of the anal were treated by prospectively designed non-randomized split course trials of radiotherapy with infusion 5-FU administration with or without MMC (Cummings et al. 1993). The addition of MMC was associated with improved tumor control rates (87% versus 58% at 4 years, p — 0.005) and improved 4-year actuarial cause-specific survival (80% versus 64%, p — 0.02). In this series as in above randomized trials, there was no long-time toxicity attribute to MMC alone.
A possible cause for the efficacy of this specific drugs might lie in the supraadditive interaction found in vitro and in the differential toxicity of MMC, being more toxic in radioresistant hypoxic tumor cells (Dobrowsky et al. 1992).
In other gastrointestinal cancers e.g. colorectal recurrent cancers, the combination of MMC, 5-FU and concomitant radiotherapy has not been of major benefit. Both studies with bolus and infusion 5-FU (with MMC and radiotherapy) have caused severe local and systemic toxicity and
Radiation Sensitizers
A Contemporary Audit
Editors
Nagraj G. Huilgol
C.K.K. Nair
V.T. Kagiya
This edition is authorised for sale in
India, Pakistan, Bangladesh, Nepal,
Bhutan, Sikkim and Sri Lanka only.
Narosa Publishing House
New Delhi Chennai Mumbai Calcutta
Dr. Nagraj G. Huilgol
Chief, Division of Radiation Oncology
Nanavati Hospital and Medical Research Centre Mumbai-400 056, India
Dr. C.K.K. Nair
Radiation Biology Division Bhabha Atomic Research Centre Mumbai-400 085, India
Prof. V.T. Kagiya
Chairman, HRF
Kinki Invention Centre Yoshida Kawahara.cho 14
Sakyo-ku, Kyoyo, 606 8305, Japan
This book is published for the Society for Cancer Research and Communication (SCARC), P.O. Box 8094, Mumbai-400 056, India
Copyright 0 2001 Narosa Publishing House
N A R O S A P U B L I S H I N G H O U S E
22 Daryaganj, Delhi Medical Association Road, New Delhi 110 002 35—36 Greams Road, Thousand Lights, Chennai 600 006
306 Shia Centre, D.B.C. Sector 17, K.U. Bazar P.O., Navi Mumbai 400 705 2F—2G Shivam Chambers, 53 Syed Amir Ali Avenue, Calcutta 700 019
All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without the prior written permission of the publishers.
All export rights for this book vest exclusively with Narosa Publishing House. Unauthorized export is a violation of Copyright Law and is subject to legal action.
ISBN 81-7319-376-2
Published by N.K. Mehra for Narosa Publishing House, 22 Daryaganj, Delhi Medical Association Road, New Delhi 110 002 and printed at Replika Press Pvt. Ltd., Delhi 110 040.
Preface
Cancer still remains as a major cause of morbidity and mortality, taking a heavy toll of human population in the developed world in spite oi several decades of clinical research and trials of variety of new and promising therapies. Use of ionizing radiation in treatment of cancer quickly followed the discovery of X-rays and radioactivity at the dawn of’ the last century. The limited success of radiotherapy has been attributed to several factors of which the intrinsic radio resistance of tumor cells, their fast repopulation kinetics, and tumor hypoxia are the major causes. The problems of fast repopulation kinetics of tumor cells are circumvented by continuous hyper fractionated accelerated radiation therapy (CHART). Several strategies were worked out and many of them have been tested in clinical trials for overcoming tumor hypoxia. Hypoxic cell radiosensitizers were shown to be very effective in enhancing tumor radiosensitivity in several studies. Hypoxia has been reported to be a cause of failure in accelerated fractionation in radiotherapy where there is lack of reoxygenation. As a result hypoxic cell radiosensitizers could have a greater effect with accelerated rather than conventional fractionation. Indeed, encouraging tumor responses have been reported in patients receiving hypoxic cell radiosensitizer with every radiotherapy fraction. Since 1970s multicentered clinical trials were undertaken for a number of compounds particularly nitroimidazoles for their hypoxic cell radio sensitizing property in radiation therapy. However, some of the major clinical trials were inconclusive and some trials even did not reveal any benefit to the patients. As tumor hypoxia is one of the major impediments in radiotherapy of cancer, laboratory and clinical research on hypoxic cell radiosensitizers continued with increasing interest. Since 1989 international co-coordinated research program of clinical study on chemical modifiers of cancer treatment using senazole (AK-2123) has been undertaken by a number of groups in several countries. Senazole is a nitrotiazole compound and is less toxic than the nitroimidazoles in vivo. It effectively sensitized the radiotherapy of different malignancies such as head and neck, nasopharynx, esophagus, lung, colorectal, rectum, bladder, breast, uterine cervix, endometrium etc. Development of target selective radiosensitizer with higher sensitizing ability is a new area of research worth exploring. Suitably modifying the groups or side chainssensitizers can be targeted to different cellular locations. Hypoxic cell sensitizers have had a chequered past and now face an uncertain future. The knowledge of hypoxia leading to radio resistance can be traced to 1909, when, Schwartz noticed that when the skin was compressed with radium application, the response to radiation treatment was decreased, representing the first evidence thathypoxia induces resistance to radiation. Hollander in 1951 showed that hypoxic £. coli required doses of radiation three times more than oxicam E. coli. In 1965, Evans demonstrated a link between radiation response and, hemoglobin. When there was despair and doubt about the relevance and efficacy of hypoxic cell sensitizers, Overgaard in his metanalysis showed that sensitizers do work in head and neck malignancies. This book carries forward the optimism with sensitizers.
We thank the contributors for their sincere efforts, which made possible to give a shape to the present book.
N.G. HUILGOL
C.K.K. NAIR
V.T. KAG I
Contents
Preface
1. The clinical efficacy of hypoxic cell radiosensitizers: The beautiful hypothesis and the ugly facts
Rajiv Sarin and K.A. Dinshaw
2. Cellular membrane in modulation of radiation
damage and improvement in cancer therapy 14
B.B. Sing/t
3. Chlorpromazine—A hypoxic cell sensitizer: A new
role for an old drug 19
N.G. Huilgol
4. Mitomyciri C (MMC) and radiotherapy 26
Werner Dobrowsky
5. In vivo disposition and elimination kinetics of
senazole following intravenous and intraarterial
administration in cancer patients 33
Mathew, C.K.K. Nair and N.G. Huilgol
6. Nitrotriazole spermidine conjugate: A DNA selective
radiosensitizer 44
C.E.K. Nair, R. Mathew, S.R. Venkatachalam,
N.G. Huilgol and S. Kapoor
7. AK-2123: An overview 53
N.G Huilgol
8. An early experience with AK-2123 administered
intra- arterially in the treatment of recurrent
hemorrhaging of cervix. 60
N.G Huilgol C.KK. Nair, Rohit Mathew and
Neela A. Chatterjee
9. An overview of radiosensitizers 67
G.K. Rath, P.E. Julka and D.K. Parida
10. Radiosensitizer for hypoxic tumor imaging 74
Mrugesan, S.I. Shetty, 0. P.D. Noronha,
A.M. Samuel, T.S. Srivastava and C.K.K. Nair
1. The Clinical Efficacy of hypoxic Cell
Radiosensitisers: The beautiful hypothesis
and the ugly facts
Rajiv Sarin* and K.A. Dinshaw2
‘Division of Radiation Oncology, Tata Memorial Hospital, Mumbai 400 012, India
2Director, Tata Memorial Hospital, Mumbai 400 012, India
Introduction
It is known for over sixty years that the tumor hypoxia can result in radioiesistance and various strategies have been employed to overcome this problem. The most extensively investigated approach in the past 25 years has been of using hypoxic cell radiosensitizers. A number of in vitro, pharmacokinetic and randomized clinical studies have been performed for various such radiosensitizers. However, these agents are still considered in visitational by most oncologists. Only in a few instances they are used in the routine clinical practice for head and neck cancers [26].
This article reviews all the important randomized clinical trials of hypoxic cell radiosensitizers. The strategies used, the results obtained and the possible reasons for the failure have been analyzed.
The Beautiful hypothesis: ’Hypoxic cell radiosensitizers can improve cure rates with radiotherapy’
This beautiful hypothesis that hypoxic cell radiosensitizers should improve the cure rates and therapeutic ratio with radiotherapy (RT) is based on the following observations:
Tumors have hypoxic cells: In their seminal paper in 1955, Thomlinson and Gray [30] demonstrated on a human lung cancer model that tumors have hypoxic cells. This hypoxic component increases with increasing tumor diameter and its distance from the capillaries. Subsequently, a number of other investigators [3, 11, 16, 22] have confirmed the presence of hypoxia in various human tumors by measuring oxygen concentration with the help of electrodes.
Clinical studies on association of hypoxia with radiation failures: A number of clinical studies have confirmed that in vivo tumor hypoxia, as measured with oxygen electrodes, results in inferior tumor control by radiation as compared to the well oxygenated tumors [3, 5, 11, 12, 18]. In addition to the reduced radiation cell killing in the hypoxic cells, it is now suggested that hypoxia induce increased expression of angiogenic factors like vascular endothelial growth factors—VEGF, which can give a growth advantage to the tumor and also increase its metastatic potential [27]. In a recent human study it was noted that hypoxic soft tissue sarcomas had a higher incidence of lung metastases as compared to the well oxygenated sarcomas [4]. Hypoxia may also result in a clonal selection of resistant cells by overgrowth of mutant p53 clone which has a diminished capacity of apoptosis [14].
Association of anemia with radiation failure: The tumor oxygenation depends upon the pattern and heterogeneity of tumor blood flow, the hemoglobin level and the oxygen unloading capacity of hemoglobin, which in turn can be affected by smoking. A number of clinical studies have shown that inferior radiation response is obtained in anemic patients [13, 23, 24, 26]. In the DAHANCA 2 trial, anemic patients had inferior local control independent of the tumor size[23].
Studies on Nitroimidazole radiosensitizers: In the 1960s and 70s, a number of nitroimidazole compounds were found to significantly enhance the radiation induced cell killing in the in vitro and transplanted tumor model experiments
Hyperbaric oxygen significantly improves tumor control rates of radiotherapy: This was confirmed in few clinical trials conducted in 1960s and 70s, but this strategy was abandoned due to its complexity and hazards [15].
The Ugly Facts.- ’None of the radiosensitizer trials, except one trial of Nimorazole, showed any significant improvement in local control or survival’
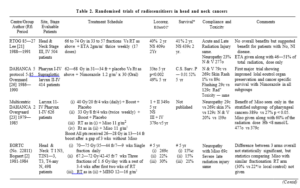
This fact is borne out in our review of the major randomized trials of chemical hypoxic cell sensitizers’ published in the last two decades (Tables 1 and 2).
Table 1. Randomized trials of radiosensitizers for gynecologic and other malignancies
| Centre/Group Author (Rf) Period | Site, Stage Evaluable Patients | Treatment Schedule | Locoreg. Control* | Survival* | Compliance and Toxicity | Comments |
| Multicentric | Cervix 11b, | XRT 40—65 Gy + ICA-variable dose, | 549o 5 yr. | 45% 5 yr. | Neuropathy 39c | Miso not always given with ICA |
| Danish cancer | 1II, IVa 331 | technique & dose rate + Placebo Vs RT-as | NS | (Crude) NS | vs 9% | Hb < 7: Poorer local control 30% |
| society Overgaard
[24] 1979— |
patients | above + Misonidazole daily Total 12g/m’ in 6 weeks. | 50% 5 yr | 39% 5 yr.
(Crude) |
Skin 2% vs 5% N&V36% vs469o | vs 57%
Heterogeneous treatment schedules in various centers. |
| 1982 | ||||||
| Multicentric | Bladder T2 | 40 Gy/20 fr/4 wks (whole pelvis) + 20 Gy/ | Complete | 41% 5 yr | N toxicity of | Increased bowel morbidity in |
| South African | Grade 3, T3 | 10 fr/2 wks (boost) Vs 40 Gy/20 fr/4 wks + | Response | NS | Misonidazole | MISO group due to unconventional |
| Study Abratt | 53 patients | 12 Gy/2 fr (boost) + MISO 3g/m2 orally and | 63% | 48*/o 5 yr | boost fractionation (6 Gy x 2). | |
| [ i 1981 — 1985 | 1g in 35 ml solvent 4h and 2h respectively prior to each 6 Gy boost fraction | NS 69% | Trial too small to detect upto 209c difference |
Miso not given for first 40 Gy/20 fr
| MRC Dische
[7] 1979— 1981 |
Cervix Figo III 139
patients |
XRT 42Gy/20 fr/4 Wks + ICA 35Gy to point A + placebo vs RT as above + Miso 500mg/ m’ with each fraction of XRT and ICA | Complete response 68% vs 647o | 557o 2 yr vs 51% 2 yr | Neuropathy 39n vs 36% | No benefit with Miso. Major deviation in Miso dose in 34/68 patients, mostly due to toxicity. | oxic Cell |
| MRC Dische | Cervix 11b, | 50 Gy/25 fr/5 wks + ICA —various techniques, | # 7S*/o 3 yr | # 63% 3 yr | N&V 37% vs65% | Sig•nificantly inferior results with | |
| [8] 1987— | III | dose rate and dose used. VS RT as above + | p = 0.009 | p = 0.044 | Diarrhoea 889c | Pim | |
| 1991 | 183 patients | PIM 750 mg/m2 slow infusion 15 min before | 60% 3 yr | 43% 3 yr | vs 80% | Significantly grater LN + patients | p |
| each fraction of XRT but not ICA | Malaise 74% | in RT + Pim arm | |||||
| (Pim) Skin 23% | Blood transfusion required in 367c | p- | |||||
| (Pim) | pim vs 23% RT alone patients | ° |
| Centre/Group | Site. Stage | Treatment Schedule | Locoreg. | Survi val* | Compliance and | Comments | |
| Author (RR | Evaluable | Control* | Toxichy | ||||
| Period | Patients |
Miso = Misonidazole; ETA = Etanidazole; PIM = Pimonidazole N & V = Nausea and Vomiting; pts = Patients; P = Placebo
* Local control and sur vival figures are actuarial or Kaplan Meier unless specified otherwise
C.S. Surv = Cancer Specific Sur vival; NS = Not significant (p > 0.5) # Estimated from the published Kaplan Meier plots
Neuropathy 25% Radiation Toxicity-same
Table 2. Randomized trials of radiosensitizers in head and neck cancers
|
Majority of these trials have used misonidazole [1, 2, 7, 10, 15, 19, 20,23, 24, 29], two trials have used etanidazole [9, 21] and single trials of nimorazole [2 1 and pimonidazole [8] each. Most of these trials are large enough to detect a major benefit of radiosensitizers in head and neck or cervical carcinoma, where locoregional failure is the main cause of death. The possible explanations for these disappointing results could be as follows:
- The original beautiful hypothesis was not based on firm foundation
Even after so many failed clinical trials of hypoxic cell radiosensitisers
we do not feel that this hypothesis is on shaky grounds. There is ample evidence to show the presence of hypoxic cells in tumours, radioresistance of hypoxic tumoui cells and the ability of radiosensitisers to make hypoxic cells more sensitive to radiation in animal experiments.
In the animal experiments, radiosensitisers were more effective when used with single large dose of radiation as compared to multifraction RT, possibly due to reoxygenation. Nevertheless, even this smaller benefit of radiosensitisers seen in multifraction RT animal experiments should have been sufficient to improve local controls in human clinical trials. It is unlikely that in a conventionally fractionated course of RT, the normal pi ocess of reoxygenation is so efficient that tumor hypoxia is no more a clinically significant problem. However one has to realize that tumour hypoxia is only one of the reasons behind radiation therapy failure. Tumour heterogeneity with intrinsic radioresistance, repopulation or inappropriate fractionation are equally important.
- The human experiments (clinical trials) to test this hypothesis were not designed and executed well
For a variety of reasons, the type of radiosensitizer, its dose/frequency and the radiotherapy regimen used in most of the randomized studies was not optimal for attaining maximum radio sensitization and clinical benefit. Both misonidazole and etanidazole a1’e associated with dose dependent peripheral neuropathy, thus limiting the maximum tolerable dose of 12 g/m2 for misonidazole and 34 g/m° for etanidazole. Pimonidazole is not associated with these neurological toxicity but produced discomfort, hot flushes, malaise and skin rashes leading to discontinuation of the drug in 28/91 (319c) patients [8]. As a result of these dose limiting toxicity, in most of the trials, radiosensitisers wei e either given with only part of the conventionally fractionated RT or else unconventional RT fractionation with dose per fraction or a split course was used.
In both the etanidazole trials [9, 21] this radiosensitizer was given with only 17 of the 33-37 daily fractions, i.e. only 46-50% of the total radiation dose was given with the radiosensitizers. Similarly, in the majority of misonidazole trials, this radiosensitizer was either given with only 33-60(o of the external radiation dose [1, 10, 20, 23, 29]; or not always
given with intracavitary treatment. In only five trials, it was planned to give misonidazole with all the radiation fractions — the MRC Head Neck [15], EORTC Head Neck [2], RTOG Lung [28] RTOG Brain metastasis trial [19] and the MRC cervix trial [7]. In four of these trials, the radiotherapy fractionation was compromised to avoid exceeding the total misonidazole dose of 13 g/m2. Thus in the MRC head and neck study 2.5 to 4.5 Gy per ft action was used and in the RTOG lung trial 6Gy per fraction was used. In the RTOG Brain metastasis trial the radiation response rates are not published. Misonidazole with cranial irradiation did not affect survival, which is not surprising since two thirds of the patients died of extracranial disease. In the EORTC Head and Neck study, a rest of 3—4 weeks was given after the first two weeks of three fractions per day RT. In this split course treatment, tumor reoxygenation and repopulation may have negated any benefit of hypoxic cell radiosensitizer. In this EORTC study, the differences in locoregional control when comparing all three treatment arms together (Conventional RT, Split Course RT and Split Course RT with misonidazole) is not statistically significant. However, the statistical significance comparing the 5 year locoregional control rate of 30% in 167 patients receiving misonidazole with split course RT versus 22to in 163 patients treated with similar RT fractionation i.e. split course, has not been published and might be significant. In the MRC cervix study the protocol was to give 500 mg/m2 misonidazole with all 20 tractions of external RT (XRT) and also with intracavitary (ICA) treatment. However, there was major deviation in the misonidazole dose on 34/68 patients, mostly due to drug toxicity. If one ignores the statistically significant improvement seen on subset analysis in the DAHANCA 2 [23] and RTOG 85—27 [21] trials as a play of chance, we are left with only two trials [8, 26] that show statistically significant difference between the two treatment arms. The issue is further complicated by one trial showing significantly inferior local control (hazard ratio 2.1) with pimonidazole [8] while other showing a significantly improved local control with nimorazole [26]. These two trials are discussed
below.
MRC Pimonidazole Trial [8]
This statistically significant adverse effect on local control and survival by pimonidazole as shown in Table 1, is an intriguing finding. It is difficult to explain these results and the investigators have considered these three hypotheses as a possible explanation:
- Pimonidazole increased morbidity leading to suboptimal radiotherapy: This is an unlikely cause since the main course of radiotherapy was the same in both groups and there were only some minor differences in the intracavitary and additional
- Pimoiiida Role resulted in ali adverse radiation response: In animal tumor models, pimonidazole in similardose/m* produced a marked reduction in tumor blood flow [6]. This vasoactive action is unique for pimonidazole and does not occur with etanidazole or misonidazole in doses used If this phenomenon observed in animal experiments occurs in human tumors also, it may aggravate hypoxia and radiation failure.
- Despite ratidomisation more favorable cases were included in the RT alone arm: In this study lymphangiography was done in only 81 cases and showed lymph node involvement in only 8/40 (20%) of patients in RT alone arm compared to 17/41 (41%) in the RT + pimonidazole arm (p = 0.036). Similarly, a higher proportion of patients required blood transfusion before RT in the RT + pimonidazole arm 33/91 (35%) compared with 21/92 (23%) in the RT alone arm (p — 046). Thus, one may speculate that even though patients were stratified by hemoglobin (Hb) level above or below 12 g/d1 a higher proportion of patients in the pimonidazole arm might have been having Hb < 11.5—12 g/d1 which was the criterion used for blood transfusion. The mean Hb levels are comparable in the two groups but the authors have not mentioned whether these Hb levels were before or after transfusion and have not discussed the reasons for a significantly hi gher requirement for transfusion before RT in the pimonidazole arm. Also, the complete or good responses rates in stage II ate comparable in the two treatment arms (43.8% and 44.1%). But in stage III, better response rates (41.8%) was observed in the RT alone compared to 26.3% in RT + pimonidazole arm (p = 0.10). The investigators in this study have pointed out that in this group of patients with late stage II (one third patients) and stage III (two third patients), the 759c 2 year control rate in the RT alone arm is higher than the expected figure of 65% from the previous MRC trials. Thus, the two year local control of 60% seen in R.T + pimonidazole arm may be significantly inferior to the RT alone arm of this study but is not much different from the expected figure oi’ 65Yo.
In summary, this unexpected finding of significantly worse results in the pimonidazole arm be a true adverse effect due to drug induced reduction in tumour blood flow. Alternatively, this may be explained by significantly higher advise agnostic factors such as lymph node involvement and requirement for blood transfusion in the pimonidazole arm.
DAHANCA Nimorazole Trial (26]
This is the only large randomized trial showing improved locoregional control and cancer specific survival with the use of a radiosensitizer along with conventional radiotherapy for head and neck cancers. The benefit was statistically significant on both univariate and multivariate analysis and was observed in all the subgroups. The possible reasons for the significant benefit seen in this trial when all the others trials failed, could be manifold. Most importantly, this is the only large trial where the radiosensitizer was given along with the majority of a con venational fractionated course of radiotherapy. Of the 33 fractions (median) of RT, nimorazole was given with 30 fractions (91%) in 49% patients; >25 (>76%) fraction in 139c patients and <25 (<76%) fractions in 38Uo patients. As discussed earlier, in most of the etanidazole and misonidazole trials with conventional radiotherapy, the drug was given along with 33—60% of the radiation fractions only. Also in this study, 86% patients obtained peak plasma nimorazole value of >25 mg/L which was considered satisfactory. The significant results of this study are based on ‘intention to treat’ basis. In fact the 34% five year locoregional control in 34 patients (15%) receiving
< 5 nimorazole treatments due to toxicity, was similar to 339o five year control in the placebo group. In contrast a 529c five year control rate was observed in the remaining 185 patients who received most of the planned nimorazole treatments. This suggests an apparent dose relationship for nimorazole.
While nimorazole was less effective than misonidazole in animal experiments, it may achieve better radio sensitization in the clinically usable doses. It has been shown the amount of nimorazole within the tumor per fraction in a 30 fraction regimen is almost twice that of etanidazoe [25]. One of the criticisms of this trial is that despite large number of patients and long follow up, this only showed a significant improvement in the locoregional control and cancer specific survival but not in the overall survival. This can be explained by the fact that of the 307 total deaths, one third (94 patients) were due to non cancer related causes. This is not surprising in a group of patients with a median age of 60 years at diagnosis
and a median follow up of almost 10 years.
Conclusion
Chemical hypoxic cell radiosensitizers showing sensitizing properties in the initial animal experiments have come a long way and an enormous clinical effort has been made to evaluate their efficacy in human cancers. The disappointing results seen in the clinical trials are multifactorial but are not sufficient to conclude that tumor hypoxia is an insignificant cause of radiation failure. The last word regarding the clinical efficacy of these drugs is yet unsaid. Hence the need of the hour is to vigorously pursue further trials of nimorazole and other promising drugs like AK- 2123 [17
References
- Abratt P., Craighead P., Reddi V.B., Sarembock L.A. A prospective randomized trial of radiation with or without oral and intravesical misonidazole for bladder cancer. Br J Cancer: 64: 968—970, 1991.
- Bogaert W. Van den, Schueren E., Horiot J., et al. The EORTC randomized trial on three fractions per day (trial No. 22811) in advanced head and neck cancer: long term results and side effects. Radiotherapy and Oncology 35: 91—99,
- Brizel D.M., Rosner G.L., Proznitz L.R. and Dewhirst M.W. Patterns and variability of tumor oxygenation profiles of human soft tissue Int.
- Radiat. oncol. Biol. Phys. 32: 1121—1125, 1995.
- Brizel D.M., Scully S.P., Harrelson J.M., et a1. Tumor oxygenation predicts for likelihood of distant metastases in human soft tissue sarcoma. Cancer Res. 56: 941—943,
- Brizel D.M., Sibley G.S., Prosnitz L.R., Scher R.L. and Dewhirst, M.W. Tumor hypoxia adversely affects the prognosis of carcinoma of the head and neck. J. Radiat. Oncol. Bio1. Phys. 38: 285-289, 1997.
- Chaplin D.J., Horsman M.R. Tumor blood flow changes induced by chemical modifiers of radition response. J. Radiat. Oncol. Biol. Phys. 22: 459—462, 1992.
- Dische S., Adams G.E., Ash, D.V., et al. The medical research council trial of misonidazole in carcinoma of the uterine cervix. The Br Jr of Radiol. 57: 491- 499, 1984.
- Dische S., chassagne D., Hope-Stone H.P., et a1. A trial of Ro 03-8799 (pimonidazole) in carcinoma of the uterine cervix: an interim report from the Medical Research Council Working Party on advanced carcinoma of the Radiotherapy and Oncology 26: 93—103, 1993.
- Eschwege F., Sancho-Garnier , Chassagne D., et al. Results of a European randomized trial of etanidazole combined with radiotherapy in head and neck carcinomas. Int. J. Radiat. Oncol. Biol. Phys. 39: 275—281, 1997.
- Fazekas J., Pajak F., Wasserman T., et al. Failure of misonidazole-sensitized radiotherapy to impact upon outcome among stage III-IV squamous cancers of the head and neck. Int. J. Radiat. Oncol. Biol. Phys. 13: 1155—1160, 1987.
- Fyles W., Milosevic M., Wong R., Kavanagh M., ct al. oxygenation predicts radiation response and survival in patients with cervix cancer. Radiother and Oncol. 48: 149—156, 1998.
- Gatenby R.A., Kessler H.B., Rosenblum J.S., et al. Oxygen distribution in in squamous cell carcinoma metastases and its relationship to outcome of radiation therapy. J. Radiat. Oncol. Biol. Phys. 14: 831—838, 1988.
- Girinsky T., Pejovic-Lenfant M.H., Bourhis J., et al. Prognostic value of hemoglobin concentrations and blood transfusions in advanced carcinomas or the cervix treated by radiation therapy: results of a retrospective study of patients. Int. J. Radiat. Oncol. Biol. Phys. 16: 37—42, 1985.
- Graeber T.G., Osmanian C., Jacks T., et al. Hypoxia mediated selection of cells with diminished apoptotic potential in solid Nature 379: 88—91, 1996.
- Henk M., Adams G.E., Ash D., et al. A study of the effect of misonidazole in conjunction with radiotherapy for the treatment of head the neck cancer. Br.
- Radiol. 57: 585—595, 1984.
- Hockel , Schlenger K., Knoop C., and Vaupel P. Oxygenation of carcinomas of the uterine cervix: a evaluation by computerized 02 tension measurements. Cancer Res. 51: 6098—6102, 1991.
- Huilgol G., Chatterjee N.; Mehta A.R. An overview of the initial experience with AK—2123 as a hypoxic cell sensitizer with radiation in the treatment of the advanced head and neck cancers. Int. J. Radiat. Oncol. Bio1. Phys. 34: 1121 —1124; 1996.
- Hockel M., Schlenger K., Ara1 B., Mitze M., Schaffer U., and Vaupel, P. Association between tumor hypoxia and malignant progression in advanced cancer of the uterine cervix. Cancer Res. 56: 450’i—4515,
- Komarnicky T., Phillips T.L., Martz K., et al. A randomized phase III trial for the evaluation of misonidazole combined with radiation in the treatment of patients with brain metastasis (RJ’OG-7916). Int. J. Radiat. Oncol. Biol. Phys. 20: 53-58, 19991.
- Lee Ding-Jen, Pajak T.F., Stetz J. A Phase I/II study of the hypoxic cell sensitizer misonidazole as an adjunct to high fractional dose radiotherapy in patients with unresectable cell carcinoma of the head and neck: a RTOG randomized study (#79-04). Int. J. Radiat. Oncol. Biol. Phys 16: 465—470,
- Lee Ding-Jen, Cosmatos D., Marcial V.A., et Results of an RTOG phase III trial (RTOG 85—27) comparing radiotherapy plus etanidazole with radiotherapy alone for locally advanced head and neck carcinomas. Int. J. Radiat. Oncol. Biol. Phys. 32: 567—576, 1995.
- Nordsmarck M., Bentzen S., M. and Overgaard J. Measurement of human tumor oxygenation status by a polarographic needle electrode. Acta. Oncol. 33: 383—389,
- Overgaard , Hansen H.S., Anderson A.P., et al. Misonidazole combined with split course radiotherapy in the treatment of invasive carcinoma of larynx and pharyx: report from the DAHANCA 2 study. Int. J. Radiat. Oncol. Biol. Phys. 16: 1065—1068, 1989.
- Overgaard J., Bentzen S.M., Kolstad P., et al. Misonidazole combined with radiotherapy in the treatment of carcinoma of the uterine cervix. Int. Radiat. Oncol. Biol. Phys. 16: 1069—1072, 1989.
- Overgaard J. Clinical evaluation of nitroimidazoles as moditiers of hypoxia in solid tumours. Res. 6: 509—518, 1994.
- Overgaard J., Hansen H.S., Overgaard M., et a1. A randomized double-blind phase III study of nimorazole as a hypoxic radiosensitizer of radiotherapy in supraglottic larynx and pharynx carcinoma. Results of the Danish Head and Neck Study (DAHANCA) Protocol 5—85. Radiotherapy and Oncology 46: 135—146,
- Shweiki , Neeman M., ltin, A., and Keshet E. Induction of vascular endothelial growth factor expression by hypoxia and by glucose deficiency in multicell spheroids: implications for tumor angiogenesis. Proc. Natl. Acad. Sci. USA 92: 768—772, 1995.
- Simpson J.R., Rauer M., Wasserman T.H., et Large fraction irradiation with or without misonidazole in advanced non-oat cell carcinoma of the lung: a phase III randomized trial of the RTOG. Int. J. Radiat. Oncol. Biol. Phys. 13: 861—867, 1987.
- Stehman F.B., Bundy B.N., Thomas G., et al. Hydroxyurea versus misonidazole with radiation in cervical carcinoma: long term follow-up of a Gynecologic Oncology Group J. Clin. Oncol. 11: 1523—1528, 1993.
- Thomlinson H, Gray L.H. The histological structure of some human lung cancers and the possible implications for radiotherapy. Br. J. Cancer 9: 539— 549, 1955.
2. Cellular Membrane in Modulation of Radiation Damage and Improvement in Cancer Therapy
B.B. Singh
Former Head, Radiation Biology & Biochemistry Division Bhabha Atomic Research Centre, Mumbai, India
Targets of Radiation Damage
Radiation damage to cells has been largely ascribed to the lesions in the genetic material. The modification of radiation effects on cells by physical or chemical means was therefore believed to be achieved mainly by interfering with the initial chemical lesions in the DNA or their subsequent repair by several enzymatic processes. Earlier, radioprotection by chemicals was demonstrated by scavenging the free radicals formed during the radiolysis of intracellular water thereby protecting the cellar DNA from the initially damaging events. Alternatively, restitution of damaged DNA molecules by sulfhydryl compounds could also lead to protection against radiation damage [1]. Subsequently, incorporation of halogenated base analogues into the genetic apparatus of cells was demonstrated to increase the radiation induced radination lethality of cells. Consequently sensitization of bacterial cells to UV light and ionizing radiation on incorporation of BudR and IudR in the DNA, formed the most convincing evidence in favor of the cellular DNA being the main target for manifestation of radiation induced lethality of living cells [23]. Similar effects also reported in mammalian cells. However there is ample evidence to suggest that the cellular membrane is an equally important target particularly in the modification of the radiation lethality of cells, which is the main effect desired in radiation treatment of malignancies. Alper [5] demonstrated the importance of cellular membrane in manifestation of oxygen effect in irradiated cells and Shenoy et at conclusively established that cellular membrane plays an important role in enhancement of radiation effects in hypoxic cells. Using a known sensitizer, iodoacetic acid labeled with I—131, they observed that on exposure to radiation, iodine atoms were released from the molecule which reacted with the membrane proteins of E. coli B/r resulting in inhibition of post- irradiation protein and DNA synthesis [7]. The toxic iodine atoms were released on reaction of hydroxyl radicals with the sensitizer which was reconfirmed by generating such species in Fenton’s reaction in presence of iodoacetic acid and alkali halides resulting in iodination of cells and increasing their sensitivity to UV and ionizing radiation [ I It was then postulated that modifications in the biophysical structure of the membrane lead to post-irradiation inhibition of DNA repair. Thus, any agent which could modify the structure of the membrane would in principle modify the cellular response to radiation.
Membrane Specific Drugs
On the abovementioned rationale, several drugs have been studied for their effect on radiation response of living cells. The foremost among these are the local anesthetics, analgesics and tranquilizers. Most of these have been assessed for their radio sensitizing ability with the sole purpose of their potential use in radiotherapy of cancer and more particularly as hypoxic cell radiosensitizers.
(a) Local Anesthetics
Procaine hydrochloride, a commonly used local anesthetic was demonstrated to preferentially enhance the radiation lethality of E. coli B/r cells under anoxia [9]. Whereas the maximum sensitization was observed when the drug was present during irradiation of cells but some sensitization was also observed when drug was added even after irradiation. Post-irradiation effect of the drug was however dependent on the time lapsed after radiation exposure when the cells were exposed to the drug. Furthermore, the drug did not enhance the lethal effect of UV exposure clearly indicating that the site of drug action was not the DNA [10]. Procaine was later shown to inhibit post-irradiation protein and DNA synthesis, which could explain its radio sensitizing effect [11]. Other local anesthetics such as lidocaine, lignocaine and tetracaine were also found to enhance the lethality of bacterial cells iiiadiated under anoxic condition [121
Employing E. coli K10d0 cells, Yatvin demonstrated a protective effect
of procaine under euoxic conditions of irradiation [13]. Using mammalian cells Djordejevic [14] reported that procaine protected Hela cells from radiation damage under euoxia but enhanced their lethality when the cells. were treated with the drug after irradiation. Yau and coworkers also confirmed the radio sensitizing effect of procaine in hypoxic CHO and L5178Y cells as well as their protection under euoxia [15, 16]. This radioprotective effect of procaine iepoi ted in euoxic mammalian cells but missed by the earlier workers in bacterial systems was to be further investigated in view of its importance in radiotherapy when the same di’tig could possibly protect the euoxic cells shh bounding the tumor at the same time sensitizing the hypoxic cells within the interiors of the tumor. It was observed that the radioprotective ability of such drugs in euoxic cells was dependent on concentration of the drug which phenomenon was extensively investigated with other membrane specific drugs since procaine did not show much promise in experimental tumors [12].
The local anesthetics by virtue of their ability to modify the cellular membrane have been studied also for enhancement of chemotherapeutic drugs such as bleomycin and pepleomycin [17, 18] and 4or circumvention of resistance of malignant cells to anthracyclin [19].
- fb) Phenothiazines
Phenothiazines include tranquilizers, antihistaminics, antipyretics and aniemetics. Chlorpromazine (CPZ) is commonly used as a tranquilizer and was shown to sensitize hypoxic ñ. coli B/r cells to y-rays [20]. Prochlorperazine (PCP), promethazine (PMZ) and trimeprazine (TMZ) also sensitized bated ia1 cells to y-rays at sub millimolar concentrations [21, 22] and a combination of procaine and CPZ gave sen.sitization gi’eater than that by oxygen [12]. When bacterial cells were irradiated in presence of these phenothiazines, DNA and protein syntheses were found to be inhibited [21]. In addition, an increase in DNA single strand breaks and subsequent inhibition of their repair was also observed [23, 24]. Whereas these observations may explain the enhancing of radiation lethality of cells, there are other biochemical processes, which get affected by the presence of phenothiazines and may contribute to the radiation effects mentioned above. A summary of these pi’ocesses is described elsewhere [25].
Phenothiazines at i’e1atively low doses have also been reported to cause radioprotection of euoxic bacteria and mammalian cells [26, 27] which has been attributed to the fluidization o4 the cellular membrane resulting in increased mobility of non-protein sulfhydryl groups thereby leading to efficient restitution of damaged oxygen sensitive sites on molecules [271 The OER values for E. coli B/r cells decreased from 2.8 to 1.7, 1.3, 1.8 and 1.6 in presence of CPZ, PMZ, PCP and TMZ, respectively. This radioprotective elect of phenothiazine was also demonstrated in vivo in Swiss mice [28].
Concomitant with its radio sensitizing effect, CPZ was also demonstrated to be preferentially toxic to bacterial and mammalian cells [29, 30j. Trifluoperazine and several hydroxylated phenothiazines also were found to be cytotoxic to hypoxic human lymphatic leukemia L1210 and P-388 murine leukemia cells [30]. The cytotoxic effect of such was found to correspond to their calmodulin inhibiting activity [31]. Cells held under chronic hypoxia in presence of CPZ showed twice as great sensitivity to prays as cells under oxygen. In addition, enhanced radiosensitivity of bacterial and mammalian cells was also observed when irradiation was car i red out at higher temperature [32].
Radio sensitizing effect of CPZ and other phenothiazines has been demonstrated in two transplantable murine solid in vivo tumour s namely a fibrosarcoma and Sarcoma l80A [33—35] as well as in a spontaneously occur ring mammary adenocarcinoma [36] in CBA mice but not in the ascites form of Sarcoma 180A [35]. Although in such experiments high doses of drugs have been used, the results are nevertheless very interesting and to considerable importance to radiotherapy.
The selective toxicity and radio sensitizing effect of phenothiazine drugs in an toxic/hypoxic cells and tumors in vivo as discussed above, and the radioprotective of these same drugs in euoxic cell systems at lower concentrations, would prove highly advantageous in treatment of human malignancies by chemo- and radiotherapies particularly in view to such drugs being presently used for other medical purposes. Clinical trials with carcinoma of cervix and head and neck cancers have been initiated and the preliminary results are highly encouraging.
References
- Bridges B.A. (1969), Adv. Radiat. Biol., 3, 123.
- Djoi djevic and Szybalski W. (1960), J. Exp. Med,. 112, 509.
- Szybalski (1967), Radiat. Res. (Suppl. 7), 147.
- Shipley V., Elkind M.M. and Prather W.B. (1971); Radiat. Res. 47, 437.
- Al per Nature (Loud), (1968), 217, 862.
- Shenoy A., Singh B.B. and Gopal-Aycnger A.R. (1968), Science (Wash), 160, 9’99.
- Shenoy A., Joshi D.S, Singh B.B. et aI. (1970), Adv. Biol. Med. Phys. 13, 255.
- Singh B.B., Shenoy M.A. and Gopal-Ayengar A.R. (1971), J. Exptl. Bio1, 9, 518.
- Shenoy A., Singh B.B. and Gopal-Ayengar A.R. (1974), Nature (Lond), 248, 416.
- George C., Shenoy M.A., Joshi D.3., Singh B.B. et at (1975), Brit. J. Radiol. 48, 611.
- Nair K.K. and Pradhan D.S. (1975), Chem. Boil. Interact. 11, 178.
- Shenoy A., George K.C., Srinivasan V.T., Singh B.B. et al, (1976) In: “Modification of Radiosensitivity of Biological Systems”, IAEA, Vienna 131.
- Yativin B., Schmitt B.J. and Dennis W, H. (1980), Int. J. Radiat. Biol. 37, 531.
- Djordejevic (1979), Radiol. 131, 515.
- Yau M. (1979), Radiat. Res. 80, 523.
- Yau M. and Kim S.C. (1980), Brit. J. Radiol. 53, 687.
- Mizano S and Ishida (1982), Biochem. Biophy. Res. Commns. 105, 425.
- Komatsu and Sakomoto K. (1985), J. Radiat. Res. 26, 426.
- Kessel (1986), Cancer Surv. 5, 109.
- Shenoy M.A., George C., Singh B.B. et at. (1975), Int. J. Radiat. Biol. 28, 519.
- Maniar S. and Singh B.B. (1983), Int. J. Radiat. Bio1. 44, 399.
- Maniar S., Nair C.K.K. and Singh B.B.(1984), Radiat, Env. Biophys. 23, 279.
- Shenoy A. and Gopalkrishna K. (1978), Int. J. Radiat, Biol. 33, 587.
- Yonei S (1979), J. Radiat. Biol, 36, 547.
- Shenoy A. and Singh B.B. (1992), Cancer Invest. 10, 533.
26: Maniar H.S. and Singh B.B. (1985), Ind. J. Exptl. Biol. 23, 100.
- Lenhert (1983), 7th Congr. Radiat. Res. Amsterdam Abs. No. B—6, 20.
- Shenoy A. and Singh B.B. (1986) Radiosens. Newsletter 5(2), 3.
- Shenoy A. and Singh B.B. (1978), Int J. Radit. Biol. 34, 595.
- Stater M., Sweet P.M. et a1. (1987), anticancer drug Design 1, 297.
- Lehnert (1987) Res. Commune. Chem. Pathol. Pharmacol. 56, 361.
- Shenoy A. and Singh B.B. (1985), Radiat. Env. Biophy. 24, 113.
- Shenoy A. and Singh B.B.(1980), Indian, J. Exptl. Biol. 18, 791.
- George C., Srinivasan V.T. and Singh B.B. (1980), Int. J. Radiat. Biol. 38, 661.
- George C. and Singh B.B. (1984), Indian J. Exptl. Biol. 22, 305.
- Shenoy A., Biaglow J.E. et al (1582), Int. J. Radiat. Oncol. Biol. Phys. 8, 725.
3.Chlorpromazine—A Hypoxic Cell Sensitizer: A new role for an old drug
N.G. Huilgol
Division o1 Radiation Oncology, Nanavati Hospital and Research Centre, Mumbai-400 056, India
Quest for an ideal radiation sensitizer led to screening of molecules with diverse properties. The search for an ideal hypoxic cell sensitizer has eluded us. Hyperbaric oxygen, perflurocarbon, and imadazoles have had a dismal outcome except in few trials.Chlorpromazine (CPZ) is a well known antipsychotic drug with a wide range of other activities of which radiation sensitizing is least discussed. The conventional sensitizers have been effective by being electron affinic like metronidazole, imadazole or senazole. CPZ, with its wide ranging actions on cell proliferation and repair, sensitizes the tumour cells by a novel and distinct mechanism.
Chlorpromazine, an anticalmodulin as well as membrane active drug was investigated by Singh and his colleagues. Maniar and Singh demonstrated the twin potential of CPZ as sensitizer and protector at a high and low cellular concentration [1]. This effect in neuroblastoma cell line was demonstrated by Abe et a1 [2]. The in vitro and in vivo studies by Singh et al have demonstrated the efficacy of CPZ as a radiation sensitizer. There has been a meager attempt to assess CPZ as sensitizer of radiation in the clinic. This article will i eview the drug and its possible new role as radiation sensitizer.
Pharmacology
Chlorpromazine is an odourless white or creamy white crystalline powder with a molecular formula C l7H l9CINz which comes under aliphatic group of phenothiazines. It decomposes to yellow, pink and finally violet colour on exposure to air. It is insoluble in water. The drug may be administered orally, intravenously or as deep intramuscular injections and also per rectally. It is readily absoroed by gastrointestinal route but the serum concentration rises slowly due to considerable first-pass metabolism in the gut wall. It is also extensively metabolized in liver. The serum levels vary between individuals. Significantly, serum concentration of CPZ and metabolites do not have a simple relation to thei-apeutic effects. Chlorpromazine is metabolized by hydro-oxygenation, conjugation with glucoronic acid, oxidation of sulfur atom and dealkylation. The plasma half-life of chlorpromazine is reported to be only a few hours, the metabolites (more than 100) are eliminated over a longer period of time. It is liberally bound to plasma proteins [3]. And, it is widely distributed in the body and crosses blood brain barrier easily.
Chlorpromazine has a wide range of activity arising from its depressant actions on the central nervous system and its alpha-adt’energic blocking and weaker antimuscarinic activities. It is a dopamine inhibitor , it inhibits prolactin-release-inhibitory factor, considered to be dopamine, thus stimulating the release of Prolactin. The turnover of dopamine in the brain is also increased. It has anti-adrenergic blocking potential with weak antimuscaranic activities. Chlorpromazine can relax skeletal muscle, induce vasodilatation, hypotension and tachycardia, CPZ also has anti-emetic, antipruritic, and antiserotinin properties. It has a weak antihistaminic properties and ganglio-blocking activity. Chlorpromazine is a membrane active drug. It can stabilize cell membrane, an impoi’tant mechanism of action when hypoxic cell sensitization is under consideration [4].
Chlorpromazine has sedative properties but patients usually develop tolerance to sedative effects. It has anti-emetic, antipi uritic, membrane stabilizing and vasodialatory effects. It destabilizes heat regulatory mechanism. It also produces tachycardia and decrease salivary and gastric secretions [4].
Chlorpromazine is administei’ed orally in the dose of 25-50 mg three times daily and gradually increased if necessary. Deep intramuscular CPZ is safer than intravenous injection. Sub-cutaneous administration is contraindicated. The usual dose by injection iE 25 to 50 mg repeated as required. A lethal dose of CPZ in a 4 year old child was 350 mg. Adults have survived 9.7 g [5].
Adverse Reactions
Adverse effects may include dry mouth, constipation, urinary retention, hydriasis, agitation, insomnia, depression, convulsion, nasal congestion, tachycardia, ECG changes, postural hypotension, miosis, blurred vision and inhibition of ejaculation.
Allergic reactions include urticaria, exfoliative dermatitis and contact sensitivity. Jaundice has been reported as of allergic origin. Prolonged therapy may lead to pigmentation of skin, cornea and also lens opacities. Hematological disorder includes fatal agranulocytosis. Extrapyramidal dysfunction has been reported which is due to the effects 9n-dopaminergic transmission. Extrapyramidal symptoms include parkinsonism like symptoms, akathisia and neuroleptic malignant syndrome.
Endocrine sequel includes amenorrhea, gynecomastia, and weight gain, altered glucose tolerance and increased serum cholesterol concentration. Chlorpromazine has not been reported to cause dependence of the type encounter ed with barbiturates or benzodiazepines. However, mild with- drawl symptoms have been encountered with patients receiving prolonged therapy.
Rationale
It is a less known fact that CPZ can potentiate cytotoxic effects of radiation at a high concentration whereas the lower cellular concentration affords a degree of protection to radiation. It being an anti-calmodul in is also shown to influence many cellular functions including inhibition of cellular proliferation and inhibition of DNA repair [6]. Levin and Weiss demonstrated that phenothiazines bind to and antagoniz• the action of calmodulin (CaM), a widely distributed multifunctional, calcium-binding protein [7].
Hirai et.al. have reported antiproliferative activity of CPZ while studying SV40 DN.A replication in a Hela cell extract. Analysis with agarose gel electrophoresis revealed that some steps of DNA chain elongation and maturation of closed circular forms were sensitive to CPZ [8].
It is also suggested that CPZ induces cell death due to marked non- specific inhibitor y effects on various processes in cells.
CPZ is also a sympathetic nerve antagonist in smooth muscles vasculature. Vasodilatation thus induced may increase tumor perfusion. Such a phenomenon may obviate a complicated perfusion determined hypoxia of a tumor.
Hypoxic cells steeped in a low pH milieu are inefficient in repair of DNA damage. CPZ, potentates this inability to repair presumably through Ca+° calmodulin complex or merely by decreasing the available intracellular ATP, crucial for energy dependent repair process. It is also noted that calmodulin antagonists like CPZ exhibit antiproliferative properties in certain cell lines [6]. The above studies demonstrate the antiproliferative and repair inhibition of DNA in various cell lines in vitro. Increased perfusion by smooth muscle relaxation may enhance perfusion of tumour to eliminate perfusion limited hypoxia. CPZ, which acts through multiple channels may be both, hypoxic cell sensitizer and cytotoxic agent,
Clinical Studies
The study reported by us in head and neck cancer is one of the earlier work exploring the potential of CPZ, as a sensitizer. Patients with Stage III and IV head and neck cancer were evaluated in a prospective randomized study treated with conventionally fractionated radiation. Patients in the study weie landomized following histological confirmation of squamous cell carcinoma. Patients with glottic and nasopharyngeal cancer were not accrued in this trial. Patients in both the groups underwent radiation with conventionally fractionated radiation. A total of 60 Gy was delivered with an individual fraction of 200 cGy daily, five days a week. Patients randomized to CPZ group received daily dose of 50 mg of CPZ orally in equally divided dose. Patients underwent weekly evaluation of acute toxicity due to radiation and CPZ. The compliance of CPZ ingestion was based on wrapper inspection. Table l shows initial response in both the groups while Fig.1 shows overall survival function estimates. Patients who received CPZ showed an initial complete response of 90%, while, only 709o complete response was seen in the control group. The pairwise comparisons of initial response indicated significant response in CPZ treated group (p = 0.01ñ). The difference is significant at 5% level of significance. The comparison of overall survival curve shows better survival for CPZ group. A p-value of 0.08 for the effectiveness of CPZ suggests a better response due to CPZ.
| Table 1 | ||
| % CR | No PR | |
| Complete Response | Partial Response | |
| Control | 70 | 30 |
| n = 20 | n — 14 | n = 6 |
| CPZ | 90 | 10 |
| n = 38 | n = 34 | n — 4 |
The adverse reactions in this trial due to the drug were acceptable. Somolesence was seen in few patients but was not a dose limiting side effect. None of the patients developed dreaded extrapyrimadal syndrome, photosensitization, or pigmentation.
Similarly intratumoural injection of CPZ in advanced cancer of cervix treated with radiation has shown radiation sensitization.
Discussion
Chlorpromazine (CPZ) is firmly entrenched as an anti-psychotic, as well as anti-emetic in the clinical practice. It has been routinely used as an
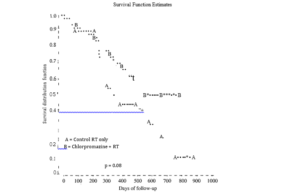
Fig. 1
anti-emetic with chemotherapy and radiation in the past. However, the potential of CPZ molecule to both sensitize and protect tissues against radiation has not been investigated extensively. It is a calmodulin antagonist. Calmodulins are involved in many intracellular enzymatic processes. Are just a few of the reactions controlled by calmodulin. W.Hait and Lee have demonstrated antiproliferative effects of CPZ in different cell lines. They studied the effects of CPZ on both murine and several human cell lines in logarithmic phase of growth. They demonstrated anti-proliferative effects of phenothiazines. They also reported that the cytotoxicity of phenothiazines was not demonstrated until 8 hours ot exposure at a concentration of 32 M [6].
The companion paper by Dr. Singh deals with the possible mechanism of CPZ mediated sensitization and protection in hypoxic and euoxic cells. Abe et.al. repoi ted the effects of anti-proliferative potentials of CPZ. They studied the effects of the drug on IMR-23, neuroblastoma cell lines. They concluded that the anti-proliferative potential is due to the cation radical of CPZ, which has marked non-specific inhibitory effects on various enzymatic process in cells [9]. The toxic effects of CPZ also may be due to the hydrophobic interaction of CPZ, which causes membrane perturbation, which result in cell death. [10]
The toxic anti-proliferative effects are generally seen at a concentration of 10“ to 10‘” M. [2]. Dr. Maniar and Singh have reported inhibition ot DNA repair following high concentration of CPZ in E. coJi. They concluded that cytotoxic potentiel of the drug could be due to changes in fluidity of cell membrane leading to facilitation of influx of oxygen causing ëoxygen effectsî, following radiation [1].
George and Singh have reported inhibition of mouse fibrosarcoma irradiated with 20 Gy of radiation and 25 mgs of CPZ per kg of body weight [11]. This showed an increased concentration of the drug in fibrosacrcoma. Preferential concentration of the drug in tumour leads to higher concentration and a gradient across normal and the tumour tissue, thus favouring sensitization of the tumour. There is a common consensus that CPZ, following administration is preferentially distributed in adrenals, kidney, liver and lungs. This differential distribution in favour of tumour created a gradient where sensitizing effect of CPZ in the tumour and preferentially in hypoxic cells becomes predominant. Chlorpromazine, which acts by its unique action on cell membrane and DNA repair, is a drug that is apart from other sensitizers. A large clinical trial should reinforce the early promise and claims of it being a radiation sensitizer.
Acknowledgement
This work is supported by the Board of Research in Nuclear Sciences (DAE). I also thank Ms. Neela Chatterjee for her help in analyzing the data and prepare the manuscript.
References
- S. Maniar and Singh B.B. Radiosensitizing and radioprotective effects of phenothiazines Maniar Indian Journal of Experimental Biology, Vol. 23; 100— 102: 1985.
- Abe, T. Sekizawa and K.Kogure, Biphasic effects of chlorpromazine on cell viability in a neuroblastoma cell line, Neuroscience Letters 71; 335—339: 1986.
- H. Curry, J. Pharm. Pharmac., 1970, 22: 193.
- Martindale, The Extra Pharmacopoeia 29th Edition; Edited by James E.F. Reynolds; 724; The Pharmaceutical Press, London, 1989.
- Martindale, The Extra Pharmacopoeia 2Sth Edition; Edited by James F. Reynolds; The Pharmaceutical Press, London, 1989.
- N. Hatt and G.L. Lee, Characteristics of the cytotoxic effects of the phenothiazine class of calmodulin antagonists. Biochemical Pharmacology Vol. 32, No. 22: 3973—78: 1985.
- M. Levin and B. Weiss, Ca’2 blocker effects of CPZ are crucial in understanding antiproliferative potential as well as protective. Molecular Pharmacology, 12: 581: 1976.
- Hirai, S. Takeda, S. Natori and K. Sekimizu, Inhibition of SV40 DNA replication in vitro by chlorpromazine. Biol.Pharm. Bull. 16(6); 565—7; Jun 1993.
- Kalyanaranan and P.G. Sohale Generation of free radical intermediates from foreign compounds by neutrophil derived oxidants J.Clin. Investigation 75; 1618—1622; 1985.
- Zilbermans, Y. Gulman and R. Koran, The effects of verapamil, lanthanum and local anesthetics on serotonin release from rabbit platelets. Biochem. Biophys. Acta 691; 10Ml14: 1982.
- C. George and B.B. Singh, Potentiation of radiation response of a mouse fibrosarcoma by phenothiazine drugs. Indian J. of Exp. Biology Vol 22; 305— 307; June 1984.
4. Mitomycin C (MMC) and Radiotherapy
W. Dobrowsky
Sonderabteilung für Strahlentherapie, Krankenhaus der Stadt Wien-Lainz Wolkersbergenstr. 1, A-1130 Vienna, Austria
Introduction
Mitomycin C (MMC) is a natural product isolated from Streptom yces caespitosus and is stable in aqueous solutions but unstable in acidic or basic environment (Crooke and Bradner 1976). MMC is activated by reduction of its quinone moiety, which releases methanol and facilitates opening of the aziridine ring to form an alkylating species. The second alkylating moiety is formed by chemical or enzymatic loss of the carbamate side-chain. The activation of MMC occurs predominantly in hypoxic regions, thereby complementing the radiation induced toxicity, which is higher in well oxygenated tumour regions (Kennedy et al. 1979).
In this article, a brief summary of preclinical data and of clinical data regarding combined therapy of anal cancers and head and neck cancers with the use of MMC are discussed.
Preclinical Studies
It vitro experiments carried out have shown that MMC is more toxic to anoxic or hypoxic cells (Rauth et al. 1983, Rockwell 1983). The differential is shown in many, but not all, rodent cell lines and is quoted to be in the range of 1.5 to 5. The chemical basis for this increased toxicity to hypoxic cells was found to be due to the one electron reduction to the semiquinone and/or two electron reduction to the hydroquinone. This leads to the loss of the methoxy group, opening of the aziridine ring and displacement of the carbamate group producing a bifunctional alkylating agent capable of monoadduct formation and/or intra or interstrand crosslink production in the DNA. In the presence of oxygen, the semiquinone form, and to a lesser degree the hydroquinone, can be back oxidized to the parent compound
where not recommended by the authors. No benefit was seen when compared to historical controls treated by radiotherapy alone (Dobrowsky 1992, Wong et al. 1991). These results are somewhat contrasted to the beneficial effects from the combination (MMC, 5-FU and radiotherapy) in some other cancers like cancers of the oesophagus and head and neck cancers (Keane et al. 1985, Coia et al. 1988, Dobrowsky et a1. 1991, Keane et al. 1985). One reason for the higher incidence of toxicity after treating pelvic recurrent lesions of rectal cancers might be the larger treatment volume and more advanced stage of disease in these patients than in patients with head and neck cancers, where the treatment volume is considerably smaller and patients often in a better performance stage.
After the good results in therapy of squamous cell cancers of the anus, the same combination was used for therapy of squamous cell cancers of the oropharynx and the oral cavity (Dobrowsky et a1. 1991). In a non- randomize study the combination of infusion days 1-5) and 50 Gy achieved a complete response, histologically verified, in 49′ o of all cases. This is clearly a higher percentage than one would assume after radiotherapy alone in these tumors. It was concluded that the administration of MMC and 5-FU increased the radiation effect on the tumour. In most cases a mutilating radical neck dissection could be omitted in all patients, without impairment of prognosis. Furthermore it was seen that the acute mucosal reaction, due to 5-FU administration was more extensive than seen after radiation alone, no late increased toxicity was noticed.
Interesting results were published from the Yale Randomized Trials in 1997. Between 1980 and 1992, two consecutive trials using MMC and Dicumarol as an adjunct to radiation therapy in head and neck cancers were evaluated (Haffty et a1. 1997). A total of 203 patients were enrolled in the two trials. Patients randomized to the drug treatment arm received MMC (15 mg/sqm by IV bolus injections) on day 5 of therapy. Patients scheduled for more than 6 weeks of radiation therapy also received a second dose of MMC (15 mg/sqm) 6 weeks after the initial dose or on the last day of therapy. In case of treatment with additional Dicumarol, this was administered orally (300 mg given them before and 200 mg administered on the day of MMC administration). Hematological toxicity was more frequent in the drug treatment arms, but were of moderate grade and no drug related deaths occurred. Non-hematological toxicities (mucositis) were not increased by additional drug therapy. After a median follow-up of 138 months, a statistically significant benefit was seen in the MMC arms with respect to cause specific survival (p — 0.005), local recurrence (p — 0.002) and local recurrence free survival (p = 0.003), whereas overall survival was not significantly different in the treatment arms (489o afte.r MMC and radiotherapy versus 42 after radiotherapy alone).
In a recently completed randomized trial the effect of MMC to accelerated hyper fractionated radiotherapy was evaluated. From October 1990 to December 1997, 229 patients with squamous cell cancers originating in the neck region were randomized to one of three treatment options (Dobrowsky, submitted 1999. The majority of patients had advanced tumors’ (T3-4: 193/229 = 84%) and lymph node metastases (NI–3: 180/229 = 79%) and most had been considered inoperable by referring specialist. Site of tumor origin was oropharynx in 95, oral cavity in 70, hypopharynx in 39 and larynx in 25 cases, respectively. Standard therapy consisting of conventional fractionation with 70 Gy in 7 weeks in 35 fractions (CF). The second treatment option consisted of a continuous hyper fractionated accelerated radiotherapy delivering a total dose of 55.3 Gy in 33 fractions over 17 consecutive days (V-CHART). The third study a:’m had identical fractionation and dose as the above accelerated treatment, with the additional administration of 20 mg/sqm Mitomycin C on day 5 of treatment (V- CHART + MMC). Main toxicity resulted from accelerated fractionation in confluent mucositis (Grade 3–4 in 95%) requiring nasogastric tube feeding, analgesics and antiphlogistic in the majority of cases. Hematological toxicity Grade 3–4 was seen after MMC administration in 29%. MMC administration did not influence mucosal reaction. Overall duration of mucositis was not different in the three treatment groups.
Local tumour control was 31% after CF, 34% after V-CHART and 48% after V-CHART + MMC, respectively tp < 0.05). Overall crude survival was 279o alter CF, 289c following V-CHART and 399c after V-CHART + MMC, respectively (p < 0.05).
Median follow up was exceeding 48 months, assessment performed February 1999. Pretherapeutic hemoglobin levels showed that these influenced the treatment outcome in CF and V-CHART but not in those cases where accelerated fractionation was combined with MMC (V-CHART
+ MMC). The mean Hb-level in patients who recurred after CF was 12.8 mg/dI vs 13.8 in those who have obtained local tumor control (p — 0.05). After V-CHART the mean was 13.8 in those who are without local recurrence compared to 13.3 in those who recurred (p — 0.07). In those treated by V-CHART + MMC however, the Hb value did not influence local tumor control. The Hb level in those who recurred was 13.2 compared to 13.4 in those who are without local recurrent tumor (p — 0.5).
It was concluded that following shortening overall treatment time from 7 weeks to 17 consecutive days and dose of radiotherapy from 70 Gy to
55.3 Gy the results in the radiotherapy only treated patients were identical. A significant improvement regarding local tumor and survival was seen following administration of MMC to the accelerated fractionated treatment. From a biologic view the findings regarding the Hb seem interesting.
Mitomycin C (MMC) and Radiotherapy 31
A number of authors have pointed out the importance of pretherapeutic Hb-levels and the response to radiation therapy in head and neck cancer s (Dische 1991, Fein et a1. 1994, Naude et al. 1995). With regard to the results considering the pretherapeutic Hb level, it seems that it is of importance for patients undergoing conventional fractionation and accelerated fractionation, but not for those who had additional administration of MMC. This indirectly suggests that hypoxic conditions are important in radiotherapy of head and neck cancers, and that this partly can be overcome by additional treatment with a drug predominately toxic to hypoxic cells. Another explanation for the improvement of tumour cells. In clinical oncology the combination of chemotherapy in those patients undergoing a very short schedul.e radiotherapy is that the addition of MMC might counteract a possible lack in reoxygenation and simultaneous radiotherapy is in may cases causing an increased effect on the tumour. The problem is that also the toxicity in normal tissue increases and very often leads to dose reductions or modifications, so that the combined therapy is not having any increased effect on the tumour. The advantage of MMC is that it apparently has not any increased, sensitising effect on radiation induced toxicity (e.g. mucosa) and that it therefore can be combined without increased local toxiciy. Further studies on the interaction and the differential toxicity are warranted as well as examination of other bioreductive d1’ugs, as Porfiromycin, which has reportedly an even larger differential toxicity towards hypoxic cells and therefore might be of clinical use.
References
Bartelink H., Roelofsen F., Eschwege F., Rougier fi, Bosset J.F., Gonzalez D.G., Pfeiffer D., van Glabbeke M., Pierart M. Concomitant radiotherapy and chemotherapy is superior to radiotherapy alone in the treatment of locally advanced cancer: Results of a phase III randomized trial of the EORTC and Gastrointestinal Cooperative Groups. J. Clin. Oncol. 1997, 15: 2040.
Cora L.R., Paul A.R., Engström P.F: Combined radiation and chemotherapy as primary management of adenocarcinoma of the oesophagus and gastroesophageal junction. Cancer 1988, 61: 643.
C ooke S.T., and Bradner W.T., Mitomycin C: A review. Cancer Treat. Rep. 1976, 3: 121.
Cummings B.J., Keane T.J., O’Sulluvan B., Wong C.S., Catton C.N. Mitomycin in anal carcinoma. Oncology 1993, 50: 63.
Dische S. Radiotherapy and anaemia—the clinical experience. Radiother. Oncol. 1991; 20 (Suppl.): 35.
Dobrowsky W., Dobrowsky E., Strassl H., Braun 0., Gritzmann N., Scheiber Y Combined modality treatment of advanced cancer of the oral cavity and oropharynx. Int. J. Radiat. Oncol. Biol. Phys. 1991, 20: 239.
Dobrowsky W. Mitomycin C. 5-fluorouracil and radiation therapy in advanced, locally recurrent rectal cancer. Br. J. Radiology 1992, 65: 143.
Dobrowsky W., Dobrowsky E., Rauth A.M. Mode of interaction of 5-fluorouracil, radiation and mitomycin C: in vitro studies. Int. J. Oncol. Biol. Phys. 1992, 22: 875.
Dobrowsky W. Continuous hyperfractionated radiotherapy with/without mitomycin C in head and cancer. Initial results from a completed randomised trial. (Submitted for publication 1999).
Fern D.A., Lee W.R., Hanlon A.L., Ridge J.A., Langer CL., Curran W.J., Coia L.R. Pretreatment hemoglobin level influences local control and survival of T1-T2 squamous cell carcinomas of the glottic larynx. J. Clin. Oncol. 1995, 13: 2077.
Hatfty B.G., Son Y.H., Papac R., Sasaki C.T., Weissberg J.B., Fischer D., Rockwell S., Sartorelli A.C., Fischer J.J. Chemotherapy as an adjunct to radiation in the treatment of squamous cell carcinoma of the head and neck: results of the Yale moitomycin randomized trials. J. Clin. Oncol. 1997, 15: 268.
Keane T.J., Harwood A.R., Beale F.A., Cummings Payne D.G., Elhakim T., Rawlinson
E.A. Pilot study of mitomycin C/5-Fluorouraci1 infusion combined with split course radiation therapy for carcinomas of the larynx and hypopharynx. J. Otolaryngol. 1986, 15: 286.
Keane T.J., Harwood A.R., Elhakim T., Rider W.D., Cummings B.J., Ginsberg R.J., Cooper J.C. Radical radiation therapy with 5-fluorouracil infusion and mitomycin C for oesophageal squamous carcinoma. Radiother. Oncol. 1985, 4: 205.
Kennedy K.A., Rockwell S., Sartorelli A.C. Selective metabolic activation of mitomycin C by hypoxic tumor cells in vitro. Proc. Am. Assoc. Cancer Res. 1979, 20: 278.
Naudé J., Dobrowsky W., Wachter S. Prognostic factors in larygeal carcinoma: primary radiotherapy versus postoperative irradiation. In. Smee R, Bridger GP, Editors. Laryngeal cancer. Amsterdam: Elsevier, 1994: 582.
Nigro N.D., Vaitkevicius V.K., Considine B. Combined therapy for cancer of the anal canal: a preliminary report. Dis. Colon Rectum 1974, 17: 354.
Papillon J. and Montbarbon M.D. Epidermoid carcinoma of the anal canal. Dis. Colon Rectum 1987, 30: 324.
Rauth A.M., Mohindra J. K., Tannock I.F. Activiy of mitomycin C for aerobic and hypoxic in vitro and in vivo. Cancer Res. 1983; 43: 4145.
Rockwell S. Cytotoxicities of mitomycin C and X-rays to aerobic and hypoxic cells cell in vitro. Int. J. radiat. Oncol. Biol. Phys. 1983, 8: 1035.
Simpson-Herren L. and Noker P.E. Diversity of penetration of anti-cancer agents into solid tumors. Cell. Prolif. 1991, 24: 355.
UKCCCR Anal Cancer Trial Working Party: Epidermoid anal cancer: results from the UKCCCR randomised trial of radiotherapy alone versus radiotherapy, 5-fluorouraci1, and mitomycin. Lancet 1996, 348: 1049.
Wong C.S., Cummings B.J., Keane T.J., Dobrowsky W., O’Sullivan B., Catton C.N. Combined radiation therapy, mitomycin C, and 5-fluorouracil for locally recurrent rectal carcinoma: results of a pilot study. Int. J. Radiat. Oncol. Biol. Phys. 1991, 21: 1229.
5.In Yiro Disposition and Elimination Kinetics of Senazole Following Intravenous and Intraarterial Administrations in Cancer Patients
Mathewl, C.K.K. Nair and N.G. Huilgol
”2Radiation Biology Division, Bhabha Atomic Research Centre, Mumbai 400 085, India
°Division of Radiation Oncology, Nanavati Hospital and Medical Research Centre, Mumbai 400 056, India
Introduction
A seminal discovery by Thomlinson and Gray (1955) provided the rationale for hypoxia it1 human tumors and that its presence compromises the effectiveness of Radiation Therapy [1]. The oxygen effect, discovered towards the beginning of the century has been interpreted in terms of the fundamental interactions of ionizing radiations with cellular target molecules and the free radicals that are created and has been the basis of efforts to modify the hypoxic cell radiosensitivity with of oxygen mimetic chemical agents generally referred to, as hypoxic cell radiosensitizers [2]. These synthetic chemical agents possess an advantage over oxygen in that they are not rapidly metabolized by the cells they perfuse through, and hence are able to penetrate greater distances. The efficacy of hypoxic cell radiosensitizers was correlated to their electron affinity in early 1960s, paving way to the design and evaluation of many electronegative compounds as radiosensitizers, most of which have failed in the clinic due to dose limiting toxicities. However, a meta-analysis by Overgaard and Horsman
[3] provided a convincing argument that high tumor concentrations achieved by non-toxic hypoxic cell radiosensitizers can compensate for low electron affinity. Senazole is a nitrotriazole derivative synthesized at Kyoto University, Japan [4, 5] and was shown to possess appreciable sensitization efficiency and negligible toxicity in pre-clinical [6] and clinical systems [7]. However, information on its clinical pharmacokinetic profile in cancer patients was scanty and limited to oral administration [8].
Pharmacokinetics is defined as the quantitative, time-dependent changes of both the plasma drug concentration and the total amount of the drug in the body, following administration by various routes. The significance of identifying the pharmacokinetics of a sensitizer lies not only in defining the factors that influence its level and persistence in the body, but also in tailoring the therapeutic use of the sensitizer to improve upon the therapeutic index.
A clinically ideal radiosensitizer should possess a few favorable pharmacokinetic profiles by virtue of moderate half-life of elimination and low peripheral tissue distribution and maximum tumor uptake. The purpose of this investigation was to estimate the plasma levels of Senazole in cancer patients as a function of time, to fit the data into the most appropriate pharmacokinetic model and thereby evaluate the pharmacokinetic parameters of Senazole by various modes of administration.
Drug and Solvents
Senazole was gift from Dr V.T. Kagiya, Kyoto University, Japan. It was tested in the studies without further purification. The sol vents, dichloromethane methanol and acetonitrite were of spectroscopic grade and obtained from M/S Sisco Research Labs., Mumbai. All other chemicals used in this study were of analytical grade.
For clinical studies involving human subjects, Senazole powder was reconstituted according to the specifications of ICRP (International co- coordinated Research Program) at an FDI approved manufacturing unit of a pharmaceutical company. Senazole was dissolved as 2% solution in saline, filtered through 0.2 Jr Millipore filters and sealed in 50 ml vials before sterilizing them by end sterilization method at 115 °C, 15 lb pressure for 60 minutes. Parenteral presentation was as 50 ml vials, which were preserved under refrigeration away from light until use.
Patients and Samples
Patients undergoing radiation therapy for stage III carcinoma of the cervix or head and neck were accrued for the trials, with their informed consent. Eligibility criteria included histologically proven epithelial cancers (carcinomas) of the uterine cervix or head and neck staged T3 or above with any combination of nodes and metastasis. All patients chosen had a Karnofsky Index of 80 and above, with reasonable life expectancy, though they had advanced disease. Patients with associated hypertension, diabetes mellitus, autoimmune-disease and renal insufficiency were excluded from the study. Routing work up included haemogram, blood chemist1’y, ECG, renal function and chest X-ray (PA). Details of the patient profile are shown in Table 1.
“fable 1. Patients’ details for intravenous pharmacokinetic modelling and analysis
| Subject | Sex | Age | Weight | Height | Diagnosis | Total Dose |
| (Yr) | (kg) | (cm) | (AU 2123) | |||
| Patient 1 | F | 55 | 50 | 138 | Cancer cervix lIIb | 0.8 |
| Patient 2 | F | 42 | 67 | 164 | Cancer cervix IIIb | 1.04 |
| Patient 3 | F | 36 | 42 | 142 | Cancer cervix IIIb | 0.77 |
| Patient 4 | F | 60 | 35 | 142 | Cancer cervix IIIb | 0.71 |
| Patient 5 | F | 71 | 54 | 152 | Cancer cervix IIIb | 0.9 |
| Patient 6 | M | 50 | 45 | 152 | Cancer Tonsil | 0.9 |
| Patient 7 | M | 45 | 65 | 187 | Cancer Tonsil | 0.99 |
| Patient 8 | F | 60 | 38 | 142 | Cancer cervix IIIb | 0.74 |
| Patient 9 | F | 60 | 58 | 154.5 | Cancer cervix IIIb | 0.5 |
| Patient 10 | F | 60 | 47 | 152.4 | Cancer cervix IIIb | 0.84 |
| Patient 11 | F | 61 | 49 | 150 | Cancer ccr vix IIIb | 0.85 |
| Patient 12 | F | 60 | 47 | 152 | Cancer cervix IIIb | 0.84 |
| Patient 13 | F | 60 | 49 | 155 | Cancer cervix IIlb | 0.86 |
Drug Administration and Pharmacokinetic Studies Pharmacokinetic modelling and analysis of the drug was done for 13 patients listed in Table 1, following i.v. administration and on 5 patients after i.a. administration. Patients were administered AK 2123 (Senazole) at a dose of 0.6 g per m2, for intravenous administration.
Intravenous Administration
AK 2123 was administered (0.6 g/m2) to thirteen patients with advanced cervical cancer staged III b with no hepatic or renal abnormalities. Exact protocol was reviewed and accepted by the hospital ethical committee; physical characteristics of the patients are summarized in Table 1. Infusion of the drug was done int1’avenously into the peripheral vein by the use of a cannula for 20 minutes, half an hour prior to the radiation therapy.
Intraarterial Administration
Patients with recurrent cancer of the cervix whei e prognosis were bleak were chosen for the intraartei ial administration with embolization. The uterine arteries on both sides were cannulated by trans femoral approach using a French i enal double curve catheter. Whenever the uterine artery cannulation was not possible, the anterior division of the internal iliac artery was cannulated. AK 2123 (Senazole), to a total dose of 2-4 g was infused as a sterile 2Vo saline solution after confirming the catheter position and demonstrating abnot’mal tumour vascularity. This was followed by the embolization of the artery using a particulate gel foam to enhance the tumour hypoxia, a condition where AK 2123 (Senazole) a hypoxic cell radio sensitizer was expected to act at its best.
Collection of Blood Samples
Serial venous blood samples were collected from patients before and up to approx. 24 hours after the drug administration, at various intervals up to 25 hours post administration of Senazole. In case of intra-arterial administration samples were collected for 36 hours. An 18 G intravenous cannula was inserted in the arm and 2 ml blood sample was initially taken each time and discarded. This was immediately followed by 5 ml blood, which was collected in heparinized vials and placed on ice. Cannula was then flushed with 2 ml sterile saline to prevent clotting. Plasma was then, separated by centrifugation at 3300 x g for 10 minutes, and stored at
—70°C until drug was extracted along with internal standard for HPLC analysis.
Standardization of HPLC Methodology and Analysis
HPLC methodology for the separation and quantification of Senazole was developed on an Applied Biosystems HPLC, with isocratic and gradient elusion modes and an variable wavelength uv-visible spectrophotometer, using a reverse phase (ODS Jr-Bonde pack, Emerck C18) column. The absorbance of the eluant was measured and the peaks were integrated using spectra Physics Integrator cum Plotter. Various solvent systems and flow rates were tried to optimize the retention time and peak characteristics of Senazole. In order to calibrate the detector response and also to take care of the inter-sample variations, 50 [i1 aliquots of misonidazole stock solution (7 mM in nanopore water) were added as internal standard to each of the 0.5 nil sample of blank plasma spiked with Senazole in the concentration range of 0-50 Jig/ml. The mixture was thoroughly vortexed for one minute, and equilibrated within 5 ml dichloromethane, for 5 minutes by vigorous shaking. 4 ml of the organic phase (dichloromethane) was removed from each sample by a micropippette, evaporated to dryness and reconstituted l09oaqueous methanol, which were used as the mobile phase. This reconstituted solution (100 (1) was subjected to further HPLC analysis. The standard equation of the calibration line using the slope of the line and the Y intercept was used for calculating the unknown Senazole concentration from the area under the peak as obtained form the detected response of the patients sample data.
HPLC method developed above was used in separating the drug from biofluids such as blood, urine and saliva collected from cancer patients treated with Senazole.
Patient samples were treated exactly as described for standard Senazole solutions and the chromatograms were obtained. From the integrated peak areas of these chromatograms and using the standard equation of the calibration curve, the actual concentrations of Senazole in g/ml were calculated. These values were plotted as a function of the time of sample collection and were subjected to the pharmacokinetics modelling to choose the most appropriate pharmacokinetic model and the pharmacokinetic parameters were calculated from the model using standard equations.
Pharmacokinetic Modelling of the Data
HPLC analysis data from patients administered with Senazole intravenously an intra-areterially wete fitted to standard pharmacokinetic one compartment and multi-compartment models, using non-linear least square regression method. The best-suited model was chosen on the basis of the model index parameter, which is proportional to the magnitude of the sum of residuals. The data was found to fit the two compartment open model.
Cz — Cue”” + Cte2,’
— D(Kz – ) e ” + D(Kz› — >z)e *’
V1(32 — 11) (V1(32 – Al)
The pharmacokinetic parameters AUC, t/2e’ V9 Vc’ Vp# CLV M•S•C•9 K21′ Kit and K 2 determined from the data using the standard equation from Rowland et. a1. [9).
Pharmacokinetic modeling was done on the basis of model selection criteria (M.S.C.), a parameter defined in iRSTRIPi (MicroMath, Salt Lake city, UT, USA) ñ an iterative least square regression package for pharmacokinetic analysis, which signifies the goodness of fit to the particular model, the higher M.S.C. being associated with better fit. Major Pharmacokinetic parameters, their standard equations, units and clinical significance in two-compartment open model system adapted from Roland and Tozer (1989) and are summarized in Table 2.
Studies on Metabolism of Senazole
Urine samples were collected from a Senazole treated patient for 24 hr post-drug administration and analyzed for the presence of urinary metabolites of Senazole. The urine samples were concentrated by evaporating with slow heating in a water bath under gentle current of nitrogen gas. The urine concentrate which contained the unmodified drug and its possible bio-transformed products eliminated through urine, were subjected to solvent
Table 2. Details of pharmacokinetic parameters
| 1. | Area under | AUC AUC – | kg hr/ml” | AUC — | Signifies the extent |
| the curve | AU | = (ti. — ‹) | of drug exposure to | ||
| (C, + C, i) 2 | the body | ||||
| 2. | Half-life | T 2d | Hr | i /2d | Fastness of |
|
Elimination |
T/2, |
hr |
= 0.693Jt
ti/2e |
distribution in the central compartment
Fastness of |
| Central | V, | L | Vc — D€lse In central
(Cj + C2) compartrr.ent |
|
| Peripheral | Vp | L | Vp = V — Vt In peripheral | |
| compartment | ||||
| 4. | Plasma | CL | Lhr- ’ | CL = Dose/ Signifies the |
| clearance | (AVC0 ) volume of | |||
| biological fluid | ||||
| (blood) that must | ||||
| be cleared of the | ||||
| drug in unit time in | ||||
| order to account | ||||
| for (plasma) | ||||
| clearance | ||||
| 5. | Intercom- partmehtal transfer rate constant | Ki› Kit
Kl2 |
H,- H, _{
H,— |
(I{C t + zCz) Signifies the rate of
(C, + C ) intercompart- I z)/Kzi mental transfer of + 2- the drug Kzi — kio |
extraction using ethyl acetate as the solvent. The organic fractions after the repeated extractions were pooled in and evaporated at room temperature using flash evaporation and the residue was reconstituted with 10% methanol in water.
Results and Discussion
The mean plasma concentration—time profile of these 13 patients is shown in figure 1 for intravenous administration. By intravenous administration the drug reaches peak plasma concentration almost instantaneously. Plasma pharmacokinetics of Senazole in cancer patients conformed to the two compartmental open model.
Table 3. Mean pharmacokinetic parameters for intravenous administration
| Sr. No. | Parameter | Mean estimate (I.V.) |
| 1 | ABC(o—i› | 202.25 ptg hr/ml |
| 2 | A UC 0_ | 217.29 (g hr/ml |
| 3 | AUC(o ) 8 | 285. 16 (g hr/ml/g |
| 4 | Ti/2d | 0.368 hr |
| 5 | T /2el | 5.18 hr |
| 6 | Cmax | 50.00 (g/g |
| 7
9 |
V V«
Vp |
26.35 L
16.18 L 10.17 L |
| 10 | Kiz | 0.5733 hr*’ |
| 11 | Kio | 0.2782 hr‘‘ |
| 12 | Kio | 0.2049 hr‘’ |
| 13 | CL | 3.7300 L hr*’ |
The elimination of the drug from plasma followed a bi-exponential decay, which indicates that there is a central and a peripheral compartment representing the fast perfusing and slow perfusing tissues with which Senazole gets equilibrated at two different rates. The central compartment normally consists of liver, circulating system, kidney, heart and such fast proliferating tissues. The peripheral compartment normally is composed of slowly perfused tissues such as fats and muscles. Tumour with its rich vascularisation at the peripheries often represents the central compartment. The drug is distributed in both the compartments.
Senazole showed a terminal elimination half-life of 5.18 hrs by intravenous administration. The mean important pharmacokinetic parameters of Senazole intravenous administration is summarized in Table 3 and the model selection criteria fOr patients are present in Table 4.
Apparent volume of distribution (V) of the drug is a very important pharmacokinetic parameter signifying the distribution for the drug in the body. Higher the value of V, higher is the distribution and binding of the dt ug. The apparent volume (V) estimated from the data was 26.35 liters for Senazole by intravenous administration. The ratio of volumes of distribution in central to that of the peripheral was 16.18:10.17. From the ratio it is clear that, though the drug is distributed relatively more in the central compartment, its distribution characteristics do not differ much in the central and peripheral compartments. This trend is further supported by the inter-compartmental transfer rate constants K 2, K2 ffnd Kit which follow the trend K2t > Kt2 >K 0 S›howing that the drugis tendency is to be
confined to the central compartment rather than the peripheral. It is obvious that the drug Senazole is eliminated from the central compartment. It has two-compartment disposition. The plasma clearance of the drug was estimated to be 0.3730 L/hr.
Table 4. Pharmacokinetic Model Selection in Intravenous Patients
| Patient | Model (Best fit) | MSC |
| Patient # 1 | 2 compartmental-open | 3.65 |
| Patient # 2 | 2 compartmental-open | 6.00 |
| Patient # 3 | 1 compartmental-open | 4.33 |
| Patient # 4 | l compartmental-open | 3.67 |
| Patient # 5 | 2 compartinental-open | 6.95 |
| Patient # 6 | 2 compartmental-open | 3.43 |
| Patient # 7 | 2 compartmental-open | 3.66 |
| Patient # 8 | 2 compartmental-open | 4.12 |
| Patient # 9 | 2 compartmental-open | 5.50 |
| Patient # 10 | 2 compartmental-open | 3.23 |
| Patient # 11 | 2 compartmental-open | 7.58 |
| Patient # 12 | 2 compartmental-open | 3.21 |
Model Selection Criteria as calculated using the computer software “RSTRIP”
In case of intra-arterial administration, the drug reached a peak plasma concentration in 1.33 hr. This is understandable because the drug was first directly infused into the artery that nourishes the tumour. Moreover further blood flow was locked by a gel foam to induce hypoxia in the tumour. Hence the drug had to be slowly absorbed into the venous circulation before it is distributed all over the body. By this mode of administration the drug was eliminated from plasma with a terminal elimination half-life of 5.12 hr. This value did not vary significantly at all from the intravenous mode suggesting that the elimination kinetics of this drug is independent of the mode of administration.
An interesting fact to note, in intra-arterial administration was that the ratio of volumes of distribution in the central to the peripheral, compartments was 19.61:3.86. This would suggest that the drug had a higher tendency to be confined to the central compartment and its peripheral tissue distribution was significantly reduced and compared to i.v. administration. It was also noted that the distribution half-life of the drug by intra-arterial administration was significantly higher than that obtained for intravenous administration. The increased distribution half life and high volume of distribution in the central compartment would suggest that the drug has a prolonged retention in the central compartment even though its elimination from plasma was not affected by the mode of administration. Increased tendency of Senazole to be confined to the central compartment is further supported by the inter-compartmental transfer rate constants, which followed the same trend as was found for intravenous administration. There was no significant difference in the other parameters such as plasma clearance
AUC (0 )t Cmaxetc- When adjusted for the same dose. The mean pharmacokinetic parameters for intra-arterial administration and the patient details are given in Table 5 and 6 respectively. In summary, intravenous and intra-arterial administrations demonstrated very favorable pharmacokinetic profile for the drug. However, intra-arterial administration has many advantages over intravenous administration.
Table S. Intraarterial administration—Patients’ details
| Subject | Sex | Age | Diagnosis | Total dose administered (g) |
| Patient l | F | 55 | Recurrent CaCx | 1.5 + 1.5 = 3 |
| Patient 2 | F | 30 | ” | 2.0 + 2.0 = 4 |
| Patient 3 | F | 50 | ” | 2.0 + 2.0 = 4 |
| Patient 4 | F | 65 | ” | 2.0 + 2.0 = 4 |
| Patient 5 | F | 30 | ” | 2.0 + 2.0 = 4 |
Table 6. Mean pharmacokinetic parameters for sanazole by intraarterial administration
| 1 | 1144.68 yg hr/ml | |
| 2 | 1270.97 yg hr/ml | |
| 3 | 303.72 kg hr/ml.g | |
| 4 | T,/2d | 2.880 hr |
| 5 | Ti/z‹a | 5.12 hr |
| 6 | Inal | 50.00 kg/ml. g |
| 7 | V. | 23.47 L |
| 8 | Vc | 19.61 L |
| 9 | p | 3.86 L |
| 10 | Kiz | 0.2899 hr* |
| 11 | Kzi | 0.5943 hr*’ |
| 12 | Kt | 0.16180 hr*’ |
| 13 | CL | 3.8900 L hr*’ |
Intra-arterial administration has been tried earlier for radiosensitizers in animal models (10). However, this approach has been totally new in clinical hypoxic radiosensitization. Intra-arterial administration with embollization increased the tumour retention of the drug and reduced peripheral tissue distribution, without compromising the mean venous plasma levels, area under the curve or elimination characteristics and it could be a preferable mode in extremely advanced i ecurrent diseases with torrential bleeding for which intra-arterial embolization is considered. However, for daily dosing in adjuvant radiation therapy one has to resort to intravenous administration. The moderate elimination half-life of 5.18 hr makes Senazole a very suitable drug for daily dosing intravenously.
The drug displayed a linear relationship between area under the curve (AUC) and the dose administered. This was obsei ved in both intravenous as well as intra-arterial cases. AUC depends on the administered dose.
Analysis of the Metabolites
The reconstituted urine extract when subjected to high performance liquid chromatography (HPLC) showed up two extra peaks along with Senazole and numerous other peaks. These two peaks consistently appeared in all the urine samples.
In an attempt to identify these metabolites, as a preliminary step, two molecules were prepared from Senazole, which could be thought of as the most probably biotransformation products of the drug. In one case the intro group of Senazole was reduced to amino group and the other, the side chain was hydrolyzed into a carboxylic acid group, as nitroimidazole and triazoles are known to undergo intracellular enzyme activated reduction of their nitro group to an amino group and the amide linkage with side chain of the molecule could be hydrolyzed by amidases. Senazole undergoes conversion in vivo and a 3-amino derivative and a derivative 3ith carboxylic acid group on the side chain are formed, which are excreted through the urine. A LC-MS (liquid chromatography ñ mass spectrometry) analysis may be useful in for the confirming the presence of these compounds in urine.
Conclusion
The tumor could represent the central compartment with its high vascularity even though it may harbor a hypoxic core due to high oxygen consumption of the malignant cells surrounding them and the resultant depletion of oxygen perfusion. The diffusion of the sensitizer was not affected because it was not rapidly metabolized by the cells they pass through. Senazole showed an increased affinity towards the central compartment.
Senazole possesses a very favorable pharmacokinetic profile for its use as a hypoxic radiosensitizer in the clinic, as evidenced by the elimination profile and higher VVp ratio.
The peripheral tissue distribution of Senazole is found to be low and the plasma elimination moderately fast. The ideal plasma elimination half-life of 5.18 hr makes it an ideal chemical adjuvant for use in radiation therapy of cancer, especially for daily dosing.
A significant reduction in peripheral distribution and increased tumor
drug concentration were achieved by administering the drug intra-arterially. The tumor radiosensitizer potential was enhanced as a result of inducing tumor hypoxia by embolizing the artery to the tumor. Presence of two metabolites of this drug in the urine of the patients suggests that, the drug is first cleaved at the amide linkage and the acid is excreted as such. It may be possible that the pharmacological action of these metabolites contributed in the occasional transient neuropathy observed in patients with multiple dosing. However, confirmation of these metabolites have to be done by IR. NMR and LC-MS (Liquid Chromatography ñ Mass Spectrometry).
References
- Thomlinson H. and Gray, L.H. British J. Cancer, 9—539—549, 1955.
- Aclams E. and Cooke M.S. Int. of Radiation Biology 15, 457–71, 1969
- Overgaard, and Horsman, M.R. Seminars in Radiation Oncology, 6(1), 11— 21, 1996.
- Jenkins C., Naylor MA,O’Neil P, Threadgill M.D., Cole S. and Stratford I.J., J. Med. Chem. 33, 2603, 1990
- Kagiya and Nishimoto S, Free Radical Research Communication 6, 1053, 1989
- Kagiya V., Researcher’s Booklet of the Fundamental and Clinical Studies on Senazole (AK 2123), Health Research Founclation, Kyoto, Japan, 1998
- Huilgol, G., Chatterjee, N.A. and Mehta, A.R., Int. J. Radiat. Oncol. Biol. Physics, 32, 5, 1996
- Huan, L.C. and B.Y., Int. J. Radiat. Oncol. Biol. Physics., 29(3), 607—10, 1994
- Rowland and Tozer, T., In Clinical Pharmacokinetics: Concepts and Applications, Lea and Felliger, USA p318, 1989
- Ebe, Suda H., Kuramitzu,’I’.Honma, Y., Nakanishi, t., Miyata, Y., Sakaguchi, M and Matsunaga, N.Int.I.Radiat,Oncol.Biol.Physics, 36, 1107, 1956
6. Nitrotriazole-Spermidine Conjugate: A DNA selective radiosensitizer
C.K.K. Nairl, R. Mathew4, S.R. VenkatachaIam2,
N.G. Huilgol4 and S. Kapoor3
Radiation Biology Division, 2Bio-Organic Division, Radiation Chemistry and Chemical Dynamics Division, Bhabha Atomic Research Centre,
Mumbai 400085, India
Division of Radiation Oncology, Nanavati Hospital and Medical Research Centre, Mumbai 400056, India
Abstract
A novel DNA selective nitrotriazole-spermidine conjugate [NTS) was synthesized by coupling 3-nitro, 1, 2, 4-triazolyl acetic acid and spermidine free base. The new compound, [NTS] was resistant to gamma radiolysis upto 350 Gy. Pulse radiolysis studies showed a solvated eletron reaction rate constant of 1.0 x l0′0 M‘ s ‘. The conjugate [NTS] sensitized hypoxic Escherichia coli cells to gamma radiatioin at 1.0 x 10 M concentration while sanazole (AK-2123), a non- polyamirio-nitrotriazolyl derivative, showed sensitization only at 1.0 x 10‘3 M. The observed high radiosensitization in case of [NTSI could be due to its specific binding to DNA and consequent inhibition of DNA repair, in addition to the enhancement of free radical damage, as spermidine moiety would confer strong binding to DNA and nitrotriazole moiety would bring about the hypoxic radiosensitization ability.
Introduction
The development of compounds which differentially increase the radiosensitivity of tumour cells could be a solution for the failure of radiotherapy of some tumours. There is a strong evidence that cellular hypoxia influences the radiocurability of several human malignancies [1-8J. Well oxygenated cells are more sensitive to ionising radiation than hypoxic cells. Oxygen has strong electron affinity and is the best known radiosensitizer [9]. There is a strong correlation between electron affinity of a molecule and its potency as a hypoxic cell sensitizer [10]. The biological properties of nitroheterocyclic compounds have been attributed to their redox properties [11, 12]. The correlation between one electron reduction potential and the ability of these compounds to act as radiosensitizers for hypoxic cells led to the development of a number of nitroheterocyclic compounds as adjuvants in radiotherapy (9]. Among these nitroimidazoles and nitrotriazoles constitute a unique class of hypoxic cell radiosensitizers [9, 13—19]. many of these compounds have undergone extensive clinical investigations and some of them were discarded due to neurotoxicity and other side effects while a few are found to be highly promising [14,18, 20].
Several nitrotriazole derivatives such as sanazole are under clinical trials as adjuvants in radiation therapy of cancer [15—18]. However these derivatives are not designed to have any cellular site specificity. Molecules can be targeted to specific cellular sites by attaching chemical moieties or groups having specific affinity for such sites [21—24]. Thus by attaching a polyamine like spermidine, a nitrotriazole compound can be targeted to DNA [21]. We have synthesized a new DNA selective nitrotriazole conjugate [NTS] by coupling 3-nitro, 1, 2, 4-triazolyl acetic acid and spermidine. As spermidine binds to the minor groove of DNA and nitrotriazole moiety confers hypoxic cell radiosensitizing property, the new compound would be a DNA selective hypoxic radiosensitizer. This paper reports our studies on this new compound, (NTS].
Materials and Methods
Sanazole (AK2123) was obtained from Dr. V.T. Kagia, Kyoto University, Japan. This drug was used in the studies without further purification. Spermidine was obtained from Sigma Chemical Co. The solvents dichloromethane was of spectroscopic grade and obtained from M/S Sisco Research Labs., Mumbai. All other chemicals used in this study were of analytical grade.
Synthesis and Characterisation of Nitrotriazole-Spermidine
Conjugate (NTS)
A new compound, a conjugate of nitrotriazole and spermidine, [NTS] (nitrotriazolyl-acetyl-spermidine) was synthesised by and coupling 3-nitro, 1, 2, 4-triazoly1 acetic acid and spermidine with dicyclohexyl-carbodi- imide (DCC) as the coupling agent. When the reaction mixture was subjected to thin layer chromatography (TLC) with methanol: chloroform mixture at 3 : 97 ratio as the mobile phase, the product [NTS] appeared as purple brown spot different from that of the reactants. This distinct band of [NTS), when cut and extracted with 40 : d0 chloroform : ethyl acetate
mixture, reconstituted with 109a methanol and analysed on HPLC, gave a distinctly different chromatogram from that of the reactants separately. Structural characterisation was done using mass and infrared spectroscopy.
Thin Layer Chromatography
TLC was performed on preconditioned silica gel plates, with appropriate solvent systems including methanol, chloroform and diethyl amine, and viewed against green background under low field U.V. of wavelength 248 nm. The fit value was noted and the purity of the compound was checked. For preparative TLC, plates were coated with silica gel (GF, 254) dried and activated in oven at 105 °C. The chromatogram was developed in a chamber saturated with the mobile phase by ascending technique. The spot was detected under U.V. light at 248 nm. The colour of the spot and the f value were noted.
High Pressure Liquid Chromatography (HPLC)
HPLC methodology for the separation and quantification of AK-2123 and NTS was developed on an Applied Biosystems HPLC with isocratic and gradient elusion modes using a reverse phase (ODS-Bonde pack, Emerck C18) column. The absorbance of the eluant was measured at 248 rim and the peaks were integrated and recorded using a Spectra Physics integrator cum plotter. Various solvent systems and flow rates were tried to optimise the retention time and peak.
Spectroscopic Studies
U.V. spectrum of AK-2123 and NTS were recorded in water and dichloromethane using a Varian 200 UV/visible spectrophotometer. I.R. spectrum of the compound was recorded as a potassium bromide pellet and various wave numbers were assigned to the known functional group in the molecule.
Pulse Radiolytic Studies
7 MeV electron pulses of with 50 ns from a LINAC with a kinetic spectrophotometer arrangement and an on-line computer facility were used for the determination of reaction rate constants of NTS with aquated electrons (e.,q-) [25]. The diameter of the beam was 2 mm at the exit window and the typical dose/pulse was -16 Gy. In order to study the contribution due to (eaq) appropriate scavenger like t-butano1 was used. Concentration of NTS used for pulse radiolysis was 1 x l0°4 M and the solutions were prepared in nanopure water. The solutions were flushed with lolar grade nitrogen with purity greater than 99.9% The transient absorption spectrum of the compounds with fill‘ was recorded. The kinetics of bimolecular reaction of the compounds with aq— WaS Calculated from the slope of the linear plot ofJobsvs solute concentration. The decay of the transients formed in nitrogen saturated solution of the compounds were followed.
Steady State Gamma Radiolysis
Steady state gamma radiolysis of AK-2123 and NTS was studied by irradiating the solutions in a 60Co Gamma Cell 220 (Atomic Energy Canada Ltd., Canada) at a dose of 17 Gy/min. The solutions of AK-2123 and NTS (10“ M ) and various alcohols (0.2 M) were prepared in 0.1 M phosphate buffer of pH 7 for these studies.
Studies on Bacterial Survival
Escherichiu coli AB 1157 was a stock strain maintained in our laboratory. Bacterial cells from a single colony were inoculated and cultured in Luria broth (1% Bacto Tryptone, 0.39c Yeast extract and 0.5% NaCl), at 37 °C overnight. Cells were harvested, washed off the medium with sterile buffer (0.067 M phosphate) and resuspended, in the buffer at a density of 1 x 10′ cells/ml, keeping always on ice. An aliquot of the cell suspension was treated with 5 x 10 3 M AK-2123 or 1 x 10a M NTS in 0.067 M phosphate buffer. Control and the treated cells were exposed to various doses of gamma radiation under oxygen free nitrogen at ice-temperature in the “Co-Gamma Cell 220. After irradiation the cells were plated on Luria agar plates (Luria Broth with 1.5% agar agar) following serial dilution. The plates were incubated overnight and the colonies were manually counted. The surviving fractions were plotted against the irradiation doses in Gy.
Results and Discussion
Characterization of NTS
The formation of NTS during the coupling of 3-nitro 1,2,5 triazolyl acetic acid and spermidine in presence of DCC was monitored by TLC with mehanol : chloroform 3 : 97 as moving phase. The R values of NTS, the reactants and AK-2123 under the experimental conditions are presented in Table 1. NTS has distinct HPLC elution profile compared to 3-nitro, 1, 2, 4-triazolyl acetic acid and AK-2123. The HPLC-retention times for NTS, 3-nitro 1, 2, 5 triazolyl acetic acid and AK-2123, with methanol : water 10:90 as the mobile phase are presented in Table 2.
The IR spectrum of NTS is given in Figure 1. It can be seen that NTS has the characteristc amide (-CO-NH-) band in the IR spectrum unlike the 3-nitro 1,2,4-triazolyl acetic acid. The NTS was further characterized by
HPLC retention times were determined on an applied biosystems instrument using RP-ODS micro-Bonde Pack C-18 column, with isocratic elution using water : methanol (90 : 10) at a flow rate of 1.0 ml per min.
mass spectroscopy and NMR. The results of these studies are not presented here. The ultraviolet absorption profiles of 3-nitro, 1, 2, 4-triazo1y1 acetic acid, NTS, and AK-2123 showed absorption at 248 nm characteristic of the nitrotriazole moiety.
Radiolytic Studies
The transient spectrum obtained on reaction of e.,q- with NTS exhibits an absorption band with max at 295 rim and a shoulder at 310 rim. The rate constant for the reaction of e. q- with NTS was determined by the kinetics of formation of the transient at 295 nm and by e,q- decay at 700 nm. The
band was observed to grow by the first other kinetics and the ob› increased linearly with NTS concentration between 0.1 and 1.0 x 10“ mol dm . The biomolecular rate constant as obtained from the slope of the linear
plot of kobs vs solute concentration was 1.0 x 10’0 dm*’mol*’s*’. The
transient showed spectral characteristics similar to AK-2123 [25]. The results on radiolysis presented in Table 3 reveal that [NTS] is more resistant to radiolysis than AK-2123.
Table 3. Steady state gamma radiolysis (350 Gy) of aqueous solutions of [NTS] and sanazole (AK-2123)
| Degradation of irradiation | (G value) |
Ak-2123 |
fNTS] |
AK-2123 |
[NTSI
N2O |
| OH (5.6) | 1.55 x 10’0 | — | 90 | ||
| N2(tbuvo) | e, – (2.8) | 3.6 x 10′0 | 1,0 x 101’ | 47 | 0.0 |
The compounds, [NTS] and AK-2123 at a concentration of 1 x 10*’ mole dm‘’ were exposed to 350 Gy gamma radiation under different conditions.
It has been reported earlier that nitro radiosensitizers such as metronidazole and other imidazoles also react with eaq- And OH [9—11, 13, ld]. The transients of metronidazole produced after the reaction with hydrated electron and hydroxyl radicals show absorption in the same spectral region as observed in the case of AK-2123. However, slight difference was observed at length greater than 340 nm region which could be due to the low molar absorptivity of the transients produced in [NTS].
Studies on Bacterial Survival
Figure 2 shows the survival curve for E. coli AB1157 cells irradiated with various doses of gamma radiation under hypoxic in the presence and absence of the NTS at a concentration of 1.0 x 10 M. It has been observed that this new compound sensitized the E. coli cells even at a low concentration such as 1.0 x 10 M while AK2123 sensitised thess cells under the same conditions only at a concentration of 1.0 x 10‘3 M or higher. Thus the modification of the 3-nitro, 1, 2, 4-triazole moiety to have groove specific DNA binding ability, by attaching a polyamine moiety on the side chain has resulted in the enhancement of radio sensitization almost 10 fold in molar terms.
Fig. 2. Survival of Escherichia cofi AB1157 after exposure to gamma radiation in presence and absence of radiosensitizers. Bacterial cells at a density of 1 x 10′ cells per ml were exposed to gamma radiation under N2 in 0.067 M sodium phosphate buffer pH 7.0 in the presence of 1 x 10‘ M [NTS] or 5 x 10*’ M AK-2123. Control (A); [NTS] (O); AK-2123 (O).
Polyamines including spermidine are known to cause DNA condensation and are shown to be radioprotectors due to their DNA groove binding property. It has been established that spermidine binds to the minor groove of the DNA and move along the groove in a threading like manner [9, 241 in general were radioorotective in nature with DNA strand breaks as the end point. AK-2123 with its nitrotriazole moiety enhanced the yield of radiation induced DNA strand breaks [17, 18]. The radiosensitization property can be enhanced on targeting of the sensitizer to the DNA and also on the mobility of the sensitizer along the DNA back bone [22, 23]. It is known that radiosensitization ability is compromised when electron affinic compounds have strong DNA binding properties due to the restriction ot mobility along the DNA back bone. Spermidine being highly interacting with DNA and having appreciable mobility (threading effect) along the minor groove of the DNA is useful for targeting an elctron affinic sensitizer to DNA. The enhancement in radiosensitization could be a product of many factors such as the DNA minor groove binding of the radiosensitizers, the mobility along the target DNA molecule, and the hypoxia specific bioreductive activation of the nitro heterocyclic moiety which imparts hypoxia selectivity. It is also quite possible that the molecule inhibits the intracellular DNA repair.
The present compound NTS containing spermidine and nitro triazole exhibited high radiosensitization efficiency in bacterial cells. The observed higher radiosensitizing ability could be due to the amplification of radiation induced damage to DNA or the inhibition of the DNA repair in irradiated cells due to its interaction with cellular DNA. The present experiments do not distinguish between these two possibilities.
References
- B. Parliament, J.D. Chapman, R.C. Urtasun, A.J. McEwan, L. Golberg,
J.R. Mercer, R.H. Mannan and L.I. Wiebe, Non-invasive assessment of human tumour hypoxia with ’°’I-iodoazomycin arabinoside: preliminary report of a clinical study, Br. J. Cancer, 65, 90—95, 1992.
- M. Henk and C.W. Smith, Radiotherapy and hyperbaric oxygen in head and neck cancer: intrim report of second clinical trial, Lancet, 2, 104,1977.
- G. Graeber, C. Osmanian, T. Jacks, D.E. Housman, C.J. Koch, S.W. Lowe and A.J. Giaccia, Hypoxia-mediated selection of cells with diminished apoptotic potential in solid tumours, Nature, 379, 88-91, 1996.
- A. Gatenby, H.B. Kessler, J.S. Rosenblum, L.R. Coia, P.J. Moldofsky, W.H. Hartz and G.J. Broder, Oxygen distribution in squamous cell carcinoma metastases and its relationship to the outcome of radiation therapy, Int. J. Radiat. Oncol. Biol. Phys., 14, 833—838, 1988.
J.M. Brown and A.J. Giaccia, Tumour hypoxia : the picture has changed in the 1990s, Int. J. Radiat. Biol., 65, 95—102, 1994.
- Hockel, C. Knoop, K. Schlenger, B. Vorndran, E. Baussman, M. Mitze,
P.G. Knapstein and P. Vaupel, Intratumoral pO2 predicts survival in advanced cancer of the uterine cervix, Radiother. Oncol., 26, 45—50, 1993.
- Okunieff, M. Hoeckel, E.P. Dunphy, K. Schlenger, C. Knoop and P. vaupel, Oxygen tension distributions are sufficient to explain the local response of human breast tumours treated with radiation alone, Int. J. Radiat. Oncol. Biol. Phys., 26, 631—636, 1993.
- A. Shenoy and B.B. Singh, Chemical radiosensitzers in cancer therapy, Cancer Investigation, 10, 533—551, 1992.
- E. Adams and M.S. Cooke Electron affinic sensitization. 1.A structural basis tor chemical radiosensitizers in bacteria, Int. J. Radiat. Biol., 15, 457— 471, 1969.
- E. Adams, I.J. Stratford, R.G. Wallace, P. Wardman and M.E. Watts, Toxicity of nitrocompounds towards hypoxic mammalian cells in vitro: dependence on reduction potential, J. Nat1. Cancer Inst., 64, 554—560, 1980.
- J. Stratford. P.O’Neil, P.W. Sheldon, A.R.J. Silver, J.M. Walling and G.E. Adams, RSU 1069, a nitroimidazole containig an aziridine group, Biochem. Pharmacol., 35, 105—109, 1986.
- E. Adams, I.R. Flockhart, C.E. Smithen, I.J. Stratford, P. Wardman and
M.E. Watts, Electron affinic sensitization VII. A correlation between structures, one electron reduction potentials and efficiencies of nitroimidazoles as hypoxic cell radiosensitizers, Radiat. Res., 67, 9—20, 1976.
- Overgaard, Clinical evaluation of nitroimidazoles as modifiers of hypoxia in solid tumours, Oncol. Res., 6, 509-518, 1994.
- Kagiya, S. Nishimoto, Y. Shibamoto, L. Zhou, J. Wang, Y. L. He, K. Sasai,
- Takahashi and M. Abe, Importance of tumour affinity of nitroazoles in hypoxic radiosensitization, Int. J. Radiat. Oncol. Biol. Phys., 16, 1033—1037.
- E. Adams and I.J. Statford, Hypoxia mediated nitroheterocyclic drugs in the radio- and chemotherapy of cancer: an overview, Biochem. Pharmacol., 35, 71 —76, 1986.
- Imamura, Radiosensitization with a 3-nitrotriazole (AK—2123), Int. J. Oncol., 6, 841-845, 1955.
- Kagiya, Researcher’s Booklet of the fundamental and clinical studies on sanazole (A K-2123), Health Research Foundation, Kyoto, Japan, 1998.
- Zhou, and S. Nishimoto, Electron affinic radiosensitizers possessing NPSH- reactive side chains: cytotoxicity and radiosensitizing activity towads hypoxic EMT6 cells in vitro, Int. J. Radiat. Biol., 67, 335—346, 1995.
- Godden and N. Howells, Induction of tumour hypoxia post-irradiation: a method for increasing the increasing the sensitizing efficiency of misonidazole and RSU 1069 in vitro, Int. J. Radiat. Biol., 55, 411—422, 1989.
21 S.R Venkatachalam and C.K.K. Nair, Synthetic strategies for designing drugs and ligands interacting with D.N.A. Proc. Ind. Natl. Sci. Acad., B62, 389— 426, 1996.
- M.S. Cowan, R. Panicucci, R.A. McClelland and A.M. Rauth, Targeting radiosensitizers to DNA by attachment of an intercalating group: nitroimidazole- linked phenanthridines, Radiat. Res., 127, 81—89, 1991.
- A. Denny, P.B. Roberts, R.F. Anderson, J.M. Brown and W.R. Wilson, NLA- 1 : a 2-nitroimidazole radiosensitizer targeted to DNA by intercalation, Int. Radiat. Oncol. Biol. Phys., 22, 553—556, 1992.
- Mathew, S. Kapoor, C.K.K. Nair, M.S. Sastry, N.G. Huilgol, C. Gopinathan,
B.B. Singh and V.T. Kagiya, Pulse and gamma radiolytic studies of the radiosensitizer sanazole (AK-2123) in presence of uracil, Radiat. Phys. Chem., 49, 2—4, 1997.
- T. Kunjappu, S.R. and C.K.K. Nair, Interactions of molecules
7. AK-2123 : An overview
N.G Huilgol
Division ot Radiation Oncology, Nanavati Hospital and Nledical Research Centre, Mumbai 400 056, India
Introduction
Mottaram et al. demonstrated insensitivity of anaerobic cells as early as 1930 [1]. Ever since then a large body of in vitro and in vivo studies has demonstrated that hypoxia compromises the effect of radiation on tumors. Indirect evidence for the existence of hypoxia in human tumors was reported over 40 years ago by Thomlinson and Gray [4]. Cells, which have low levels of oxygen, have much greater resistance to sparsely ionizing radiation. The presence of hypoxic cells in human tumors is one of the principle reasons for the failure of radiation therapy. Intensive laboratory and clinical efforts to overcome tumor hypoxia have focused on oxygenating, radio sensitizing or killing the maximally radioresistant fraction of tumor cells [2, 3]. This is termed as the “hypoxic fraction”. Hypoxic cells in human and animal tumors are though to arise primarily through two distinct mechanisms. The first is termed as chronic, of diffusion- limited hypoxia. The second mechanism of hypoxia in tumor’s is through transient, intermittent changes in blood flow. This is termed as acute or perfusion-limited hypoxia [4, 5]. Hypoxia within solid tumors is of clinical significance; i.e. hypoxic tumor cells. are more resistant to radiation and to some forms of chemotherapy, than well-oxygenated cells. Killing of hypoxic tumor cells by low LET or radiation requires 2 to 3 times the radiation dose necessary under oxygenated conditions [6, 7, 8].
Historically, the use of hypoxic cell sensitizers has not resulted in quantum jumps in survival. Yet, it remains that hypoxic cells with their inherent lack of sensitivity to radiation reduce the chances of cure. Head and neck cancers are reported to harbor significant amount to hypoxic cells, large enough to deter cure due to radiation [6,9]. An ideal model to assess any sensitizer should have more than 40 percent as hypoxic cells and tumor should not be very aggressive to overwhelm to the benefits of the treatment. Not surprisingly, many molecules with the potential for sensitizing hypoxic cells have been clinically tested. The halogens, niridazoles and many other bio reductive drugs have been tested without any sensational success. The reasons for failure of sensitizers are discussed elsewhere in this book.
The earlier sensitizers failed due to high toxicity or inappropriate administration of the drug with radiation. Besides, hypoxia confers on added virulence to the neoplastic cells by inducing mutations of p53.
Table 1. Newer molecules screened for sensitization potential
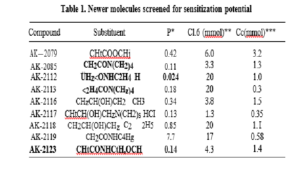
Inevitably, conventional hypoxic cell sensitizers alone may not be sufficient to address the problem of hypoxia. The search for more effective hypoxic cells sensitizers has led to the screening of many newer molecules, which are listed in Table 1. AK-2123, a nitro-triazole was inducted into a clinical ttial following in vitro and vivo studies regarding safety. Prof. V.T. Kagiya is the inventor of the molecule (Fig. 1).
Fig. 1. Molecular Structure of AK-2123
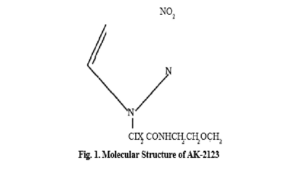
Pre-Clinical Studies and Rationale
Pi e-clinical data of AK-2123 has proved its safety and efficacy in vitro. Kagiya has shown its cytotoxic potential fo1’ KB tumor cells when these ceI1s were incubated in the presence of AK-2123. It however did not show any cytotoxic activity against proliferating Flow 7000 (normal) cells. Cytotoxic potential of the drug in hypoxic environment is significantly higher than in aerobic conditions. Jmamura has shown a relative toxicity of 6.6 for AK-2123 in hypoxic V79—37A cells [11].
AK-2123 acts best under hypoxic condition. It is not only a good hypoxic cell sensitizer of radiation but has shown to be a chemo-sensitizer for example of Adriamycin and vincristine [12].
Sensitization potential of AK 2123 compared to MISO is slightly less. Dam [13] in his studies with Chinese hamster ovarian CHO cells has shown a dose enhancement ratio of 1-2 for AK-2123 while 1-5 for hJISO. AK-2123 was chosen for further studies from the molecules list in Table 1, for reasons of fewer’ toxicities and lower cost. Preliminary laboratory studies demonstrated an acceptable toxicity profile. Annes index showed a how mutagenicity and was less than that reported for MISO (’Table 2).
Incubation of AK-2123 with V79- 37A(A) cell showed a relative toxicity of 6.6 in hypoxic conditions. A similar study of drug in CHO cells showed that a pretreatment of hypoxic cells with AK-2123 and subjected to radiation showed an enhancement by a factor of 2. Post-incubation effect of AK-2123 was more than MISO, in a study reported by Imamura (1995) in V79-379A cell line [13]. In vivo study ot the drug in JBII male mice of 8—10 weeks showed a LD50 of 2720 mg/kg. Single oral administration of 1000 mg/kg of Ak-2123 to ICR male mice (5 weeks) and Wistar male rats (7 weeks) did not show any ethologic changes.
A daily administration of AK-2123 at 500 mg/kg consecutively for 14 days to ICR male mice did not show any sub-acute toxicity. The ‘Rotard’ performance test with 14 days of oral administration to ICR-male mice showed an average score of 3.8 and 2.5 mts. at 200 mg/kg and 300 mg/kg of AK-2123 while it was 2.d and 1.7 in MISO. Neurotoxicity of AK-2123 observed in this study with male mice is considerably less than MISO AK-2123 also penetrates the obtain less efficiently than most other imidazole due to low octanol/water co-efficient at 0.14 to 0.16.
Animal studies have shown hypoxic cell sensitization potential of AK-2123 as well as cytotoxic and immunomodulating effects. However, it is the potential to sensitize hypoxic cells, which is the dominant effect that is undergoing a clinical evaluation in different institutions of vivo studies have showed the feasibility of administering the drug orally and parentally. Initial toxicological studies in ICR male mice when administered with AK-2123 at a dose of 500 mgs/kg and 1 mg/kg did not reveal ethologic, hematologic, toxicities (Kagiya 1985 Unpublished data). Kagiya reported on enhancement ratio of =1.71 at the administered dose of 430 mgs/kg in EMT6 bearing mouse. Tumor dose of 30 Gy single session was delivered following 40 minutes of AK-2123 administered intraperitoneally in this experiment. We reported a phase I study in Sensitizer’ News Letter in 1991 to establish the feasibility of administering the drug safely. A dose range of 200 mg to 600 mg per meter square was evaluated and safety of administering 600 mg/m2 of the drug was demonstrated in this study. A dose limiting neurotoxicity was not evident in this study
Clinical Trials
The drug has been evaluated for various sites in different centers. But, there are very few randomized trials. Some of the clinical experience is as follows.
Garcia, in 1989 reported the use of AK-2123. Clinical benefits were shown in his initial reports. The drug was well tolerated [15]. AK-2123 has been administered orally, intravenously, intra-tumoral and intra- arterial [16, 17, 18, 19, 20]. It’s well absorbed following oral, ingestion though; nausea and vomiting are the acute toxicities limiting per oral use. Intratumoural injection as suggested by Balmakhanov has technical difficulties and cannot be used in all anatomical sites [18]. Intra-arterial administration of Platinol, Peplomycin and Doxorubicin as a neo-adjuvant to surgery and radiation in the treatment of cancer of cervix has been reported by Fujiwaki-lida et al. The five and ten year survival is indeed impressive [20].
Similar, intra-arterial administration of AK-2123 in advanced cancer of cervix with foam embolization showed that the drug quickly gets into the plasma, i.e. central compartment. Our study showed a high plasma level of AK-2123, following intra-arterial administration [19] thus precluding administration of a substantially higher dose of AK-2123. Advanced cancer of head and neck as well as cancer of cervix are good models to assess any sensitizer as they harbor significant hypoxic core. Clinical studies on AK-2123 have shown an initial promise [15, 16, 17, 18, 19, 20].
Xu have demonstrated an excellent initial response in esophageal cancer [21]. Seven patients were radiated to a total dose – 60 Gy in 6 to 7 weeks. AK-2123 was administered 2—3 times a week at a dose = 900 mgs to 1000 mgs/m°. Total dose not exceeding 13 g was given in six weeks. Five of 7 patient (71.4(o) showed a complete response. However, 17.89o showed temporary paranesthesia in the extremities. Kose has reported a response rate of 62.5 percent following RT + AK-2123. In his study patients with colonic cancer‘ were iii adiated prior to surgery. Patients received 32 Gy in 8 fractions. Radiation was delivered on alternate days. AK-2123 was given 2 hours before radiation. A combination of clinical, endoscopic and imaging showed a complete response of 62.5 percent in those who ieceived AK-2123 as compared to 3.00 per cent in the control [22]. However, Berdo has not shown any complete response in his studies of advanced rectal cancer with radiation and AK-2123. Berdov reported lethargy and loss of appetite as the immediate side effects [23]. The dose limiting neuropathies to the extent of 13% have been reported by Senapati et al. [24]. The incidence of neuropathy reported by these authors varies from 7 to 139r. This has not been the universal experience, other investigators China, Russia and Pakistan, have not reported any patients with neuropathy.
For hypoxic cell sensitizer to be effective it must be administered when hypoxia is a significant deterrent for radio curability. Besides, drug should be present in adequate concentration during radiation. Administration of any hypoxic cell sensitizer with a short plasma half-life like AK-2123 allows frequent dosing. Accelerated fractionation of radiation delivery ensures minimum reoxygenation over time besides, repopulation. This clinical tourniqiiet sets an appropriate background for both testing and using the sensitizer.
Huilgol et al. have demonstrated effectiveness of AK-2123 in respectable head and neck tumor treated with AK-2123 and accelerated radiation. Patients with advanced head and cell canceres, which were potentially respectable, were randomized to the study and, the control group. Patients were radiated thrice a day at an interval of 04 hours, with no radiation treatment administered on weekends. A total of 54 Gy was delivered in 13 days. Patients received AK-2123 at 600 mg/m2 between the 1st and 2nd fraction. The drug was administered on all days of the treatment. Patients treated with AK-2123 had a complete response in all the patients where as only 44.44% (4 of 9) had CR in those treated with radiation alone. This study has shown the effectiveness of AK-2123 when used appropriately [25].
AK-2123 is a new molecule to reach the clinic toe evaluation as hypoxic cell sensitizer. The dose limiting toxicity of neuropathy was not seen in any of the patients treated for head and neck cancer while, patients with neuropathy were seen in patients with cancer of cervix.
The interim result that was announced by the multinational group under IAEA, investigating AK-2123 with radiation in cancer of cervix has been encouraging (personal communication). The interim report yet to be published has shown a promising initial response and two year survival. The long term benefit awaits a final analysis.
Conclusion
The quest for an ideal hypoxic cell sensitizer may not end with AK-2123, as neuropathy though seen in a small percentage of patients is a serious dose limiting toxicity. The drug however has proved its efficacy in improving initial response in both cancer of cervix and oro-phyrangea1 cancers. There are many small studies of patients with cancer of oesophagus and lung where addition of AK-2123 has demonstrated increased response. The mechanism of AK-2123 is similar to imadazoles that it is an electron donor. It has an interesting property of being an immunostimulant at low doses. AK-2123, in conjunction with continuous accelerated radiation has shown the best results so far. The drug when delivered daily over a short period of time in the range of 500—600 mgs, is both effective and safe. The incidence of neuropathy in this dose range would probably be around 3 percent, which is clinically acceptable.
References
1. Mottaram J.C. A factor of importance in the radiosensitivity of tumors. Dr J.C. Radiology 9: 606—614, 1936).
2 C. Khalil, A.A. Nordsmark, M. Herdsman The relationship between carbon monoxide breathing, tumour, oxygenation and, local tumour control in the C3H mammary carcinoma in vivo. Br. J. Cancer 69, 50—57 1994.
- Overgaard Sensitization of hypoxic tumor cells clinical experience. Int. J. Radiat. Biol. 56: 801—811, 1909.
- Thomlinson R. and Gray L. H., The histological structure of some human lung cancers and the possible implications for radiotherapy. Br. J. Cancer 9, 539—549, 1955.
- Thomlison H. and Craddock E.A., The gross response of an experimental tumor to single doses of X-rays. Br. J. Cancer 21, 108-123; 1967.
- Gray L.H., Conger A.D., Ebert M., Hornsey S. and Scott O.C.A., The concentration of oxygen dissolved in tissues at the time of irradiation as a factor in Br. J. Radiol. 26, 638—548; 1953.
- Brown J.M., Evidence foe acutely hypoxic cells in mouse fumoirs, and a possible mechanism for Br. J. Radiol. 52, 650—656, 1979.
- Sutherlan M. and Franko A.J., On the nature of the radio biologically hypoxic fraction in human tumors. Int. J. Radiat. Oncol. Biol. Phys. 6, 117—120, 1980.
Elkind M.M., Swain R.W., Alwscio T., Sutton H., Moses W.B., Oxygen, nitrogen, recovery and radiation therapy. In: Cellular radiation biology. Baltimore: Williams and Wilkins: 442—461,
- Hall E.J. Radiobiology for the radiologist, 3rd ed. Philadelphia: Lippincott; Chapter 7 pp 137—160;
- Imamura Radiosensitization with 3 nitrotriazole (AK 2123). Int. J. Oncology. 6:841—845, 1995.
- Chang X., Chemosensitization of AK-2123 for ADS, ACR, in vitro and for cyclophosphamide. Abstract of the 10th International conference on chemical
- Dam A.M. Cytotoxic and radiosensitizing effects of AK 2123 and MISO in vitro comparative study on CHO cells-sensitization Newsletter. 3(3) 3-5,
- Huilgol N.G., Ghooi R.B. and Mehta A.R. Preliminary report on the clinical study of AK-2123, a hypoxic cell Radiation Sensitizer News Letter, March 1991.
- Garcia , Peru, Radiosensitization Newsletter Vol. 8 No. 3; pp2; July 1989.
- Gai‘cia , Peru, Tolerable oral dose of AK-2123 Radiosensitization Newsletter Vol. 10, No. l ; Jan. 1991.
- Huilgol N.G., Chatterjee N., Singh B.B. and Mehta A.R AK-2123 a hy poxic cell potentiator of radiation—A report on the use of AK-2123 in head and neck cancer. Journal of Clinical Radiotherapy Oncology 10(1): 9—10;
- Balmukhanov S.B., Seitkazina G.D., Koposova R.P., and Eselbaieva G.O. Radiosensitizing effect of AK-2123 on experimental tumours Radiosensitization Newsletter 9, No. 3; pp 12—21 ; July 1990.
- Huilgol G., Soni A., Chatterjee N., Nair C.K.K., Mathew R. Case studies of intra-artierial administration of AK-2123 with embolisation in re-radiation of cancer of cervix.
- Fujiwaki , lida V., Ohnishi W., Watanbe Y., Ryuiko K., Takahashi K., Miyazaki K., Intra-artierial neoadjuvant chemotherapy foltowed by radical surgery and radiotherapy for stage llb cervical carcinoma. Anti Cancer Research 17(5B): 357 I —3755; 1997 Sep.—Oct.
- X.Y., Abstract the 9th International Conference on chemical modifiers of cancer treatment 367—368. 22—26 Aug 1995 Christ Church, Oxford U.K.
- Kosse V , The combined colorectal cancer treatment using AK-2123 sensitizer, Radiosensitization Newsletter, 12(1), 1993.
- Berdor B.A. Prospective clinical trial of the AK-2123 as chemical sensitizer for locally advanced rectal cancer. Sensitization news letter 2, 4—7, 1995.
- Senapati S.N., Samanta D.R., Giri S.K., Mohanti B.K., Nayak C.R., and Kagiya V.T. AK-2123 as a radiosensitizer in Stage JIIB carcinoma cervix Sensitization Newsletter Vol. 4, No. 3; July 1997.
- Huilgol N.G., Chatterjee N., and Mehta A.R. An overview of the initial experience with AK-2123 as a hypoxic cell sensitizer with radiation in the treatment of advanced head and neck cancers. Int. Journal of Radiation Oncology Biology Physics 1996; Vol. 32, No. 5: 1121—1124.
Cancer of Cervix
N.G. Huilgol’, Abhijit Soni2, C.K.K. Nair*, Mathew* and Neela A. Chatterjee’
Division of Radiation Oncology, Department of Radiology2, Nanavati Hospital and Madical Research Centre, Mumbai 400 056, India
Department of Biochemistry, Bhabha Atomic Research Centre, Mumbai 400 085, India
Introduction
Radiation for locally advanced lesions has not yielded satisfactory results; 40 to 50% of stage II cancer of cervix recur [1]. A good deal of these patients presents in an advanced state for various social and financial reasons. Inevitably, a large proportion of patients fail radical radiation or radiation with any adjuvant treatment. Persistent disease following radical treatment is treated symptomatically in our institution. However, frequent pelvic hemorrhage with episodes of torrential bleeding can cause considerable morbidity. Hemorrhage in such patients is potentially life threatening. Vascular embolization or ligation of both uterine arteries has been suggested [2]. Vascular ligation or occlusion leads to decreased perfusion of tumors. This procedure primarily performed to control hemorrhage is also an optimal clinical setting to test hypoxic cell sensitizers. Traditionally, hypoxic cell sensitizers in the laboratory are tested by totally occluding the blood supply or nitrogen breathing animals. Embolization, or arterial ligation simulates a laboratory scenario of inducing total hypoxia as in the clinic. Hence, it was proposed to test the feasibility of administering a hypoxic cell sensitizer intra-arterially along with embolization, a modality by which a high concentration of the drug is expected in the tumor.
AK-2123 is a nitro-triazole. It has an OER of 1.8 and octanol to water co-efficient of 0.14-0.17. Animal experiments and phase I studies have shown efficacy and safety of the drug. Our preliminary experience of the drug in head and neck cancer have shown promising results [3]. The molecule is undergoing a phase III trial under the aegis of International Atomic Energy Agency (IAEA), Vienna, Austria. The interim report of the study is very encouraging. The initial response of patients with cancer of cervix treated with AK-2123 showed a significantly better initial response (IAEA-personal communication of the interim meeting). We have reported similar effectiveness of AK-2123 in head and neck cancer [3]. Hence it was tested in an intra-arterial mode. The following study was designed to test feasibility of intra-arterial administration of AK-2123, Pharmacokinetics of AK-2123 following such an administration and to document acute toxicities related to the drug.
Material and Method
Patients with intractable vaginal bleeding due to advanced recurrent cancer were included for the study. Patients had either a frozen pelvis or a large cervical disease with total parametrial involvement. All these patients had radical radiation in the past. Patients had received 50 Gy in 5 weeks to the entire pelvis followed by 20 Gy to point A in single insertion of intracavitory radiation. None had chemotherapy following resonance. Intractable bleeding was defined as failure to control bleeding with vaginal packing for 10—15 days, with a continuous fall in haemoglobin, or recurrent bouts of bleeding, leading to anemia (Hb < 6 g/dl).
Eight patients were recruited for the study following an informed consent. Patient were informed that the primary procedure is embolization and injection of AK-2123 intra-arterially was investigational. All patients except one received radiation following embolization and intra-arterial injection of AK-2123. Homeostatic pelvic radiation was delivered in spite of prior radiation. The choice of the dose oiAK-2123 was empirical but guided by our initial experience with intravenous administration. A dose of 1 g/m2, administered intravenously was tolerated very well in our clinic. Hence a dose of 4 g of AK-2123 was administered. The dose was divided into two and each of the uterine artery was cannulated to deliver 2 g of the drug. In patient no. 4 cannulation was possible from only one side. All the drug was administered from one side only. A mid pelvic dose of 800 cGy to 1200 cGy in four to eight fractions was delivered in 4 to 10 fractions. Re- radiation though not common was resorted to ensure a sustained haemostasias. Initial response of tumor was assessed at the end of radiation. Vital parameters and renal profile were monitored. Gastro-intestinal side effects like nausea and motility disorders were monitored but were deemed unlikely due to AK-2123. Tumour response was assessed immediately after radiation as well as after three weeks (Table 1). The maximum response was recorded and considered for the study though it is not the objective of this analysis to study the effectiveness of AK-2123.
Intra-Arterial Embolisation
Procedure is done under local anesthesia. Selective cannulation of the right uterine is achieved via the left femoral approach using a 5 French renal double curve catheter. For the left uterine cannulation right femoral approach was used. A co-axial catheter system (3 to 2 Fr. Tracker (Balt)) was occasionally used to achieve selective cannulation when vessel caliber was small. After confirming catheter position and demonstrating abnormal tumor vascularity, 100 ml of 2% AK-2123 (aqueous) was slowly infused over a period of 20—30 minutes. This was followed by embolization of the uterine artery using particulate gel foam. The uterine artery embolized up to half the length of its main stem. Pre- and post-embolisation check angiogram was done to confirm complete embolization (Figs. 1 and 2). Patients received 2 g of the drug from each side except in one patient.
Results
All patients had cessation of bleeding immediately atter the procedure. Patient no. 8 refused radiation and was discharged after 2 days of the procedure without any active bleeding.
The first patient died a week after the completion of radiation. The patient had a frozen pelvis which did not respond to radiation. She died of
An Early Experience with AK-2123 Administered Intra-Arterially
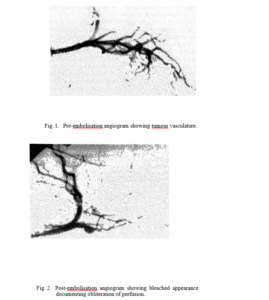
acute renal failure. Renal scan in this patient showed poor renal function with post renal obstruction. Hence, the renal failure was considered to be of obstructive etiology than related to the drug. Injection of Conray could also have precipitated acute renal failure.
One patient had a complete response following 20 Gy of pelvic radiation and 4 g. Of the drug administered through both urine arteries. This patient who was chemotherapy naive had a very large exophytic central disease occupying upper third vagina and unilateral parametrial disease reaching the pelvic wall. Thus one patient had a complete response, two none at all and four had partial response. In one patient radiation was not delivered. Patients who showed partial response had a significant regression of the central than the parametrial disease. Two of the eight patients developed peripheral neuropathy. Patients complained of tingling and numbness in both the hands and feet. No clinically evident sensory motor deficits were elicited. These symptoms receded over 2-3 weeks.
Pharmacokinetics
The pharmacokinetic data of AK-2123 following intra-artery a1 administration is shown in Table 2. AUC(0- ) at 303-720 is nearly similar to i.v. administration (study conducted in our lab). Cmax in plasma was achieved within 1.33 hours. Plasma clearance half life was (T1/2e1)
5.12 per hr. The drug was eliminated unchanged in urine. The above data suggests a quick entry of the drug in plasma with an AUC almost similar to intravenous administration. It suggests a lower initial retention of the drug in the tumor mass.
Table 2. Pharmacokinetics
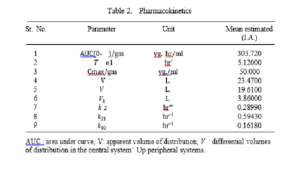
AUC : area under curve, V: apparent volume of distribution; V : differential volumes of distribution in the central system’ Up peripheral systems.
An Early Experience with AK-2123 Administered Intra-Arterially
Discussion
Intra-arterial route of administration of cytotoxic drugs have been described by various authors in the treatment of liver, mead and neck cancers and breast cancer [4, 5, dJ.
Intra-arterial administration of the generally confers higher localization of the drug. Thus reducing systemic toxicity. Intra-arterial administration of the drug in this study was well tolerated though 2 of the 8 patients developed transient neuropathy. One patient who died of renal failure probably did so due to the progressive disease causing obstructive uropathy and is presumed to be unrelated to the procedure. Hemorrhage stopped in all patient even in the patient who did not undergo radiation. One of the eight patients showed a remarkable and unexpected complete response indicating the effectiveness of the strategy.
Any hypoxic cell sensitizer to be effective must be present in a high enough concentration in an adequately hypoxic environment. It was assumed that intra-arterial administration of AK-2123 would increase tumor concentration of the drug while, embolization would induce hypoxia. But, the pharmacokinetic studies showed a ,pattern of venous equilibration, plasma halt-life and renal excretion, which was similar to intravenous administration.
Intra-aiterial administration of the drug perhaps ensures a higher cellular concentration but follows a kinetics that is not any different than parental administration. Systemic dose limiting toxicity of neuropathy was seen at a dose of 4 g. Administration of AK-2123 at an escalated dose even per arterial is fraught with danger as the AUC, between the two routes is not significant.
A better approach would be to inject microspheres embedded with AK- 2123 in the intrauterine artery that can ensure embolization and sustained release of drug with hyper fractionated radiation. Such a technique will ensure higher dose of the tumor without mimicking intravenous administration. Administration of AK-2123 intra-arterially is feasible and safe. Yet, the drug is quickly transferred to central compartment. The avowed benefit of delivering higher dose of the drug, to a localized tumor, thus ensuring a better therapeutic ratio seems unlikely in this case.
References
- S. Clifford Chao, J.F. Williamson, P.W. Grigsby and C.A. Perz, Uterosacral space involvement in locally advanced carcinoma of the uterine cervix. Int. J. Radiat. Oncol. Biol. Phys. Vol. 40, No. 2; 397—403 1998.
- D. Hatch, Cervical cancer in Ref: Practical Gynecologic Oncology Ed. Jonathans Berck, Weville F. Hacker. Publishers Williams & Wilkins 198
- Huilgol G., Chatterjee, N. A., and Mehta A.R. An overview of the initial experience with AK-2123 as a hypoxic cell sensitizer with radiation in the treatment of advanced head and cancers. International JGurnal of Radiation Oncology Biology Physics 1121—1123; Vol. 34, No. 5: 1996.
- Cakmakli, S., Ersoz, S., Tug, T., Karaayvaz, M. and Akgul, H. Intra-arterial infusion chemotherapy in the treatment of locally advanced breast cancer. Acta Oncologica 489—491: 36 No. 5; 1997.
- Stephens, O. Intra-arterial induction chemotherapy in locally advanced stage III breast cancer. Cancer 645—650: 66: 1990.
- Current Trends in Cancer Treatment. Editors Nagraj G. Huilgol and A.R. Mehta, Nachiket Academic Publications
9.An Overview of Radiosensitizers
G.K. Rath, P.K. Julka and D.K. Parida
Department of Radiation Oncology, All India Institute of Medical Sciences, New Delhi, India
There is a need in clinical practice to enhance the differential effect of the radiation in tumor and moral tissues. Radiosensitizers are the compounds which when combined with radiation enhances greater tumor inactivation than would have been expected from the additive effects of each modality. The response of radiation to various tumors are still not very clear. Tumor heterogeneity, necrosis and lack of oxygen may account for the varied response. Therefore it is of prime importance to look for certain agents that modify the response of cells to radiation in a well-controlled fashion. When carefully used these agents sensitize the tumor cells to radiation and at the same time protect normal tissues, thereby increasing the therapeutic ratio. Some of the sensitizers have already established their places in the clinical practice and rest are in the laboratory, or are under going different trials. The use of radiation sensitizers in clinical practice started in late 1930s, when Gray [1] first quantified the effects of oxygen on radiation response. In fact radiation acts more effectively in presence of oxygen. Experimental tumors are found to contain hypoxic cells and have dramatic effect when treated with radiation after oxygenation. 02 electrodes both inside the tumor as well as normal cells directly measure the oxygen tension. As the normal cells are already oxygenated, adding further oxygen will sensitize only tumor cells. Treating the patients with hyperbaric oxygen was not much in clinical use for its associated grade
IV complications like necrosis and gangrene. It is clear that some combinations have made a profound difference in the outcome of of treatment in several tumors.
Radiation kills cell through a very complex chain of events. It is believed that the damage, which is most significant for mutation and loss of cell viability, is the damage in DNA. Various dose schedules are tried to obtain the optimum result. Certain agents are used to sensitize the cells to radiation and thus called radiosensitizers. These may be either chemical, pharmacological or physical agents, which increase the lethal effects of radiation when administered in conjunction with it. There are many agents, which enhance the cell sensitivity to radiation, but it will be of clinical importance, that they only sensitize the tumor cells and not normal cells. The basic aim of using radiosensitizers for having a greater tumor control with less radiation dose ending up in minimal complications. The two principal types of radiosensitizers those are in clinical practice since long are halogenated pyrimidines and hypoxic cell sensitizers. Faster cell cycle in tumor incorporates more pyrimidines than surrounding normal cell. There is a long list of agents used as radiosensitizers and some of the agents commonly used in clinical practice and the agents who have a promising role in future are described as follows.
Electronafinic Compounds
Over the last decade new compounds belonging to this group have been tried in clinical practice to improve the radiosensitivity of hypoxic tumor cells, which are generally resistant to low LET radiation. The most extensively used compounds are misonidazole, metronidazole, nimorazole and nitrofurantoin. The 2-niti otriazole is the most potent sensitizer due to its higher electron affinity than other compounds of its class. It has been established that presence of 10 mM of misonidazole in the hypoxic tumor cell increase the radio sensitizing effect compared to normal aerated cell. Because of its greater effectiveness at lower concentration misonidazole is the leading compound as radiosensitizer [2]. Misonidazole has been known to produce following effects; radio sensitization of hypoxic tumor cells, direct killing of hypoxic tumor cells, radio sensitization of certain normal tissues, neurotoxicity, mutagenicity to bacteria as well as mammalian cells under toxic and hypoxic conditions and oncogenic transformation in vitro and in vivo. The sensitizer enhancement ratio (SER) for mammalian hypoxic cells in vitro varies from 1.2 to 2.4. The cell killing potential of the drug is greatly enhanced when the temperate is raised by few degrees. The largest trial in head and neck cancers performed in Denmark using misonidazole as radiosensitizer clearly shows the survival advantage. But the neuropathy associated with this compound limited its use. Another positive result has been published by the Danish Head & Neck Cancer Group (DAHANCA) in a clinical trial of the 5-nitroimidazole,
i.e. nimarazole used with radiotherapy for the treatment of head and neck region [3]. With a high dose of nimorazole (1.2 g/m2)along with radiotherapy the side effects observed are within acceptable limits. Nimorazole was administered an hour before radiation and the patients were treated six days a week. Subsequently another compound named etanidazole having a shorter half-life in vivo was tried with better result. The compounds, which contain the nitro-heterocyclics can show oxygen mimetic property and act as i adiation sensitizers. Simple nitroheterocycles include 5-nitro- imidazoles such as metronidazole, nimorazole and analogues the 2- nitroimidazole, i.e. misonidazole act as radiation sensitizers owing to the presence of alkylating functions in their structures. These agents must be present in the cell at the time of irradiation for enhancing the radiation effect. Ultimately these agents increase the radiation induced DNA damage, thus not allowing the repair process to be complete. Numerous studies have established that the efficiency of radiation sensitization increases with the electron affinity properties of the compounds. The intermolecular electron migration within intracellular DNA, followed by electron trapping by the sensitive is a process in competition with recombination whereby the overall number of free radicals is increased in the presence of the sensitizer. This hypoxic cytotoxicity property was first observed in multicellular spheroids treated with metronidazole [4]. The efficiency directly related to the electron affinities of there compounds. Metronidazole was the first nitro heterocyclic compound to be investigated clinically as a hypoxic-cell radiosensitizer. Urtasun ct a1. reported a prolonged disease free survival in glioblastoma multiformat patients treated with metronidazole as radiation sensitizer. Both metronidazole and misonidazole could not be used in clinically frequently for their neurological toxicity. The lipophilic properties of the misonidazole analogue account for its toxicity and laboratory experiment produced a less lipophilic drug called etanidazole of which phase I and II studies have already established less neurological complications. Several other clinical trials are in progress using other novel sensitizers including substituted nitroheterocyclic compounds.
2-Deosy-Glucose
This compound is an inhibitor of glycolysis and transport of glucose. It is known for its sensitization of tumor cells of radiation therapy. Radiation cells kill occurs for various types of DNA damage including double strand breaks. These damages can be repaired through different pathways. Studies have shown that the cellular process leading to the repair and fixation of radiation damage require continuous flow of the metabolic energy supplied by glycolytic pathways and therefore the agents those inhibit the glucose transport and glycolysis differentially inhibit the repair processes leading to an enhancement of the radiation damage [5]. 2-DG had successfully tried in phase I and II trials at our center in case of human cerebral gliomas [6].
Hematoporphyrin Derivatives (HpD)
Since these compounds inhibit certain enzymes of the respiratory metabolism, resulting in an increase in the glucose usage and glycolysis, it enhances the energy-linked radio sensitizing effects of 2-DG in cancer cells. The hematoporphyrin derivatives PS-3 potentiates the radio sensitizing effect of 2-DG in cancer cells, possibly by further reducing the energy supply leading to an irreversible inhibition of DNA repair and increases cytogenetic damage and cell death [7]. It is a nitrotriazole compound with a potential for hypoxic cell sensitization As the tumor consists of hypoxic cells this compound act as a sensitizer to radiation. The mechanism of action ofAK-2123 is like other nitroimidazole compounds. Even the clinical trials shows less neurotoxicity compared to other hypoxic sensitizers, nausea and gastric discomfort are the significant dose limiting toxicities [8, 9]. Despite many limiting complications and not so encouraging results, research is still on to find out a satisfactory hypoxic cell sensitizer.
Heat
Treatment of tumors with heat was known long time back. Towards the last decade of nineteenth century William B Coley [10], a Surgeon in United States developed mixed bacterial toxin to produce pyrexia to treat cancer and subsequently known as the father of modern hyperthermia. As heat enhances blood flow used to obtain a better penetrating effect of the external beam radiation Muller et al (1912) established the principle of thermal penetration of X-ray was better. Hyperthermia enhances the effect of radiation on cell when administered concurrently [11]. The exact mechanism though not known but the radiosensitivity depends on the temperature and heating time. The repair of potentially lethal damage can also be reduced by additional heat treatment leading to an increase in radiation cell kill. Heat interacts with radiation mainly through two different mechanism, i.e. hyperthermic radiosensitization and direct hyperthermic cytotoxicity. The hyperthermic radiosensitization is expressed as the thermal enhancement ratio (TER) and produces optimum result with simultaneous application. The therapeutic ratio does not alter when the temperature of the tumor is raised above the normal tissue. However technical feasibility and complex thermometry limit the clinical utility of hyperthermia as radiosensitizer.
Inhibitors of Anaerobic Glycolysis
This group includes 5-Thio-Glucose (5-TDG) and butyric acid. 5-TDG kills the hypoxic cells by inhibiting the uptake of glucose and stopping the anaerobic glycolysis. It sensitizes the hypoxic cells to radiation. It also potentiates the cell killing effects of hyperthermia on hypoxic cells in vitro. However, butyric acid inhibits anaerobic glycolysis by reducing lactic acid dehydrogenase activity leading to stoppage of anaerobic g1ycolysis. Sodium butyrate can be used specifically to sensitize neuroblastoma cells, as theses are more sensitive to inhibitors of anaerobic glycolysis than other cell type (12, 13J.
TX-1877
This compound along with its analogs were designed, synthesized and evaluated for their in vitro and in vivo radiosensitization, tumour growth control, suppression and immunopotentiation. In-vitro radiosensitizing ability of TX-1877 and its analogs with their partition co-efficient values of more than 0.050 were comparable to misonidazole at their doses of 1 mM. Tumour regrowth was suppressed evidently 20 days after the treatment in the irradiated group with TX-1877 and KIN-806 plus radiation [14].
Nitric Acid (NO)
The effects of nitric acid on the radiosensitivity of SCK tumor cells in oxic and hypoxic environments in vitro have been extensively studied. The NO donor compounds 2, 2-diethyl-1-nitroso-oxyhydrazine sodium salt (DEA/NO) and spermine/nitric acid complex (SPER/NO) release NO at half-lives of 2.1 min and 39 min respectively at pH 7.4. It has been seen that the aerobic cells were not radiosensitized by DEA/NO or SPER/NO. At higher concentrations SPER/NO was found to be cytotoxic in aerobic conditions but not in hypoxic conditions. DEA/NO was mildly toxic to the cells both in aerobic and anaerobic conditions. The NO released from nitrous oxide donors is as effective as oxygen to radiosensitize the hypoxic cell in vitro [15]. Its application to the radiosensitization of hypoxic cells in solid tumors is to be further investigated.
Significant antitumor activity has been reported when radiotherapy is administered with 13-cis-retinoic acid (cRA) and interferon-a 2a (IFN-n) in the treatment of advanced stage cer vi cal and skin cancers. Radiosensitization is minimal when ME-180 cells were treated with either cRA or IFN-n before radiation. There is substantial benefit by the use of cRA and IFN-o as a combination radiosensitizer in selected human carcinomas [1d].
Radiation sensitizers along with various altered fractionation schedule of radiotherapy have been used in an attempt to increase the biologic equivalent dose to the tumor burden and improve local control. It is of tremendous importance to know the cellular radiosensitivity as a pai’ameter determining the clinical response of the tumor to radiotherapy. The relation between lymphocyte and radiosensitivity have been assayed using a limiting dilution and soft agar clonogenic assay to obtain the surviving fraction at 2 Gy (SF2) and the result shows that patients having radiosensitive lymphocytes had significantly increased risk of developing late coplications. Increased radiosensitivity is always associated with an increased risk of morbidity [17]. However, further work must be continued to ti’anslate laboratory benefits of various radiosensitizers into clinical application. A better understanding of the tumor microenvironment and the effet of modulators such as carbogen, nicotinamide and glutathione depletors are interesting avenues under evaluation. The applicatioin of radiosensitizers will not be universal in all solid tumors but could be specifically diverted to tumor sites according to their characteristic cell kinetics and presence or absence of hypoxia. However, a few new tests could bring some optimism in a not-too far distant future.
References
- Gray A.J., Dische S., Adams G.E., Flockhart I.R., Foster J.L. Clinical testing of the radiosensitizer Ro-07-0582:I. Dose, tolerance, serum and tumour concentrations. Clin. Radiol. 1976; 27: 151—7.
- Ash D. , Peckham M.J., Steel G.G. The quantitative response of human tumours to radiation and misonidazole. Br. J. Cancer l979;40:883—89.
- Hansen H.S., Overgaard J. Jorgensen K.E. DAHANCA (Danish Head and Neack Cancer)—20 years of co-operation. Ugeskr Laeger 1958; 160 (6): 821—
- Adams G.E., Clarke E.D., Gray P., Jacobs R.S., Starford I.J. Wardman P., Watts E., Patrick J., Wallace R.G., Smithers C.E. Structure-activity relattonship in the development of hypoxic cell radiosensitizers: II. cytotoxicity and therapeutic ratio. Int. J. Radiat Biol. 1979; 35: 151—60.
- Jain K., Kalia V.K., Sharma R., Mahajan V., Menon M. Effects of 2-Deoxy- D-glucose on glycolysis, proliferation kinetics and radiation response of human cancer cells. Int. Radiat. Oncol. Biol. Phys. 1985; 11: 943—50.
- Mohanti K., Rath G. K., Anantha N., Kannan V., Das B.S., Jena A., Ravichandran R., Sahi U.P., Kumar R., Kapoor N., Kalia V.K., Dwarkanath B.S., Jain V. Improving cancer radiotherapy with 2-Deoxy-D-Glucose: phase 1/11 clinical trials on human cerebral glioma. Int. J. Radiat. Oncol. Biol. Phys. 1996; 35(1): 103—11.
- Dwarkanath S., Adhikari J.S., Jain V. Hematoporphyrin derivatives potentiate the radiosensitizing effects of 2-deoxy-D-glucose in cancer cells. Int. J. Radiat Oncol Biol. Phys. 1999; 43(5): 1125—33.
- lmamura Radiosensitization with a 3-nitrotriazole (AK-2123). Int. J. Oncol. 1995; 6: 841-45.
- Shibamotoy Y. Radiosensitization in vitro by 3-nitrotriazole. J. Radiat. Oncol. Biol. Phys. 1986; 12: 1063—66.
- Coley B. The treatment of malignant tumours by repeated inoculation of erysipelas-with a report of ten original cases. Am. J. Med. Sci. 1893; 105: 487—93.
- Hall E.J., Aster M., Geard C., Bigalow J. Cytotoxicity of Ro-07—0582: Enhancement by hyperthermia and protection by Br. J. Cancer 1977; 35: 809—15.
- Bushway A., Whistler R.L. Regression of cancer growth by 5-thio-D-glucose.
- Carbohydr. Nucleosides 1975; 2: 399—404.
- Sakamoto A., Prasad K.N. Effect of DL-glyceraldehyde on mouse neuroblastoma cell Cancer. Res. 1972; 32: 532—37
- Kasai , Nagasawa H., Kuwasaka H., Oshodani T., Nishioka A., Ogawa Y., Yoshida S., Inayama S., Inomata T., Hori H. TX—1877: Design, synthesis and biological activities as a BRM-functional hypoxic cell sensitizer. Int. J. Radiat. Oncol. Biol. Phys. 1998; 42(4): 799—802.
- Griffin R.J., Makepeace C.M., Hur W.J., Song C.W. Radiosensitization of hypoxic tumour cells in vitro by nitric Int. J. Radiat Oncol. Biol. Phys. 1996; 36(2): 377—83.
- Ryu , Kim. O.B., Kim S.H., He S.Q., Kim J.H. In vitro radiosensitization of human cervical carcinoma cells by combined use of 13-cis-retinoic acid and interferon-alpha2a. Int. J. Radiat. Oncol. Biol. Phys. 1988; 41(4): 869—73
- West M., Davidson S.E., Elyan S.A., Swindell R., Roberts S.A., Orton C.J., Coyle C.A., Valentine H., Wilks D.P., Hunter R.D., Hendry J.H. The intrinsic radiosensitivity of normal and tumour cells. Int. J. Radiat. Biol. 1958; 73(4): 409—13.
10. Radiosensitizer for hypoxic Tumor Imaging
S. Murugesanl, S.J. Shetty, O.P.D. Noronha,
A.M. Samuel’, T.S. Srivastava2 and C.K.K. Nair
Radiation Medicine Centre, Bhabha Atomic Research Centre Mumbai 400 085, India
Departmentof Chemistry, Indian Institute of Technology, Bombay Mumbai 400 076, India
RadiationBiology Division, Bhabha Atomic Research Centre Mumbai 400 085, India
Abstract
Technetium-S9m labeled cyclam N-2’ methoxyethyl 1-2-(3’-nitro-1’-triazole) acetamide (cyc1am-AK-2123) had been synthesized, radiolabe led and characterized as a hypoxic tumor imaging agent.. Radiochemical purity was greater than 95%. Marker biodistribution was measured in normal Wistar strain rats at different time intervals after i.v. administration. In vivo distribution and scintigraphic imaging studies were performed after i.v. injection into mammary tumor-bearing rats using a gamma camera and associated computer.’ Intratumor partial oxygen pressure ( and oxygen saturation measurements were performed to estimate the oxygenation status of the tumors. Tumor to muscle ratio (T/M) of ’9 ’Tc-cyclam-AK-2123 was 8.5 which was compared with other tumor seeking radiopharmaceuticals, viz. ”’Tc-(V) DMSA (3.07), ’9 Tc-citrate (5.29) and 20IT1C1 (3.29). T/M ratios were also evaluated in comparison with radioiodinated iodoazomycin galactopyronoside (1251-IAZG). The ratio obtained was 18 for ”’Tc-cyclam A K-2123 and 20 for ‘2’I-IAZG, respectively. The increased concentration of radioactivity in these tumors suggests that this agent could be labeling hypoxic cells and have utility as an imaging agent.
Introduction
It is known that tumor cells can be resistant to radiation and chemotherapeutic treatments owing to low oxygen tension and hypoxia (Tannock, 1972). In order to overcome this component of tumor resistance, a number of hypoxic-cell radiosensitizers have been developed. Some 2-nitroimidazole and nitrotriazole derivatives were found useful for clinical application since they combine low neurotoxicity with high radio sensitizing activity. These compounds entel’ cells by diffusion and can be enzymatically reduced intracellularly. In the absence of an adequate supply of oxygen, they can undergo further reduction to reactive species which can bind to cell components (Edwards, 1993). The enzymes responmsible for the bioreduction of nitro compounds within cells have not identified but xanthine oxidase (XOD) (de Jong, 1990) and P450 cytochrome C reductase are potential candidates (Joseph et al., 1994).
Detection of hypoxia within tumors by the imaging with radiolabeled markers was first proposed by Chapman (1979) who showed that misonidazole could selectively bind to hypoxic cells in mouse tumors (Chapman et al., 1981). Since then, “F-fluoromisonidazole was developed and evaluated for detection of hypoxia in tumors (Koh et a1., 1991; Rasey et al., 1996) and myocardium (Martin et a1., 1992) using positron emission tomography (PET), while radioiodinated [1231]-iodoazomycin arabinoside (IAZA) has been evaluated with single photon emission tomography (SPECT) (Mannan et a1.,’1991; Parliament et al., 1996). The usefulness of
9″Tc-labelled PnAO-2-nitroimidazole derivative (BMS-18132) for the detection of myocardial ischemia has been reported by Shi et al. 1995; DiRocco et al. 1993 and for tumor hypoxic by Ballinger et al., (1996). Chapman et al (1998) have recently reviewed the preclinical and clinical studies associated with these nuclear medicine procedures.
We herein report the synthesis, radiolabeling and preclinical testing of a novel hypoxic marker based upon the radiosensitizer AK-2123 which was found to concentrate in hypoxic cells and is currently undergoing Phase III clinical trials for the treatment of advanced head and neck cancers (Nagraj et a1., 1996). The octanol to water quotient of AK-2123 is ten times lower than misonidazole (MISO). Therefore it was expected to exhibit lower neurotoxicity when administered to cancer patients (Nagraj et a1., 1996). Our present study was undertaken to label this radiosensitizer with Tc-99m, a radionuclide which is cheap and readily available in all nuclear medicine clinics, and determine its potential for marking hypoxic regions within animal tumors. Since AK-2123 lacked appropriate structural moieties to complex with Tc-99m we converted it into a cyclam derivative. The present study reports the synthesis of cyclam AK-2123, its radiolabeling with Tc-99m and the evaluation of this agent in normal and hypoxic tumor-bearing animals.
Materials and Methods
Tc 4 used in this study was obtained by the solvent extraction method and further processed (NOronha, .1984) All other chemicals and reagents used for synthesis were of analytical grade and obtained from local sources. AK—2123 as a crystalline powder was obtained as a gift from Professor Kagiya, Kyoto Japan, Cyclam—AK-2123 was synthesized as described
below. The other agents that were used for purposes of comparison were
,2011101 ”!סף-כש DMSA and 99mTc-citrate. 2(“Tier was obtained from Amersham International Plc while the others were prepared in our in— house radiopharmacy. 99mTc-(V) DMSA was obtained by adding 99m TCOZ to Sn (11) DMSA at an alkaline pH (Ohta et al., 1984) and ”!פדה-»61₪ was prepared as per the method described by Ercan et al. .(1993)
AK-2123 (A) was refluxed for 5.0 h with an equimolar amount of ethyl chloroacetate in the presence of anhydrous K2C03 and dry acetonitrile as solvent (Scheme .1( The reaction products were purified on a neutral alumina column with methanol/toluene-(l : 9( as eluent to give the pure product, N-alkylated AK-2123 (B). TheN-alkylated AK 2123 thus obtained was subjected to alkaline hydrolysis, by stirring with aqueous NaOH in methanol for 5h in the dark to yield AK—2123 acid (C). TheAK—2123 acid was treated with thionylcloride to produce AK-2123 acid chloride which,
in excess, was reacted with cyclam to yield the AK-2123 cyclam complex
(D). This compound was purified on neutral alumina with methanol/toluene (1 : 9) as eluent, and its chemical structure was determined by UV-vis, IR,
NMR and FAB-mass spectroscopy.s
- Radiolabeling of Cyclam AK-2123 Complex
2.0 mg of cyclam AK-2123 was dissolved in 1.5 ml physiological saline taken in a 10.0 ml reaction vial. 200 y1 of Sn2 ion (200 kg) from a
solution containing 2.0 mg of Sn CI22H in 0.1 N dil HCI was then added followed by the addition of 0.5 ml of 99‘TcO4 (74 MBq). The reaction mixture was incubated at room temperature for 10 min. The pH
was adjusted to 7.5 with 0.01 M NaHCO3 solution and the reaction mixture was passed through a 0.22 km membrane filter to free the product from larger colloidal particulates. Radiochemical purity was determined by ascending paper chromatography using physiological saline as the developing solvent.
- Biodistribution Studies in Normal Rats
Wistar strain male rats (250—350 gm) were injected intravenously under light ether anesthesia with 0.4 ml of 99‘Tc-cyclam AK-2123 (12 MBq). The animals were sacrificed at various time intervals under ether anesthesia. The organs of interest were excised, weighed and counted for radioactivity using a Nal (TI) scintillation detector attached to a single channel spectrometer. The amount of 9‘Tc-cyc1am AK-2123 detected in each organ was calculated as per cent administered dose.
- Studies in the Experimental Group of Animals
- Development of Animal Model with Chemical -Induced Mnmmary Tumor
Fifty-day-old Sprague-Dawley rats were injected (i.p.) with 0.5 ml (50-mg) of N-nitroso-N-methylurea (NMU) in a single dose to induce mammary tumors. The carcinogen was freshly prepared in physiological saline, adjusted to pH 4.0 with dilute acetic acid and injected within 20 min as described by Thomson (1991). Animals were examined twice weekly for tumor growth. When the tumor size was more than 20 mm diameter, approx. time (4 months) the animals were subjected to scintigraph imaging studies. Subsequently the tumors were dissected and sent for histological characterization.
- In Vivo Biodistribution and Imaging Studies in Tumor-Bearing Rats
NMU induced mammary tumour-bearing rats were anesthetized by ether and 0.4 ml of ”‘Tc-cyclam AK-2123 (40 MBq) was injected i.v. through the trail-vein. In vivo imaging studies were performed at 5 h post injection by acquiring 600 kilo counts on a low energy all purpose (LEAP) collimator at an energy setting centered at 140 KeV and a 20% window. The radioactivity associated with regions of interest (ROI) over tumor, liver, kidneys, heart, thigh muscle, bladder, intestines, etc., were measured (Ozcan et a1., 1996) at the same pixel count. Then tumor to muscle (T/M) ratios were calculated.
The other radiopharmaceuticals, “‘Tc-(V) DMSA, ”‘Tc-citrate and
20’TICI, were injected into the same animals after the clearance and decay of ”‘Tc-cyclam AK-2123 and T/M ratios were calculated and compared
with those of”‘Tc-cyclam AK-2123 (Table 3). T/M ratio were determined in EMT-6 tumors growing in SCID mice and compared with ‘*5I- iodoazomycin glucopyranoside (IAZG) which is considered to be the optimal hypoxia marker at this time in Dr. Chapman’s laboratory, Fox Chase Cancer Center, Philadelphia USA.
- Measurement of pO2 in Tumor Animal
Intratumor partial oxygen pressures were measured using a blood gas analyzer (Corning). Tumor-bearing rats were anesthetized by ether and tissue fluids were aspirated through a heparinized sterile syringe with 20 gauge needle. The samples were quickly analyzed for p and care was taken to minimize exposure of the samples to air and other oxygen sources.
- Measurement of Octanol-Buffer Distribution Coefficient Octonol-saturated sodium phosphate buffer (0.05 M; pH7.4) and buffer- saturated octanol (2.0 ml each) were placed into a stoppered tube with 105 to 10 dpm of the ”‘Tc-cyclam AK The stoppered tube was vigorously hand shaken for 1.0 min, allowed to settle for 1.0 min and then retaken for an additional 1.0 min. The phases were allowed to settle for 2.0 min. Aliqnots (50/Al) of each phases were counted for radioactivity. The ratio of the dpm octanol to dpm-buffer gave the distribution coefficient.
Results
Chromatographic assay of”‘Tc-cyclam AK-2123 showed a radiochemical purity of greater than 95% and the labeled product was relatively stable for more than 3 h at room temperature. The results of biodistribution studies carri ed out in Wistar rats at different time intervals after administration of radiolabeled cyclam AK-2123 are shown in Table 1. The blood uptake was 2d.7% at 5 min post-injection and fell to 6.1% at 4 h, indicating that this labeled compound cleared tiom the systemic circulation within 4.0 h after administration. Liver uptake was 10.29o at 5 min post-
Table 1. Biodistribution of ””Tc-cyclam AK-2123 in Wistar rats after i,v, administration (= % administration dose/organ at different time periods)
| Organ | 5 min | 0.5 h | th | 2 h | 4 h |
| Blood | 26.67 + 3.63 | 14.13 + 0.91 | 12.73 + 1.85 | 6.99 + 0.50 | 6.14 + 0.58 |
| Muscle | 30.21 + 2.81 | 24.76 + 2.88 | 17.09 ñ 2.34 | 10.81 I 1.35 | 10.54 ñ 1.24 |
| Bone | 2.81 + 0.69 | 1.82 + 0.74 | 2.01 + 0.07 | 1.27 0.22 | 1.42 ñ 0.03 |
| Thyroid | 0.12 + 0.02 | 0.12 + 0.03 | 0.13 + 0.04 | 0.06 + 0.008 | 0.04 + 0.02 |
| Stomach | 1.15 + 0.36 | 1.13 + 0.16 | 1.09 + 0.14 | 1.12 I 0.07 | 1.05 ñ 0.35 |
| Heart | 0.44 + 0.14 | 0.30 + 0.08 | 0.29 ñ 0.07 | 0.15 ñ 0.01 | 0.11 + 0.08 |
| Liver | 10.17 + 2.21 | 7.89 + 0.85 | 5.86 + 0.24 | 5.67+ 0.55 | 6.48 + 0.35 |
| Lungs | 1.47 + 0.43 | 1.20 + 0.10 | 1.10 ñ 0.30 | 0.65 I 0.06 | 0.61 ñ 0.08 |
| S. Intestines | 4.25 I 0.20 | 10.98 + 0.65 | 16.10 + 0.89 | 12.08 + 0.43 | 10.24 I 2.48 |
| L Intestines | 1.39 + 0.44 | 0.94 + 0.15 | 1.28 + 0.36 | 0.93 + 0.36 | 8.07 + 1.57 |
| Kidneys | 10.94 I 0.98 | 9.51 +1.42 | 9.20 Al.27 | 10.41 I 1.02 | 10.54 + 0.36 |
| Urine | 3.15 + 0.001 | 14.99 + 2.49 | 29.96 + 3.25 | 33.66 + 4.55 | 33.67 + 3.76 |
| Remaining
car cass |
57.73 + 6.83 | 52.04 + 2.70 | 45.62 + 3.20 | 30.62 + 2.95 | 28.26 + 2.31 |
ii = 6.
Blood, muscle, bone assumed to be 5%, 40% and 69c of the total body weight, respectively.
9 uptake of urine is cumulative excretion up to the time of sacrifice of the rats.
injection and dropped to d.5% at 4 h. The radioactivity associated with the intestines increased over this time. The peak uptake by small intestine was 16.1% at 1 h which subsequently cleared into the large intestine (8.19 at 4 h). The kidney uptake at 5 min was 10.9% and remained near this level over the 4 h of investigation and the radioactivity in the urine confirmed the renal excretion of this marker.
These observations clearly indicate that the compound is excreted through both hepatobiliary and renal systems. The thyroid uptake was very low (0.12% at 5 min, 0.04% at 4 h) indicating that there was negligible free pertechnetate present in the formulated product.
The stomach uptake remained relatively constant over the 4 h post injection at -1.19c, suggesting that uptake was not due to free pertechnetate.
/n vivo biodistribution studies were also performed in mammary tumor- bearing rats at 5 h post injection using a gamma camera computer system. Scintiimages and T/M ratios obtained are shown in Fig. 1 and Table 2, respectively. Tumor radioactivity was (7.6%) compared to muscle (0.9%) and the T/M ratio was 8.5. Interestingly there was relatively high uptake (1.7%) observed in heart tissue. The same rats were also subjected to in vivo biodistribution studies using other tumor-seeking radiopharmaceuticals. The T/M ratio obtained from these studies are shown in Table 3. The T/M ratio is 8.5, 3.1, 4.5 and 3.0 for ”‘Tc-cyclam AK-2123, ”‘Tc-(V) DMSA, ”‘Tc-citrate and Tl-201, respectively.
The ratios T/M can provide a measure of hypoxic tissues and were
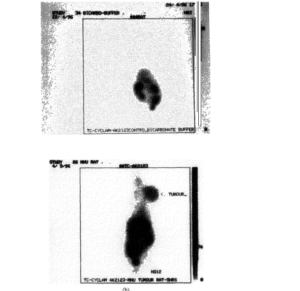
Table 2. Biodistribution study of ””‘Tc-cyclam AK-2123 in NMU-induced mammary tumor-bearing rats at 5 hr post-injection using gamma camera computer system
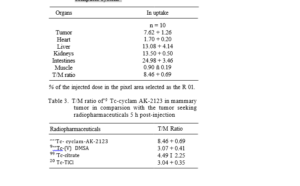
Biogas assays were performed to estimate the oxygen status in MNU induced tumors. Intratumor pO2and oxygen saturation levels measured in these tumors were 20 mmHg and 35to, respectively. The values associated with most oxygenated tissues were 60-80 mmHg and 99-100%, respectively. these results indicate that the average oxygenation of these tumors was much lower than most normal tissues.
Octanol-buffer distribution coefficient(p-value) study with 9‘Tc-cyc1am AK-2123 was carried out and the result obtained is shown in Table 4. The partition coefficient of this marker obtained was 0.1.
Discussion
The existence of hypoxic cells in soild tumors is one of the important limiting factors of treatment by radiation therapy (Gray, 1961). Many factors including deficient tumor vascularization and local microcirculation disturbances contribute to the formation of hypoxic microenvironments within tumors (Sutherland, 1988; Vaupel et a1., 1988). It is widely accepted that the use of a non-invasive method to accurately assess hypoxia within
Table 4. Hypoxic marking properties of nuclear medicine agents in mouse
| IAZA | I-125 | 4.8 | EMT-6/ | > 8 | 6.8 | Mannan |
| BALBC | et aI. 1991 | |||||
| IAZXP | 1-125 | 1.3 | EMT-6 | > 6 | 17.3 | Chapman |
| CB17/ | et a1. 1996 | |||||
| 1cr Scid | ||||||
| IAZG | 1-125 | 0.63 | EMT-6 C | > 4 | 20 | Chapman |
| BIT/ | et al 1996 | |||||
| 1cr Scid | ||||||
| BMS | Tc-99m | 40 | KHT/ | 4—8 | 3.8 | Ballinger |
| 181321 | C3H | et al 1996 | ||||
| BMS | Tc-99m | 12 | KHT/ | 4 | 3.5 | Meloet al. |
| 194796 | C3H | 1997 | ||||
| HL-91
Cyclam |
Tc—99m
Tc-99m |
0.8
0.1 |
EMT-6/
BALB/c EMT-6 |
4
> 4 |
18 |
Mannan
et al. 1955 Present |
| AK 2123 | CB 17/1cr | study | ||||
| Scid |
# — Octonal/water partition co-efficient. Time after marker administration to animals.
tumors would help to improve cancer management. 2-nitroimidazoles are known to be selectively trapped in hypoxic tissues (Linder et al., 1992). Localization of these derivatives in tumors has been non-invasively positron emission tomography (PET) (Rasey et al., 1996) and single photon emission tomography (SPECT) (Urtasum et al., 1996). Misonidazole (MISO) and dimethylmisonidazloe (DEMISO), derivatives of 2-nitroimidazole, are effective radiosensitizers against animal tumor cells in vitro and in vivo, but produced central and peripheral nervous system toxicity in humans. Therefore, there was a need for developing less neurotoxic sensitizers such as etanidazole and nitrotriazole derivatives like AK-2123, the one we used in this study. The AK-2123 is a hypoxic radiosensitizer (Huan and Hua, 1994) with lower in vivo neurotoxicity than misonidazole (MISO). There have been no serious side effects reported (Garcia-Angulo 1919). Therefore, we proposed to label this radiosensitizer with ”‘Tc for possible non-invasive detection of tumour hypoxia. The AK-2123 does not contain chelating moieties and therefore it cannot be directly labelled with ”‘Tc. Cyclam is a molecule with good chelating properties and was employed in our laboratory to synthesis a cyclam porphyrin. We exploited this chemistry expertise to produce a possible hypoxic marker which incorporated AK-2123 as a bioreducible moiety.
In-vivo biodistribution studies were carried out in NMU induced mammary tumor bearing rats at 5 h post-injection (using gamma camera). It showed (Table 3) that tumor to muslce (T/M) ratio obtained was high (8.5). This study demonstrated that these NMU induced mammary tumor may contain hypoxic zones which were further corroborated by low p levels obtained in the same tumor. Hence the radio labelled compound was indicative of hypoxia marker. However it still remains to be proved that there is a dose response relation between degree of hypoxia and concentration of the compound in the tumor.
It was documented by Linder et al. (1992) that the accumulation of BMS 181321 in hypoxic cells is due to enzymatic reduction (possibly by the enzyme xanthine oxidase) of the nitro group to reactive species in the absence of oxygen. The same mode of localization may operate for this agent. However, further work is needed to prove this hypothesis. 9“Tc- cyclam AK-2123 is mainly excreted though the hepatobiliary pathway which is similar to “‘Tc-BMS 181321 (Ballinger et a1., 1996) and therefore the high levels of abdominal activity might interfere with the visualization of tumors within the abdominal region. However, this limitation would be greatly overcome by the use of SPECT imaging procedures.
The maximal T/M ratio (4.2 at 4 h; 3.34 at 6 h) obtained by Ballinger et a1. (1996) is much lower than those obtained by this study (T/M = 8.5 at similar time interval) thereby indicating the superior characteristic of this compound for hypoxia tumor detection. An optimal marker should exhibit high hypoxia specific uptake into cells and larger T/M ratio of specific uptake in tumor models (Chapman et al 1998). Large T/M ratio of (8.5) obtained in this study as compared to conventional tumor seeking radiopharmaceuticals (Table 3) exhibits the characteristics of optimal marker for hypoxia. T/M ratio of ”‘Tc-cyclam AK—2123 (18) was comparable to that of ‘°5I-IAZG (20), thereby confirming that this compound could be hypoxic tumor imaging agent (Table 4).
Moreover an ideal nuclear medicine hypoxia marker should have a partition coefficient (lipid/water solubility) which promotes rapid and nearly equal distributions to body tissues. The properties of various hypoxic marker developed as of date in nuclear medicine are shown in Table 4. An ideal hypoxic marker should have a partition coefficient between 0.01 to
0.1. The partition coefficient of 9“Tc-cyclam AK 2123 obtained in this study was 0.1 which fell in the optimum range of an ideal hypoxic marker and this value is less than that of 251-IAZE (0.63), ”‘Tc-BMS 18321 (40), “‘Tc-BMS 194796 (12) and 9“Tc-HL91 (0.8). T/M ratio of 9“Tc-
cyclam AK-2123 obtained in this study is comparable to those that of ‘251- IAZG and ‘251-IAZXP and much superior or ’251-IAZA (Table 4). These observations indicate that this compound meets the major requirements of an ideal marker which can be proved when it is tested on humans.
Conclusions
This study demonstrates a specific accumulation of ”‘Tc-cycl am AK-2123 into hypoxic tumors. This agent could have potential for detecting hypoxia and predicting radio resistance. It could also be useful in detecting myocardial ischemia and cerebral infarct.
Acknowledgments
The authors thank Dr. Nagraj G. Huilgol, Department of Radiation Oncology, Nanavati Hospital, Mumbai, India; Prof. Kagiya of Japan for Providing a gift sample of AK-2123 used in this study, and also thank Drs. J.D. Chapman and R.F. Schneider, Department of Radiation Oncology, Fox Chase Cancer Centre, Philadelphia, USA for their assistance with the EMT-6 tumor studies.
References
- Ballinger J.R., Kee J.W.M., Rauth A.M. (1956) In vitro and in vivo evaluation of a Technetium-99m-labeled 2-nitroimidazole (BMS 181321) as a marker of tumor hypoxia. J. Nucl. Med. 37, 1023—1031.
2 Chapman J.D. (1979) Hypoxic sensitizers: implications for radiation therapy.
- Eng. J. Med. 301, 1429—1432.
- Chapman D., Franko A.J„ Sharplin J. (1981) A marker for hypoxic cells in tumor with potential clinical applicability. Br. J. Cancer 43, 546—50.
- Chapman D., Coia L.F., Stobbe L.R., Engelhardt E.L., Fenning M.C., Schneider
R.F. (1996) Prediction of tumor hypoxia and radioresistance with nuclear medicine markers. Br. J. Cancer 47, 204—206.
- Chapman J.D., Engelhardt E.L., Stobbe C.C. Schneider R.F., Hanks G.E. (1998) Measuring Hypoxic and predicting tumor radioresistance with nuclear medicine assays. Radiother and Oncol. 46, 229-237.
- Shi Q.Z., Sinusas A.J., Dione D.P., Singer M.J., Young L.H., Heller E.N., Rinker B.D., Wackers F.J.T., Zard B.L. (1995) Technetium-99m-nitroimidazo1e (BMS 1811321) : a positive imaging agent for detecting myocardial ischemia.
- Nucl. Med. 36, 1078—1086.
- DeJong W., Vander Meer P., Nieukoop A.S., Huizer T., Stroeve R.J., Bos E. (1990)Xanthine oxidoreductase activity in perfused hearts of various species, includng humans. Cir. Research 67, 770—773.
- DiRocco J., Kucznski B.L., Pirro J.P., Baer A., Linder K.E., Ramalingam K., Cry J.E., Chan Y.W., Raju N., Narra R.K., Nowotnik D.P, Nunn A.D. (1993) Imaging ischemic tissue at risk of infarction during stoke. J. Cereb Blood Flow Metab 13, 744—762.
- Edwards D.I. (1993) Nitroimidazole drugs—action and resistance Mechanisms of action. J Antimicrob Chemother 31, 9—20.
- Ercan T., Aras T., Unlenen E., Unsal I., Hascelik Z (1993) 9’ ‘Tc-citrate
Radiosensitizer her for Tumor Imaging versus “Ga-citrate for the scintigraph visualization of inflammatory lesions. Nuclear. Med. Biol. 20, 881—887.
- Garcia-Angulo A.H. (1991) Sensitized radiotherapy with both moderate high dose of K. 2123 and external beam radiotherapy combined with hydralazine induced hypoxia. Radiosensitizer Newsletter 10, 56.
- Gerecht M., Chapman J.D. (1983) The labelling of EMT-6 tumors in BALB/ C mice with “C-misonidazole. Br. J. Radiol. 56, 745—753.
- Gray L. (1961) Radiologic basis of oxygen as a modifying factor in radiation therapy. Am J. Roentgenol 85. 803—815.
- Huan L.C., Hua B.Y. (1594) Clinical Pharmacokinetic study and sensitive effect of AK Int. J. Rad. Oncol. Biol. Phys. 29, 607-610.
- Joseph , Jaiswa1A.K., Stobbe C.C., Chapman J.D. (1994) The role of specific reductases in the intracellular activation and binding of 2-nitroimidazoles. Int.
- Radiat. Oncol. Biol. Phys..29, 351—355.
- Koh W.J., Rasey J.S., Evans M.L., Grierson J.R., Lewellen T.K., Graham M.M., Krohn K.A., Griffin W. (1992) Imaging of hypoxia in human tumors with (l8F) fluoromisonidazole Int. J. Rad. Oncol. Biol. Phys. 22, 199—212.
- Linder K.E., Cyr J.E., Chan Y.W., Ragu N., Ramalingam K., Nowotnik D.P. (1992) Chemistry of a Tc-PnAo-nitroimidazole complex that localizes in hypoxic tissue J. Nucl. Med. 33, 919.
- Mannan H., Somayaji V.V., Lee J., Mercer J.R., Chapman J.D., Wiebe L. (1991) Radio iodinated I-(5-todo-5-deoxy-beta-D-arabinofuranosyl)-2- nitroimidazole (lodozomycin arabinoside: IAZA): a novel marker of tissue hypoxia. J. Nucl. Med. 32, 1764—1770.
- Mannan H., Zhang Z., Wiebe L.I., McEwan A.J. (1995) HL—91 : a putative, non-rutro containing agent for hypoxia-specific scintigraphy (abstract # p2l — 314) 43rd Annual Meeting of the Radiation Research Society, San Jose, CA.
- Martin G.V., Caldwell J.H., Graham M.M., Grierson J.R., Kroll K., Cowan M.J., Lewellen T.K., Rasey J.S., Casciari J.J., Kkrohn K.A. (1992) Non- invasive detection of hypoxic myocardium using fluorine- 18-fluoromisinidazo1e and positron emission J. Nucl. Med. 33, 2202—2208.
- Melo T., Ballinger J.R., Rauth A.M. (1997) Comparison of BMS 194796 nitroimidazoles labelled with Technetium-99m as markers for tumour hypoxic, 45th Annual Meeting of the Radiation Research Society. Providence, RI. Abstract P 11—243.
- Nagraj H., Neela C., Mehta A.R. (1996) An overview of the initial experienc with AK-2123 as a hypoxic cell sensitizer with radiation in the treatment of advanced head and neck cancers. Int. J. Radiat. Oncol. Biol. Phys. 34, 1121— 1124.
- Norohna OPD (1983) Solvent extraction technology of 9’Mo-”'”Tc generator system. Performance characteristics. In: radiophramaceuticals and Labeled compounds, Proc of an Int. Conf. (Tokyo), IAEA; 223-232.
- Ohta H., Yamamoto K., Endo , Mori T., Hamanak D., Shimazu A., Ikekubo K., Makimoto K., Lida Y, Konishi J., Morita R., Hata N., Horiuchi K., Yokoyama A., Torizuka K., Kuma K. (19984) A new imaging agent for medullary carcinoma of the thyroid. J. Nucl. Med. 25, 323—325.
- Ozcan Z., Burak Z., Ozcan C., Basdemir G., Ozacar T., Erdem S., Duman Y. (1996) Is T1—201 a reliable agent in tumor imaging? Nucl. Med. Comrnun. 17, 805—809.
- Parliament B., Chapman J.D., Utrasun R.C., McEwan A.J., Goldberg L., Mercer J.R., Mannan R.H., Wiebe L.I., 1992. Non-invasive assessment to a clinical study. Br. J. Cancer 65, 90—95.
- Urtasun C., Parliament M. B., McEwan A.J., Mercer J.R., Mannan R.H., Wiebe L.I., Morin C., Chapman J.D. (1996) Measurement of hypoxic in human tumors by non-invasive SPECT imaging of iodoazomycin arabinoside. Br. J. Cancer 74, S209—212.
- Rasey J.S., Nelson J., Chin L., Evans M.L. Grunbaum Z. (I590) Characteristics of the binding of labelled fluoromisonidazole in cells in vitro. Radiat. Res. 122, 301—308.
- Rasey S., Koh N.J., Evans M.L., et al. (1996) Quantifying regional hypoxia in human tumors with position emission tomography of [18 F] fluoromisonidazole: a pretherapy study of 37 patients Int. J. Radiat. Oncol. Biol. Phys. 36, 417—428.
- Sutherland R.M. (1988) Cell and environmental interactions in tumor microregions: The multicellular spheroid Science 240, 177—184.
- Thompson J., Adlakha H. (1991) Dose responsive induction of mammary gland carcinoma by the intraperitoneal injection of 1 methyl-1-nitrosourea. Cancer Res. 51, 411—3415.
- Tannock I.F. (1972) Oxygen diffusion and the distribution of of cellular radiosensitivity. J. Radiol. 45, 515—524.
- Urtasun C., Koch C.J., Franko A.J., Raleigh J.A., Chapman J.D. (1985) A novel technique for measuring human tissue hypoxia at the cellular level. Br.
- Cancer 54, 453-457.
- Vaupel , Frinak S., Bicher H.I. (1988) Heterogeneous oxygen partial pressure and pH distribution in CMH mouse mammary adenocarcinoma. Cancer Res. 55, 141—146.












![Creating an Audience: Social Implication of Aesthetic Experience[i]](https://sciphiweb.com/wp-content/uploads/2025/03/Creating-an-Audience-Social-Implication-of-Aesthetic-Experience-100x100.jpg)



Leave a Comment
Your email address will not be published. Required fields are marked with *